Global 22q11.2 Deletion Syndrome Market
Global 22q11.2 Deletion Syndrome Market Size, Share, By Diagnostic Method (Chromosomal Microarray Analysis (CMA), Fluorescence In Situ Hybridization (FISH), qPCR & MLPA (Targeted Deletion Testing), Next-Generation Sequencing (NGS), and Prenatal Screening & Diagnostic Testing (NIPT, Amniocentesis, CVS)), By Clinical Manifestation (Congenital Heart Defects, Immunodeficiency, Endocrine Disorders, Developmental & Cognitive Disorders, Neuropsychiatric Disorders, and Palatal Anomalies) By End User (Hospitals & Clinics, Genetic Testing Laboratories, Specialty Care Centers, and Research & Academic Institutes) and By Region (North America, Europe, Asia-Pacific, Latin America, Middle East, and Africa), Analysis and Forecast 2025-2035
Report Overview
Table of Contents
Market Snapshot
- Market Size (2025): USD 649.2 Million
- Projected Market Size (2035): USD 1241.3 Million
- Compound Annual Growth Rate (CAGR): 6.7%
- Largest Regional Market: North America
- Fastest Growing Region: Asia Pacific
- 3rd Largest Region: Europe
- Base Year: 2025
- Historical Period: 2021–2024
- Forecast Period: 2025–2035
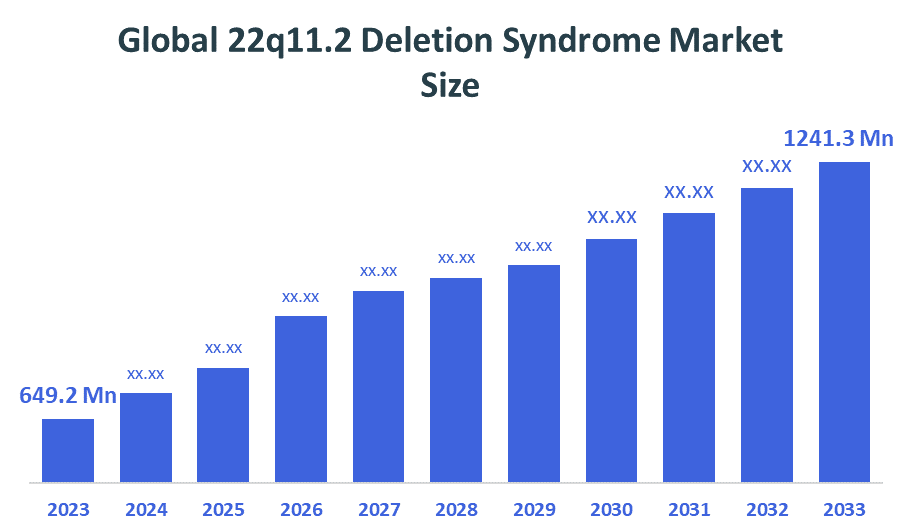
According to Decision Advisors, the Global 22q11.2 Deletion Syndrome Market Size is expected to grow from USD 649.2 million in 2025 to USD 1241.3 million by 2035, at a CAGR of 6.7% during the forecast period 2025-2035. The global 22q11.2 Deletion Syndrome market is driven by increasing adoption of chromosomal microarray analysis for accurate diagnosis, rising neonatal screening for congenital heart defects, growing focus on neuropsychiatric symptom management, and expanding clinical pipelines targeting immunological deficiencies and multisystem complications.
Market Overview/ Introduction
The global 22q11.2 deletion syndrome market refers to the ecosystem of diagnostic, therapeutic, and patient management solutions addressing a rare genetic disorder caused by a microdeletion on chromosome 22. 22q11.2 deletion syndrome, also known as DiGeorge syndrome or velocardiofacial syndrome, is characterized by a wide range of clinical manifestations, including congenital heart defects, immunodeficiency, endocrine disorders, and neuropsychiatric conditions. The market focuses on improving early detection and long-term disease management through advanced medical approaches. The scope of this market includes genetic diagnostic technologies such as chromosomal microarray analysis and next-generation sequencing, along with multidisciplinary care involving cardiology, immunology, and neurology. Increasing emphasis on early diagnosis and continuous monitoring is expanding the treated population globally. Future opportunities lie in the development of targeted therapies for neuropsychiatric and immunological complications, expansion of newborn screening programs, and growing adoption of precision medicine, which are expected to enhance clinical outcomes and drive market growth.
- The National Institutes of Health (NIH) is a leading research authority supporting the 22q11.2 Deletion Syndrome market, with significant federal funding directed toward rare disease research and genomic studies, strengthening innovation in diagnosis and long-term disease management.
- The National Health Service (NHS) in the United Kingdom plays a critical role in the 22q11.2 Deletion Syndrome market by implementing structured care frameworks such as coordinated pediatric-to-adult transition programs, improving patient outcomes and continuity of care.
- The National Organization for Rare Disorders (NORD) supports the 22q11.2 Deletion Syndrome market through patient assistance programs, awareness initiatives, and improved access to diagnosis and treatment, enhancing overall disease management and patient support systems.
Notable Insights: -
- North America holds the largest regional market share of approximately 45% in the global 22q11.2 deletion syndrome market.
- Asia-Pacific is the fastest growing region, accounting for approximately 25% of the global 22q11.2 deletion syndrome market share.
- Europe is the second largest region, accounting for approximately 30% of the global 22q11.2 deletion syndrome market share.
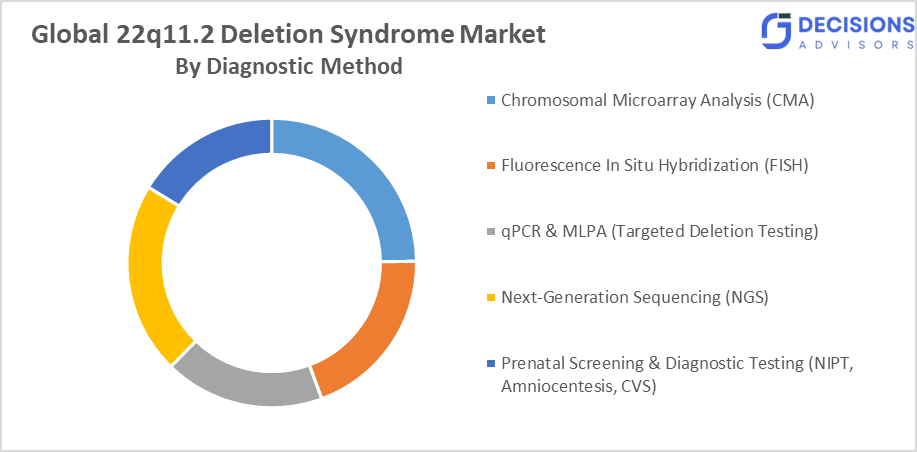
- By diagnostic method, the Chromosomal Microarray Analysis (CMA) segment held a dominant position and is projected to grow at a CAGR of approximately 10.4% during the forecast period.
- By clinical manifestation, the congenital heart defects segment held a dominant position and is anticipated to grow at a CAGR of approximately 9.1% during the forecast period.
- By end user, the hospitals & clinics segment is the dominating segment, accounting for over 60% of the global market share in 2025.
- The compound annual growth rate of the global 22q11.2 deletion syndrome market is 6.7%.
- The market is likely to achieve a valuation of USD 1241.3 million by 2035.
What is role of technology in grooming the market?
Technology is significantly advancing the global 22q11.2 Deletion Syndrome market by improving detection of the 22q11.2 microdeletion and enabling earlier clinical intervention. High-resolution tools such as chromosomal microarray analysis are widely used to accurately identify sub microscopic deletions, which are often missed by conventional methods. Next generation sequencing is further supporting comprehensive genomic profiling in complex cases. Artificial intelligence is being integrated to interpret genetic variations and assist in identifying associated neuropsychiatric risks, including schizophrenia and developmental disorders. In addition, digital health platforms enable continuous monitoring of multisystem complications such as cardiac defects and immune dysfunction. These technological advancements are enhancing diagnostic efficiency, supporting personalized care, and strengthening long term disease management outcomes.
How is Recent Developments Helping the Market?
Recent developments are directly strengthening the 22q11.2 deletion syndrome market by expanding both diagnostic and therapeutic opportunities. Advancements in pipeline therapies targeting neuropsychiatric symptoms are creating a new revenue stream beyond traditional diagnostics, enabling the market to transition toward treatment-driven growth. At the same time, increased adoption of high-precision genetic testing is improving early detection rates, significantly expanding the diagnosed patient population and boosting demand for testing services. Updated clinical guidelines and structured care frameworks are further standardizing diagnosis and long-term disease management, increasing patient retention across healthcare systems. Collectively, these developments are enhancing commercialization potential, increasing healthcare spending per patient, and driving sustained market expansion.
Market Drivers
The global 22q11.2 deletion syndrome market is driven by the increasing clinical reliance on chromosomal microarray analysis as the primary diagnostic tool for detecting 22q11.2 microdeletions, enabling early and accurate identification of the condition. The high incidence of congenital heart defects in affected patients is prompting routine neonatal screening, directly increasing diagnostic volumes. Rising clinical focus on managing neuropsychiatric manifestations such as schizophrenia, anxiety, and developmental disorders is driving demand for targeted therapies under development. Additionally, growing adoption of multidisciplinary care models in hospitals is increasing long-term patient management. Expanding clinical pipelines, particularly for immunodeficiency and thymus-related therapies, are further accelerating market growth by addressing critical unmet medical needs specific to this syndrome.
Restrain
The global 22q11.2 deletion syndrome market is restrained by limited availability of disease-specific therapies, with most treatments focusing on symptom management rather than targeted cures. High cost of advanced genetic testing and underdiagnosis in low-resource settings further restrict early detection and comprehensive patient management across regions.
Competitive Analysis:
The report offers the appropriate analysis of the key organizations/companies involved within the global 22q11.2 deletion syndrome market, along with a comparative evaluation primarily based on their product of offering, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborative analysis focusing on the current news and developments of the companies, which includes product development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
Top Companies in Global 22q11.2 Deletion Syndrome Market
- Zynerba Pharmaceuticals
- Nobias Therapeutics
- Sumitomo Pharma
- Enzyvant
- Roivant Sciences
- Bausch Health Americas
Government Initiatives
|
Country |
Key Government Initiatives |
|
UK |
The UK government, through the National Health Service, has implemented structured care frameworks such as coordinated pediatric-to-adult transition programs to improve management of complex genetic disorders like 22q11.2 deletion syndrome. |
|
US |
The government supports rare disease research through strong funding programs led by federal agencies, while initiatives promoting newborn screening and genetic testing improve early diagnosis and long-term disease management. |
|
China |
China is strengthening its healthcare system through national health reforms, increasing investment in genetic testing infrastructure, and improving access to early diagnosis for rare genetic disorders. |
|
Germany |
The government supports rare disease management through advanced healthcare policies and funding for genetic research, alongside the adoption of standardized clinical guidelines to improve diagnosis and treatment outcomes. |
|
Japan |
The Japanese government promotes rare disease research and precision medicine initiatives, focusing on early detection of genetic and neuropsychiatric conditions through expanding access to advanced diagnostic technologies. |
Market Segmentation
The global 22q11.2 deletion syndrome market share is classified into diagnostic method, clinical manifestation, and end user.
- The Chromosomal Microarray Analysis (CMA) segment dominated the market in 2025, and is projected to grow at a substantial CAGR of approximately 10.4% during the forecast period.
Based on the diagnostic method, the global 22q11.2 deletion syndrome market is divided into chromosomal microarray analysis (CMA), fluorescence in situ hybridization (FISH), qPCR & MLPA (Targeted Deletion Testing), next-generation sequencing (NGS), and prenatal screening & diagnostic testing (NIPT, Amniocentesis, CVS). Among these, the chromosomal microarray analysis (CMA) segment dominated the market in 2025, and is projected to grow at a substantial CAGR of approximately 10.4% during the forecast period. It is because CMA offers high-resolution genome-wide detection of microdeletions, making it the gold standard for diagnosing 22q11.2 deletion syndrome. Its increasing clinical adoption, accuracy, and ability to detect atypical deletions drive its dominance, supported by growing awareness and expanding access to advanced genetic testing technologies.
- The congenital heart defects segment dominated the market in 2025, and is anticipated to grow at a significant CAGR of approximately 9.1% during the forecast period.
Based on the clinical manifestation, the global 22q11.2 deletion syndrome market is divided into congenital heart defects, immunodeficiency, endocrine disorders, developmental & cognitive disorders, neuropsychiatric disorders, and palatal anomalies. Among these, the congenital heart defects segment dominated the market in 2025, and is anticipated to grow at a significant CAGR of approximately 9.1% during the forecast period. This dominance is driven by the high prevalence of cardiac abnormalities in affected patients, leading to early diagnosis and immediate medical intervention. Increased screening in neonates and advancements in pediatric cardiology further contribute to segment growth.
- The hospitals & clinics segment dominated the market in 2025, accounting for approximately 60% of the total market share, and is projected to grow at a substantial CAGR during the forecast period.
Based on the end user, the global 22q11.2 deletion syndrome market is divided into hospitals & clinics, genetic testing laboratories, specialty care centers, and research & academic institutes. Among these, the hospitals & clinics segment dominated the market in 2025, accounting for approximately 60% of the total market share, and is projected to grow at a substantial CAGR during the forecast period. This is attributed to the availability of integrated diagnostic and treatment facilities, higher patient inflow, and access to multidisciplinary care including cardiology, immunology, and genetic counseling, making hospitals the primary point of diagnosis and management.
What is the Reason of the Region Dominance?
The dominance of certain regions in the global 22q11.2 deletion syndrome market is primarily driven by advanced healthcare infrastructure, high adoption of genetic testing technologies, and strong awareness regarding rare genetic disorders. Regions such as North America lead the market due to early diagnosis, widespread availability of chromosomal microarray testing, and favorable reimbursement policies. The presence of well-established diagnostic laboratories, skilled healthcare professionals, and strong research funding further supports market growth. Additionally, increasing implementation of newborn screening programs and growing focus on precision medicine contribute significantly to regional dominance.
Strategies to Implement for Growth of the Market in Non-Leading Regions
Growth in non-leading regions can be supported through strategic investments in healthcare infrastructure, expansion of genetic testing capabilities, and increasing awareness of rare genetic disorders. Improving access to affordable diagnostic technologies such as CMA and NGS can significantly enhance early detection rates. Collaborations with local healthcare providers, government bodies, and research institutions can strengthen diagnostic networks and treatment accessibility. Expanding telehealth and genetic counseling services can further improve patient reach in remote areas. Additionally, awareness campaigns, training programs for healthcare professionals, and supportive government policies can drive market penetration and long-term growth.
Regional Segment Analysis of the 22q11.2 Deletion Syndrome Market
- North America (U.S., Canada, Mexico)
- Europe (Germany, France, U.K., Italy, Spain, Rest of Europe)
- Asia-Pacific (China, Japan, India, Rest of APAC)
- South America (Brazil and the Rest of South America)
- The Middle East and Africa (UAE, South Africa, Rest of MEA)
North America is anticipated to hold the largest share of the global 22q11.2 deletion syndrome market over the predicted timeframe. North America is anticipated to hold the largest share of the global 22q11.2 deletion syndrome market over the predicted timeframe. This market is accounting for approximately 45% of the total market share, driven by advanced healthcare infrastructure, high adoption of genetic testing technologies, and strong awareness regarding rare genetic disorders. The presence of well-established diagnostic laboratories, early screening programs, and favorable reimbursement frameworks further supports market dominance. Additionally, increasing research funding and availability of advanced genomic technologies contribute significantly to the region’s leadership position.
Asia-Pacific is expected to grow at a rapid CAGR in the global 22q11.2 deletion syndrome market during the forecast period. Asia-Pacific is expected to grow at a rapid CAGR in the global 22q11.2 Deletion Syndrome market during the forecast period. This region holds approximately 25% of the total market share and is driven by a large population base, rising healthcare expenditure, and increasing awareness of genetic disorders. Rapid improvements in healthcare infrastructure and expanding access to genetic testing in countries such as China and India are further accelerating market growth.
Europe is the second largest region in the global 22q11.2 deletion syndrome market. Europe is the second largest region in the global 22q11.2 deletion syndrome market, accounting for approximately 30% of the total market share. The region’s growth is driven by strong government support for rare disease research, well-established healthcare systems, and increasing implementation of genetic screening programs. Countries such as Germany, France, and the United Kingdom play a key role due to high healthcare spending and advanced diagnostic capabilities.
Future Market Trends in Global 22q11.2 Deletion Syndrome Market: -
- Increasing Adoption of High-Precision Genetic Testing
The market is witnessing strong momentum toward advanced diagnostic technologies that enable earlier and more accurate detection of chromosomal abnormalities. Improved accessibility and declining testing costs are further expanding the diagnosed patient population and supporting market growth.
- Shift Toward Personalized and Multisystem Care Management
Treatment approaches are evolving from generalized care to personalized management addressing multiple clinical manifestations, including cardiac, immunological, and neuropsychiatric conditions. This shift is increasing demand for integrated care models and specialized treatment pathways.
- Expansion of Early Screening and Awareness Programs
Rising awareness among healthcare professionals and growing implementation of prenatal and neonatal screening programs are driving early diagnosis. This trend is enhancing timely intervention, improving patient outcomes, and strengthening long-term demand across diagnostic and healthcare services.
Recent Development
- March 2026, Nobias Therapeutics continued Phase 2 clinical evaluation of NB-001, a non-stimulant mGluR-targeting therapy, focusing on anxiety and ADHD-related manifestations, reflecting increasing innovation in symptom-specific treatment approaches.
- January 2026, Harmony Biosciences progressed ZYN002 (Zygel), a transdermal cannabidiol gel, toward late-stage clinical development for neuropsychiatric symptoms associated with 22q11.2 deletion syndrome, strengthening the targeted rare disease therapy pipeline.
- August 2025, The American Academy of Pediatrics released updated clinical guidelines for 22q11.2 deletion syndrome, establishing a structured surveillance framework that supports early diagnosis and improved long-term patient management.
- April 2025, Industry focus on orphan drug development for 22q11.2 deletion syndrome intensified, with companies leveraging regulatory incentives such as market exclusivity and accelerated approval pathways to expand rare disease therapeutic pipelines.
Market Segment
This study forecasts revenue at global, regional, and country levels from 2020 to 2035. Decision Advisor has segmented the global 22q11.2 deletion syndrome market based on the below-mentioned segments:
Global 22q11.2 Deletion Syndrome Market, By Diagnostic Method
- Chromosomal Microarray Analysis (CMA)
- Fluorescence In Situ Hybridization (FISH)
- qPCR & MLPA (Targeted Deletion Testing)
- Next-Generation Sequencing (NGS)
- Prenatal Screening & Diagnostic Testing (NIPT, Amniocentesis, CVS)
Global 22q11.2 Deletion Syndrome Market, By Clinical Manifestation
- Congenital Heart Defects
- Immunodeficiency
- Endocrine Disorders
- Developmental & Cognitive Disorders
- Neuropsychiatric Disorders
- Palatal Anomalies
Global 22q11.2 Deletion Syndrome Market, By End User
- Hospitals & Clinics
- Genetic Testing Laboratories
- Specialty Care Centers
- Research & Academic Institutes
Global 22q11.2 Deletion Syndrome Market, By Regional Analysis
- North America
- US
- Canada
- Mexico
- Europe
- Germany
- UK
- France
- Italy
- Spain
- Russia
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- Australia
- Rest of Asia Pacific
- South America
- Brazil
- Argentina
- Rest of South America
- Middle East & Africa
- UAE
- Saudi Arabia
- Qatar
- South Africa
- Rest of the Middle East & Africa
Frequently Asked Questions (FAQ)
Q: What makes patient identification challenging in the 22q11.2 deletion syndrome market?
A: The condition presents with highly variable symptoms affecting cardiac, immune, and neuropsychiatric systems, often leading to misdiagnosis or delayed diagnosis. This increases dependence on advanced genetic testing methods and specialized clinical evaluation for accurate identification.
Q: How does lifelong disease management impact the market structure?
A: Patients require continuous, multidisciplinary care across cardiology, immunology, and psychiatry, creating long-term demand for healthcare services. This sustained engagement increases overall healthcare spending per patient and strengthens recurring revenue streams within the market.
Q: Why is the market shifting beyond diagnostics toward therapeutic development?
A: Growing clinical focus on managing neuropsychiatric and immunological complications is driving investment in targeted therapies. This shift is expanding the market from being primarily diagnosis-driven to a more treatment-oriented ecosystem with higher growth potential.
Q: How do healthcare infrastructure gaps affect market expansion?
A: Limited access to advanced genetic testing and specialized care in developing regions leads to underdiagnosis and delayed treatment. This restricts market penetration and highlights the need for improved healthcare infrastructure and diagnostic accessibility globally.
Check Licence
Choose the plan that fits you best: Single User, Multi-User, or Enterprise solutions tailored for your needs.
We Have You Covered
- 24/7 Analyst Support
- Clients Across the Globe
- Tailored Insights
- Technology Tracking
- Competitive Intelligence
- Custom Research
- Syndicated Market Studies
- Market Overview
- Market Segmentation
- Growth Drivers
- Market Opportunities
- Regulatory Insights
- Innovation & Sustainability
Report Details
| Scope | Global |
| Pages | 240 |
| Delivery | PDF & Excel via Email |
| Language | English |
| Release | Mar 2026 |
| Access | Download from this page |
