Global Achondroplasia Market
Global Achondroplasia Market Size, Share, By Type (Surgical Treatment, Pharmacological Treatment, Physical Therapy), By Diagnosis Method (Genetic Testing, Physical Examination, Imaging Techniques), By End User (Hospitals, Specialty Clinics, Home Care Settings), and By Region (North America, Europe, Asia-Pacific, Latin America, Middle East, and Africa), Analysis and Forecast 2025 ? 2035.
Report Overview
Table of Contents
Market Snapshot
- Market Size (2025): USD 148.6 Million
- Projected Market Size (2035): USD 675.1 Million
- Compound Annual Growth Rate (CAGR): 16.34%
- Largest Regional Market: North America
- Fastest Growing Region: Asia Pacific
- 2nd Largest Region: Europe
- Base Year: 2025
- Historical Period: 2021–2024
- Forecast Period: 2025–2035
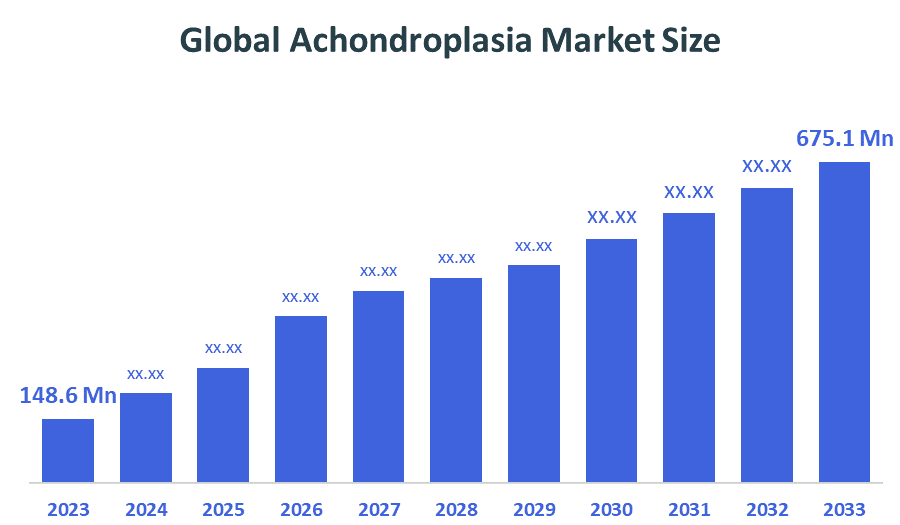
According to Decision Advisors, the Global Achondroplasia Market Size is expected to grow from USD 148.6 million in 2025 to USD 675.1 million by 2035, at a CAGR of 16.34% during the forecast period 2025-2035. The global achondroplasia market is driven by the growing use of treatments that target specific diseases results in more patients receiving these specialized therapies, which change their disease progression. The most important treatment innovation is vosoritide, which acts as a C-type natriuretic peptide (CNP) analog that prevents excessive FGFR3 activity to enable normal bone development.
Market Overview/ Introduction
The achondroplasia market refers to the global industry focused on the development, production, and commercialization of therapies, diagnostics, and supportive care for achondroplasia, a rare genetic disorder affecting bone growth and causing disproportionate dwarfism. The process includes all research and development activities, regulatory approval procedures and construction of healthcare facilities, which together work to enhance diagnosis capabilities and treatment effectiveness and the quality of life of patients. The achondroplasia market is also affected by growing public awareness and market advocacy activities. Organizations dedicated to supporting individuals with achondroplasia are actively promoting education and resources, which may contribute to a more informed public and healthcare community.
The PROPEL 3 trial design received positive feedback from the United States Food and Drug Administration FDA and the European Union European Medicines Agency EMA because they considered it to be suitable as a registrational study, which would support a marketing application.
BioMarin Pharmaceutical Inc. announced in October 2023 that the U.S. Food and Drug Administration (FDA) had approved its supplemental New Drug Application (sNDA) for VOXZOGO (vosoritide), which helps treat achondroplastic pediatric patients with open epiphyses who need to increase their linear growth.
- The U.S. Food and Drug Administration (FDA) has approved Yuviwel (navepegritide) for injection to improve growth in pediatric patients aged two years and older with achondroplasia.
- BioMarin Pharmaceutical, Voxzogo generated an increase in the revenue share between 900 million and 935 million in 2025.
- Tyra Biosciences Inc. develops advanced precision treatments that target extensive research possibilities in the Fibroblast Growth Factor Receptors study. The company will expand its research on
- TYRA-300 to treat achondroplasia after it received positive preclinical results from a study that it conducted with the Imagine Institute, located in Paris, France.
Notable Insights: -
- North America holds the largest regional market share, approximately 45.2% in the global achondroplasia market.
- Asia Pacific is the fastest-growing region in the global achondroplasia market.
- By type, the pharmacological treatment segment held a dominant position in market share in 2025.
- By diagnosis method, the genetic testing segment is the dominating one in global market share in 2025.
- The compound annual growth rate of the global achondroplasia market is 16.34%.
- The market is likely to achieve a valuation of USD 675.1 million by 2035.
What is the role of technology in grooming the market?
Technology plays a crucial role in shaping the growth of the achondroplasia market by enhancing diagnosis, treatment, and patient management. Advanced genetic testing technologies can detect FGFR3 mutations with complete precision because they operate through all stages of fetal development, which results in better patient outcomes. The field of biotechnology has created targeted therapies through its research innovations, which developed CNP analogs to treat the disease because they treat its root causes instead of only its visible signs. The combination of digital health tools with telemedicine platforms helps to enhance specialist care accessibility for patients who live in remote areas and locations with limited medical facilities. The combination of artificial intelligence (AI) and data analytics enables organizations to optimize their clinical trial processes while discovering new patient groups and making their drug development process more efficient. Imaging technologies provide better capabilities to observe bone growth patterns and how patients respond to their treatment. The medical field uses technology to advance precise medicine because it creates new treatment methods that decrease patient treatment times while making healthcare services more available, which drives the worldwide growth of the achondroplasia market.
Market Drivers
The achondroplasia market is driven by the scholarship about rare genetic disorders, which has increased because diagnostic methods have progressed, especially through genetic testing. The development of targeted therapies, which include CNP analogs has become the standard treatment method. The development of rare disease research receives additional support from government orphan drug programs and their financial backing. The market expansion receives support from three factors, which include rising healthcare costs, better treatment access in developing countries and progress in biotechnology.
In January 2025, BridgeBio Pharma, Inc., a new type of biopharmaceutical company focused on genetic diseases, provided updates on its commercial progress for Attruby (acoramidis), the status of late-stage pipeline programs, and anticipated 2025 milestones.
Restrain
The achondroplasia market is hindered due to high treatment costs and relapse risks, which create obstacles to market development. The global healthcare system faces challenges because there are not enough qualified specialists to deliver optimal care to patients. The healthcare system in developing regions faces three major challenges, which include restricted access to approved medications, a need for continuous psychotherapy, and limited mental health diagnosis capabilities.
Competitive Analysis:
The report offers the appropriate analysis of the key organizations/companies involved within the achondroplasia market, along with a comparative evaluation primarily based on their product of offering, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborative analysis focusing on the current news and developments of the companies, which includes product development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
Top Companies in Global Achondroplasia Market
- BioMarin Pharmaceutical Inc.
- Ascendis Pharma A/S
- Pfizer Inc.
- Roche Holding AG
- Novartis AG
- Ipsen S.A.
- QED Therapeutics
- OPKO Health Inc.
- Novo Nordisk A/S
- Sanofi S.A.
Government Initiatives
|
Country |
Key Government Initiatives |
|
UK |
The UK government drives growth through the Rare Diseases Framework and England Rare Diseases Action Plan 2026, focusing on faster diagnosis, improved access to therapies, and streamlined clinical trials. Regulatory reforms aim to reduce approval timelines and enhance patient access to innovative treatments. |
|
India |
India is gradually strengthening its rare disease framework through national policies and funding programs. However, limited financial support, high treatment costs, and reliance on external funding remain challenges. Increasing awareness and policy focus are expected to improve access and market growth in the coming years. |
|
China |
China is expanding its rare disease ecosystem through National Rare Disease Lists, treatment guidelines (including achondroplasia), and a national diagnosis network. Inclusion of rare disease drugs in reimbursement lists and policy reforms are improving access, though broader legislation is still evolving. |
Market Segmentation
The achondroplasia market share is classified into type, diagnosis method, and end user.
- The pharmacological treatment segment dominated the market in 2025, and is projected to grow at a substantial CAGR during the forecast period.
Based on type, the achondroplasia market is divided into surgical treatment, pharmacological treatment, physical therapy. Among these, the pharmacological treatment segment dominated the market in 2025 and is projected to grow at a substantial CAGR during the forecast period. The growth of the segment is driven by the growing usage of specific drug treatments that target diseases through CNP analogs which include vosoritide. The treatments provide permanent solutions because they treat the root genetic problem while delivering prolonged advantages and decreasing medical problems, which makes them more acceptable than surgical procedures and physical therapy treatment methods.
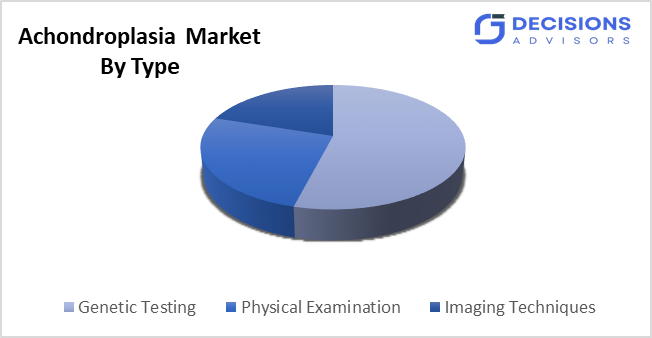
- The genetic testing segment accounted for the largest share in 2025, and is anticipated to grow at a significant CAGR during the forecast period.
Based on the diagnosis method, the achondroplasia market is divided into genetic testing, physical examination, imaging techniques. Among these, the genetic testing segment accounted for the largest share in 2025 and is anticipated to grow at a significant CAGR during the forecast period. The system achieves precise diagnosis through its ability to detect FGFR3 gene mutations, which provide results that exceed the accuracy of physical examinations and imaging tests. The combination of advanced molecular diagnostics becoming more widely used, the ability to detect conditions during prenatal development and the healthcare providers' increasing understanding of these developments leads to the system's dominant position in clinical settings and the growing demand for its use.
- The hospitals segment dominated the market in 2025, and is projected to grow at a substantial CAGR during the forecast period.
Based on the end user, the achondroplasia market is divided into hospitals, specialty clinics, home care settings. Among these, the hospitals segment dominated the market in 2025 and is projected to grow at a substantial CAGR during the forecast period. The segmental growth is driven by the system's main services, which include specialized diagnostic tools, multidisciplinary medical care and advanced treatment methods, along with surgical procedures. The combination of complex condition requirements and ongoing treatment needs makes hospital facilities the preferred choice for patients and healthcare staff members who need complete treatment services.
Regional Segment Analysis of the Achondroplasia Market
- North America (U.S., Canada, Mexico)
- Europe (Germany, France, U.K., Italy, Spain, Rest of Europe)
- Asia-Pacific (China, Japan, India, Rest of APAC)
- South America (Brazil and the Rest of South America)
- The Middle East and Africa (UAE, South Africa, Rest of MEA)
North America is anticipated to hold the largest share of the achondroplasia market over the predicted timeframe.
North America is anticipated to hold the largest share of the achondroplasia market over the predicted timeframe. North America dominates the market with approximately 45.2% share in 2024. The region operates under governance that depends on three factors: its advanced healthcare system, its dedication to rare disease research funding, and its capacity to implement new treatment methods, CNP analogues. The organization benefits from three factors, which include favorable regulatory conditions, high public awareness, and the presence of major pharmaceutical companies. North America serves as the leading global market because the United States conducts numerous clinical trials and provides superior reimbursement systems.
Asia Pacific is expected to grow at a rapid CAGR in the achondroplasia market during the forecast period. This region is projected to expand at a CAGR of around 8.7%, driven by strong healthcare investments and increasing awareness of rare genetic disorders. Asia-Pacific accounts for approximately 21–21.5% of the global market share (2024) and is expected to grow rapidly compared to North America and Europe. The patient population size in China and India, with better diagnostic methods and government programs create growth for the industry. The regional market expansion of APAC will experience acceleration from three factors, which include better healthcare infrastructure, increased pharmaceutical investments and wider access to genetic testing.
Europe is the 2nd largest region to grow in the achondroplasia market during the period. Europe is the second-largest region in the Achondroplasia market during the forecast period. The region becomes better because it has modern healthcare systems and strong regulatory systems, and people have grown their knowledge about rare diseases. The market experiences continual growth because countries such as Germany, France and the UK adopt new therapeutic methods and increase their research efforts.
Strategies to Implement for Growth of the Market in Non-Leading Regions
To drive growth of the Achondroplasia market in non-leading regions, companies should focus on expanding awareness and improving diagnosis through education programs for healthcare professionals and patients. Strengthening access to genetic testing and early screening results in better outcomes through faster medical intervention. Distribution networks become more efficient through partnerships between local healthcare providers and government organizations. Companies should use tiered pricing models and orphan drug incentives to make their treatments more accessible to patients. Local clinical trials and regulatory approval processes need funding because they help products reach the market faster. Healthcare systems need better infrastructure, with increased telemedicine use, so they can effectively manage patients who live in rural regions. Patient advocacy organizations and non-governmental organizations should work together to raise awareness and eliminate stigma while improving treatment uptake in developing markets.
Future Market Trends in Global Achondroplasia Market: -
1. Rise of Targeted Therapies
The market is shifting toward disease-modifying treatments such as CNP analogs (e.g., vosoritide) that target the FGFR3 pathway. These therapies aim to address the root cause rather than symptoms, significantly improving patient outcomes and driving long-term market growth.
2. Growth of Gene and Precision Medicine
Emerging gene-editing technologies and precision medicine approaches are expected to transform treatment. Companies are focusing on therapies that directly modify genetic mutations, offering potential long-term or permanent solutions for achondroplasia.
3. Increasing R&D and Clinical Trials
Rising investment in research and development and a strong pipeline of innovative drugs are accelerating market expansion. Multiple therapies are in clinical stages, and successful trials are expected to introduce new treatment options in the coming years.
Recent Development
In February 2026, Ascendis Pharma A/S announced that the U.S. Food & Drug Administration (FDA) approved under the FDA’s Accelerated Approval Program for YUVIWEL, the first and only once-weekly treatment indicated to increase linear growth in children 2 years of age and older with achondroplasia with open epiphyses and the only one to provide continuous systemic exposure to CNP over the weekly dosing interval.
In September 2024, BridgeBio Pharma, Inc., a commercial-stage biopharmaceutical company focused on genetic diseases, announced that the FDA had granted Breakthrough Therapy Designation to oral infigratinib under development for children with achondroplasia.
In February 2024. BridgeBio Pharma, Inc. and Kyowa Kirin Co., Ltd announced a partnership wherein BridgeBio’s affiliate, QED Therapeutics, grants Kyowa Kirin an exclusive license to develop and commercialise infigratinib for achondroplasia, hypochondroplasia, and other skeletal dysplasias in Japan
How is Recent Developments Helping the Market?
Market Segment
This study forecasts revenue at global, regional, and country levels from 2020 to 2035. Decision Advisors has segmented the achondroplasia market based on the below-mentioned segments:
Global Achondroplasia Market, By Type
- Surgical Treatment
- Pharmacological Treatment
- Physical Therapy
Global Achondroplasia Market, By Diagnosis Method
- Genetic Testing
- Physical Examination
- Imaging Techniques
Global Achondroplasia Market, By End User
- Hospitals
- Specialty Clinics
- Home Care Settings
Global Achondroplasia Market, By Regional Analysis
- North America
- US
- Canada
- Mexico
- Europe
- Germany
- UK
- France
- Italy
- Spain
- Russia
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- Australia
- Rest of Asia Pacific
- South America
- Brazil
- Argentina
- Rest of South America
- Middle East & Africa
- UAE
- Saudi Arabia
- Qatar
- South Africa
- Rest of the Middle East & Africa
Frequently Asked Questions (FAQ).
Q: What role does genetic testing play in the achondroplasia market?
A: Genetic testing plays a critical role by enabling accurate and early diagnosis through detection of FGFR3 gene mutations. It supports prenatal screening and helps clinicians plan timely interventions. Increasing adoption of advanced molecular diagnostics is boosting demand and strengthening its position within the overall Achondroplasia market.
Q: Who are the key players in the achondroplasia market?
A: Key players include BioMarin Pharmaceutical, Ascendis Pharma, Pfizer, Roche, Novartis, Ipsen, and Novo Nordisk. These companies focus on developing targeted therapies and expanding their product pipelines. Strategic collaborations, clinical trials, and regulatory approvals are central to their efforts in strengthening global market presence.
Q: How is technology influencing the achondroplasia market?
A: Technology is improving diagnosis through advanced genetic testing and enhancing treatment with targeted therapies. AI and data analytics support drug discovery and clinical trials, while telemedicine expands access to specialists. These advancements are enabling precision medicine, improving patient outcomes, and accelerating innovation in the Achondroplasia market.
Q: What is the role of clinical trials in this market?
A: Clinical trials are essential for developing and validating new therapies for achondroplasia. They help assess safety and efficacy, leading to regulatory approvals. Increasing investment in clinical research and a strong pipeline of innovative drugs are accelerating market growth and expanding available treatment options.
Check Licence
Choose the plan that fits you best: Single User, Multi-User, or Enterprise solutions tailored for your needs.
We Have You Covered
- 24/7 Analyst Support
- Clients Across the Globe
- Tailored Insights
- Technology Tracking
- Competitive Intelligence
- Custom Research
- Syndicated Market Studies
- Market Overview
- Market Segmentation
- Growth Drivers
- Market Opportunities
- Regulatory Insights
- Innovation & Sustainability
Report Details
| Scope | Global |
| Pages | 210 |
| Delivery | PDF & Excel via Email |
| Language | English |
| Release | Apr 2026 |
| Access | Download from this page |
