Global Advanced Hepatocellular Carcinoma with CPB Liver Cirrhosis Market
Global Advanced Hepatocellular Carcinoma with CPB Liver Cirrhosis Market Size, Share, By Type (Gin, Brandy, Cane, Vodka, Flavoured Advanced Hepatocellular Carcinoma with CPB Liver Cirrhosis, Rum, Natural Advanced Hepatocellular Carcinoma with CPB Liver Cirrhosis, Whiskey, and Tequila) By Distribution Channel (Supermarkets and Hypermarkets, On-Trade, Specialist Retailers, Online Convenience Stores, and Others) By Alcohol Volume (Below 40%, 40% to 60%, and Above 60%) and By Region (North America, Europe, Asia-Pacific, Latin America, Middle East, and Africa), Analysis and Forecast 2025-2035
Report Overview
Table of Contents
Market Snapshot
- Market Size (2025): USD 732.61 Million
- Projected Market Size (2035): USD 5243.7 Million
- Compound Annual Growth Rate (CAGR): 21.75%
- Largest Regional Market: North America
- Fastest Growing Region: Asia Pacific
- 3rd Largest Region: Europe
- Base Year: 2025
- Historical Period: 2021–2024
- Forecast Period: 2025–2035
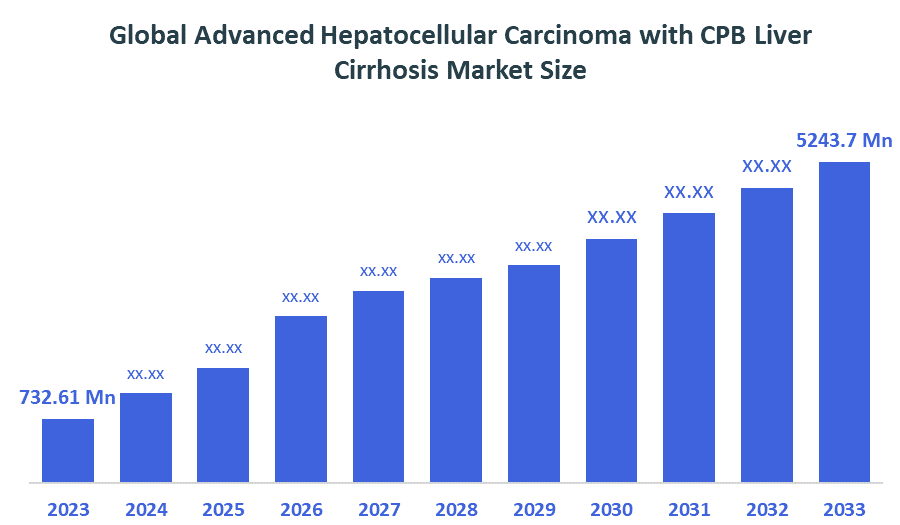
According to Decision Advisors, the Global Advanced Hepatocellular Carcinoma with CPB Liver Cirrhosis Market Size is expected to grow from USD 732.61 million in 2025 to USD 5243.7 million by 2035, at a CAGR of 21.75% during the forecast period 2025-2035. The global advanced hepatocellular carcinoma with CPB liver cirrhosis market focuses on diagnosis and therapies for patients with impaired liver function. Rising liver disease prevalence, early screening, expanding healthcare infrastructure, and adoption of systemic and combination therapies drive market growth worldwide.
Market Overview/ Introduction
The global advanced hepatocellular carcinoma with CPB (Child-Pugh B) liver cirrhosis market involves diagnosis, treatment, and management of liver cancer in patients with moderately impaired liver function. HCC is the most common primary liver cancer, often arising in cirrhotic livers, and CPB cirrhosis indicates compromised liver function with moderate symptoms, including jaundice, ascites, and impaired synthetic function. Patients at this stage are high-risk and require specialized systemic, targeted, or combination therapies. The market scope includes diagnostic solutions such as imaging and biomarker tests, and therapeutic interventions including systemic therapies, targeted therapies, immunotherapies, and supportive care. It covers hospitals, specialty oncology clinics, and cancer research institutes as primary end users. Increasing prevalence of hepatitis B and C, rising liver cirrhosis cases, and improved healthcare access across developed and emerging regions contribute to market expansion. Future opportunities lie in novel combination therapies, immunotherapy advancement, precision medicine, and expansion of early-detection programs, enhancing treatment adoption and improving patient outcomes globally.
- The US National Cancer Institute (NCI) supports multiple research programs for advanced hepatocellular carcinoma (HCC) with Child?Pugh B cirrhosis, funding clinical trials, combination therapy studies, and novel drug development, strengthening the global treatment pipeline and improving patient outcomes.
- Ayushman Bharat, Pradhan Mantri Jan Arogya Yojana (PM?JAY) in India provides cashless treatment for advanced HCC, including surgery, chemotherapy, and targeted therapies, increasing accessibility for economically vulnerable populations and expanding treatment adoption in domestic and regional markets.
- China’s 15th Five-Year Plan emphasizes oncology research, upgraded free trade zones, and better intellectual property protection, indirectly supporting the growth of advanced HCC treatment access and adoption of innovative therapies in both domestic and international markets.
- Merck & Co., Inc. is a leading global company in advanced HCC therapies, with its portfolio including pembrolizumab and other investigational agents, strengthening its position in the global market and driving ongoing clinical research for CPB cirrhosis patients.
- Rashtriya Arogya Nidhi (RAN) in India provides financial support to low-income patients for expensive cancer therapies, facilitating uptake of systemic, targeted, and combination therapies in advanced HCC with CPB cirrhosis.
Notable Insights: -
- North America holds the largest regional market share, approximately 42%, in the global advanced hepatocellular carcinoma with CPB liver cirrhosis market.
- Asia Pacific is the second largest regional market, accounting for approximately 34% of the global advanced hepatocellular carcinoma with CPB liver cirrhosis market.
- Europe is the third largest regional market, contributing approximately 30% of the global advanced hepatocellular carcinoma with CPB liver cirrhosis market.
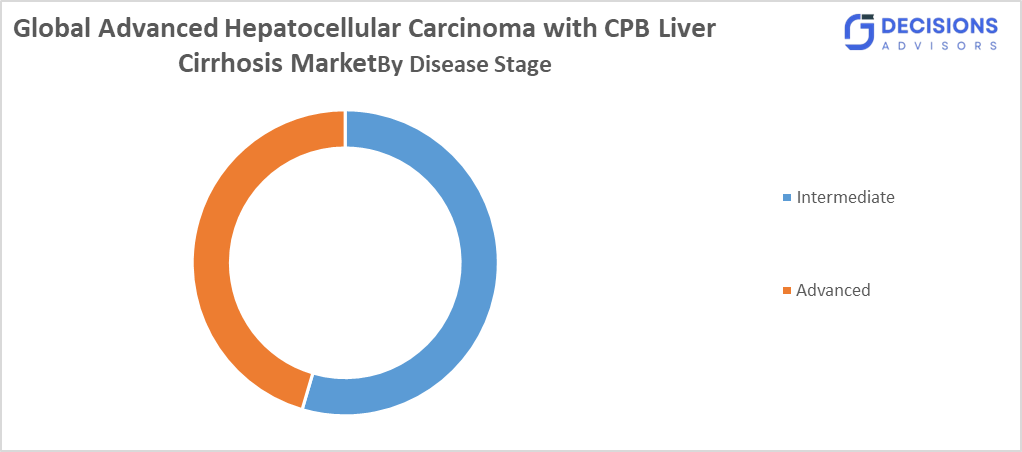
- By Disease Stage, the Intermediate segment held a dominant position with a CAGR of approximately 18% during the forecast period.
- By Treatment Type, the Systemic therapies segment held a dominant position with a CAGR of approximately 22% during the forecast period.
- By End User, the Hospitals segment held a dominant position with a CAGR of approximately 20% during the forecast period.
- The compound annual growth rate of the global advanced hepatocellular carcinoma with CPB liver cirrhosis market is 21.75%.
- The market is likely to achieve a valuation of USD 5243.7 million by 2035.
What is role of technology in grooming the market?
Technology plays a pivotal role in shaping the advanced HCC market by enhancing diagnostics, enabling precision therapies, and accelerating drug development. Advanced imaging, AI-driven liver function assessment, and liquid biopsy technologies improve early detection and accurate staging, particularly for patients with compromised liver function (CPB). Digital platforms and big data analytics streamline clinical trial design, patient recruitment, and real-world evidence collection, reducing time to market. Moreover, innovative drug delivery systems and molecular targeting technologies increase therapy efficacy and safety. Collectively, these technological advancements boost treatment adoption, optimize outcomes, and expand market potential globally.
How is Recent Developments Helping the Market?
Recent developments in the global advanced hepatocellular carcinoma with CPB liver cirrhosis market are driving growth and shaping future opportunities. The expansion of clinical trials and investigational therapies globally underscores sustained R&D activity, attracting investment and partnerships. Phase?III trials targeting Child?Pugh B patients, such as namodenoson, address unmet clinical needs, enabling broader patient access and improving outcomes. Positive early-phase study results enhance physician confidence, encouraging adoption of emerging therapies. Strategic discontinuation of less effective trials reallocates resources toward promising candidates, optimizing market potential. Collectively, these developments accelerate innovation, support commercialization, and strengthen the market across regions worldwide.
Market Drivers
The global advanced hepatocellular carcinoma with CPB liver cirrhosis market is driven by the rising prevalence of chronic liver diseases, hepatitis B and C infections, and increasing cirrhosis-related HCC cases, particularly in Asia Pacific and Africa. Expanded screening programs and greater awareness enable earlier diagnosis, facilitating timely treatment initiation. Advances in diagnostic imaging, biomarker identification, and risk stratification support personalized therapy selection. Ongoing clinical research and emerging combination regimens, including immunotherapy plus targeted therapy, are accelerating treatment adoption. Improved healthcare infrastructure, increased healthcare spending, and enhanced access to systemic and targeted therapies further fuel market growth. Patient demand for innovative and effective therapies, along with government support for oncology research and expanded reimbursement in developed countries, reinforces long-term market potential.
Restrain
Market growth is restrained by limited clinical data for Child?Pugh B patients, high costs of advanced therapies, and variable reimbursement policies. Restricted access to specialized oncology care in low- and middle-income countries, stringent regulatory requirements, and treatment safety uncertainties for CPB subgroups further hinder adoption. Diagnostic and therapy disparities, combined with limited patient awareness, continue to challenge broader market penetration.
Competitive Analysis:
The report offers the appropriate analysis of the key organizations/companies involved within the global advanced hepatocellular carcinoma with CPB liver cirrhosis market, along with a comparative evaluation primarily based on their product of offering, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborative analysis focusing on the current news and developments of the companies, which includes product development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
Top Companies in Global Advanced Hepatocellular Carcinoma with CPB Liver Cirrhosis Market
- Merck & Co., Inc.
- Bayer AG
- Roche Holding AG
- Eli Lilly and Company
- Bristol?Myers Squibb Company
- Exelixis Inc.
- AstraZeneca Plc
- Innovent Biologics Inc.
- BeiGene
- Sanofi SA
- Eisai Co., Ltd.
- Jiangsu Hengrui Medicine Co. Ltd.
Government Initiatives
|
Country |
Key Government Initiatives |
|
UK |
The UK’s NHS England has established minimum standards for hepatocellular carcinoma (HCC) surveillance, outlining quality ultrasound and monitoring pathways for high?risk patients to improve early detection and encourage regular screening and follow?up care. This supports earlier diagnosis and better clinical outcomes for liver cancer patients across the healthcare system. |
|
US |
The U.S. National Cancer Institute (NCI) supports extensive liver cancer research focused on prevention, early detection, and treatment innovations. NCI?funded programs include studies to identify high?risk cirrhotic populations, improve screening techniques, and explore biomarkers and therapeutics that may benefit advanced HCC patients. |
|
China |
China’s national liver cancer diagnosis and treatment guidelines (2024 edition) have been updated to reflect the latest evidence?based practices with the aim of standardizing HCC care and improving survival rates. These guidelines are part of a broader national health strategy aligned with Healthy China 2030 to strengthen cancer management and treatment nationwide. |
Market Segmentation
The global advanced hepatocellular carcinoma with CPB liver cirrhosis market share is classified into disease stage, treatment type, and end user
- The Intermediate segment dominated the market in 2025, and is projected to grow at a substantial CAGR of approximately 18% during the forecast period.
Based on the disease stage, the global advanced hepatocellular carcinoma with CPB liver cirrhosis market is divided into intermediate and advanced. Among these, the intermediate segment dominated the market in 2025, and is projected to grow at a substantial CAGR of approximately 18% during the forecast period. This dominance is due to earlier diagnosis in patients with compensated liver function, enabling timely treatment initiation and better management outcomes. Improved screening programs and regional healthcare awareness have further strengthened the adoption of treatments at this stage, particularly in developed countries.
- The Systemic therapies segment dominated the market in 2025, and is projected to grow at a substantial CAGR of approximately 22% during the forecast period.
Based on the treatment type, global advanced hepatocellular carcinoma with CPB liver cirrhosis market is divided into systemic therapies, targeted therapies, immunotherapies, and supportive care. Among these, the systemic therapies segment dominated the market in 2025, and is projected to grow at a substantial CAGR of approximately 22% during the forecast period. Its dominance is attributed to wide accessibility, established clinical efficacy, and cost-effectiveness for patients not eligible for surgical interventions. Increasing R&D and combination therapy trials further reinforce its strong market position globally.
- The Hospitals segment dominated the market in 2025, and is projected to grow at a substantial CAGR of approximately 20% during the forecast period.
Based on the end user, the global advanced hepatocellular carcinoma with CPB liver cirrhosis market is divided into hospitals, specialty oncology clinics, and cancer research institutes. Among these, the hospitals segment dominated the market in 2025, and is projected to grow at a substantial CAGR of approximately 20% during the forecast period. Hospitals are preferred due to advanced infrastructure, comprehensive oncology care, availability of multidisciplinary teams, and access to clinical trials. Their central role in patient management ensures high treatment adoption, especially in urban and semi-urban regions.
What is the Reason of the Region Dominance?
The dominance of certain regions in the global advanced hepatocellular carcinoma with CPB liver cirrhosis market is primarily driven by the high prevalence of liver disease, well-established healthcare infrastructure, strong clinical trial activity, and increasing access to advanced therapies. Regions such as Asia-Pacific lead the market due to the large patient populations in countries like China, Japan, and India, where chronic hepatitis B and C contribute to high HCC incidence. Expanding screening programs, rising awareness, and increased adoption of systemic and targeted therapies further support market growth. Additionally, government initiatives, investment in oncology care, and regional R&D efforts strengthen the market’s regional dominance.
Strategies to Implement for Growth of the Market in Non-Leading Regions
Growth in non-leading regions can be supported through strategic investments, expansion of healthcare infrastructure, and enhanced access to advanced therapies. Companies can increase market penetration by introducing cost-effective treatment options, patient assistance programs, and therapies adapted to local healthcare needs. Expanding hospital networks, specialty oncology clinics, and clinical trial participation can also improve therapy accessibility in emerging markets. Additionally, partnerships with local healthcare providers, government programs, and patient advocacy groups can enhance awareness and adoption. Investment in regional research, training, and telemedicine solutions may further strengthen treatment delivery and market growth.
Regional Segment Analysis of the Advanced Hepatocellular Carcinoma with CPB Liver Cirrhosis Market
- North America (U.S., Canada, Mexico)
- Europe (Germany, France, U.K., Italy, Spain, Rest of Europe)
- Asia-Pacific (China, Japan, India, Rest of APAC)
- South America (Brazil and the Rest of South America)
- The Middle East and Africa (UAE, South Africa, Rest of MEA)
Asia Pacific is anticipated to hold the largest share of global advanced hepatocellular carcinoma with CPB liver cirrhosis market over the predicted timeframe. Asia Pacific is anticipated to hold the largest share of the global advanced hepatocellular carcinoma with CPB liver cirrhosis market over the predicted timeframe. This market is accounting for approximately 34% of the total market share, the dominance of this market is driven by the high prevalence of liver disease and cirrhosis in countries such as China, Japan, and India, expanding healthcare infrastructure, rising awareness, and the growing adoption of advanced systemic therapies targeting Child?Pugh B patients.
North America is expected to grow at a rapid CAGR in the global advanced hepatocellular carcinoma with CPB liver cirrhosis market during the forecast period. North America is expected to grow at a rapid CAGR in the global advanced hepatocellular carcinoma with CPB liver cirrhosis market during the forecast period. The region is holding approximately 42% of the global market, driven by well-established healthcare systems, high adoption of systemic and targeted therapies, and extensive clinical trial activity, particularly in the United States, which contributes the majority of regional revenue.
Europe is the third largest region to grow in global advanced hepatocellular carcinoma with CPB liver cirrhosis market during the period. Europe is the third largest region to grow in the global advanced hepatocellular carcinoma with CPB liver cirrhosis market during the period. This market accounts for approximately 30% of the total global share, supported by strong cancer care networks, reimbursement policies, and the adoption of advanced systemic and combination therapies, with countries such as Germany, France, and the United Kingdom contributing significantly to overall regional demand.
Future Market Trends in Global Advanced Hepatocellular Carcinoma with CPB Liver Cirrhosis Market: -
1. Expansion of Targeted and Combination Therapies
Increasing development of targeted agents and immunotherapy combinations will continue to drive treatment personalization and efficacy, especially for patients with compromised liver function, creating new market opportunities.
2. Adoption of Precision Diagnostics
Advanced imaging, liquid biopsy, and AI-based liver function assessment will improve early detection, accurate staging, and treatment selection, supporting higher adoption rates of innovative therapies.
3. Emergence of Biomarker-Driven Therapies
Growth in therapies guided by molecular and genetic biomarkers will optimize patient outcomes, foster premium therapy adoption, and expand the market for precision medicine in advanced HCC globally.
Recent Development
- In September?2025, A comprehensive industry report showed that the global advanced liver cancer drug pipeline remains robust, with 50+ companies actively advancing clinical candidates, reflecting sustained R&D momentum that underpins future treatment innovation and market expansion in HCC therapies.
- In April?2025, A Phase?III multicenter clinical trial of namodenoson was actively enrolling patients with advanced hepatocellular carcinoma and Child?Pugh Class B7 cirrhosis to evaluate its efficacy and safety compared with placebo, indicating significant clinical focus on this underrepresented CPB subgroup.
- In October?2025, A late?stage Phase?III trial combining pembrolizumab (Keytruda) with lenvatinib plus transarterial chemoembolization in unresectable HCC was stopped after interim analysis indicated it was unlikely to meet its primary overall survival goal, illustrating the competitive and challenging nature of developing new combination regimens for advanced liver cancer.
Market Segment
This study forecasts revenue at global, regional, and country levels from 2020 to 2035. Decision Advisor has segmented the global advanced hepatocellular carcinoma with CPB liver cirrhosis market based on the below-mentioned segments:
Global Advanced Hepatocellular Carcinoma with CPB Liver Cirrhosis Market, By Disease Stage
- Intermediate
- Advanced
Global Advanced Hepatocellular Carcinoma with CPB Liver Cirrhosis Market, By Treatment Type
- Systemic Therapies
- Targeted Therapies
- Immunotherapies
- Supportive Care
Global Advanced Hepatocellular Carcinoma with CPB Liver Cirrhosis Market, By End User
- Hospitals
- Specialty Oncology Clinics
- Cancer Research Institutes
Global Advanced Hepatocellular Carcinoma with CPB Liver Cirrhosis Market, By Regional Analysis
- North America
- US
- Canada
- Mexico
- Europe
- Germany
- UK
- France
- Italy
- Spain
- Russia
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- Australia
- Rest of Asia Pacific
- South America
- Brazil
- Argentina
- Rest of South America
- Middle East & Africa
- UAE
- Saudi Arabia
- Qatar
- South Africa
- Rest of the Middle East & Africa
Frequently Asked Questions (FAQ)
Q: What specific patient population is targeted in the advanced HCC with CPB liver cirrhosis market?
A: The market focuses on hepatocellular carcinoma patients with moderate liver impairment (Child?Pugh B). These patients require specialized systemic, targeted, or combination therapies due to compromised liver function, making standard treatments less safe and less effective.
Q: How does regional healthcare infrastructure affect the market growth?
A: Strong healthcare infrastructure, including hospitals, oncology clinics, and cancer research centers, enables early diagnosis, access to advanced therapies, and clinical trial participation. This increases adoption rates, improves patient outcomes, and drives overall market growth in developed regions.
Q: Are there any emerging therapy trends in this market?
A: Yes, combination therapies, immunotherapy-based regimens, and biomarker-guided treatments are expanding. These approaches personalize therapy, enhance treatment efficacy, and provide safer options for patients with impaired liver function, particularly those with Child?Pugh B cirrhosis.
Q: How do government programs impact patient access to therapies?
A: National insurance schemes, patient assistance programs, and screening initiatives improve affordability and accessibility of advanced HCC treatments. These programs help reduce disparities, promote adoption of systemic and targeted therapies, and expand treatment
Check Licence
Choose the plan that fits you best: Single User, Multi-User, or Enterprise solutions tailored for your needs.
We Have You Covered
- 24/7 Analyst Support
- Clients Across the Globe
- Tailored Insights
- Technology Tracking
- Competitive Intelligence
- Custom Research
- Syndicated Market Studies
- Market Overview
- Market Segmentation
- Growth Drivers
- Market Opportunities
- Regulatory Insights
- Innovation & Sustainability
Report Details
| Scope | Global |
| Pages | 245 |
| Delivery | PDF & Excel via Email |
| Language | English |
| Release | Mar 2026 |
| Access | Download from this page |
