Global Allergy Diagnostics Market
Global Allergy Diagnostics Market Size, Share, and COVID-19 Impact Analysis, By Product Type (Fungal Allergy Diagnostic and Neutral Lactase Enzymes), By Allergen Type (Inhaled Allergens, Drug Allergens, Food Allergens and Others), By Test Type (In-Vitro Allergy Tests and In-Vivo Allergy Tests), and By Region (North America, Europe, Asia-Pacific, Latin America, Middle East, and Africa), Analysis and Forecast 2025-2035
Report Overview
Table of Contents
Global Allergy Diagnostics Market Size Insights Forecasts to 2035
- The Global Allergy Diagnostics Market Size Was Estimated at USD 6.63 Billion in 2024
- The Market Size is Expected to Grow at a CAGR of around 9.85 % from 2025 to 2035
- The Worldwide Allergy Diagnostics Market Size is Expected to Reach USD 18.64 Billion by 2035
- North America is expected to grow the fastest during the forecast period.
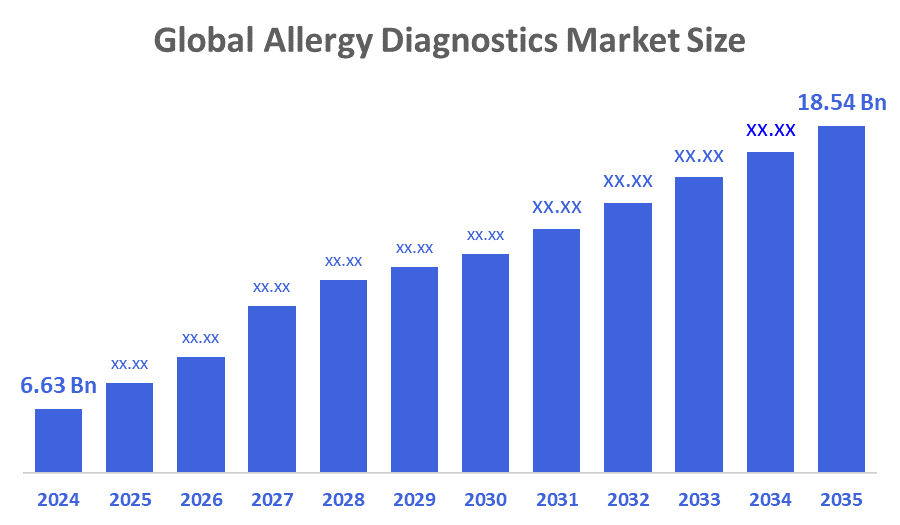
According to a research report published by Decisions Advisors and Consulting, the Global Allergy Diagnostics Market Size was worth around USD 6.63 billion in 2024 and is predicted to grow to around USD 18.64 billion by 2035 with a compound annual growth rate (CAGR) of 9.85 % from 2025 to 2035. The growing use of AI and molecular diagnostics, the development of point-of-care testing and at-home care, favourable government reimbursement policies, and the rise in tailored therapies, particularly immunotherapy and targeted allergy treatments, are the main factors driving the market's growth.
Market Overview
The allergy diagnostics market includes methods that assist in identifying particular allergens that trigger allergic reactions. These include blood and skin testing, as well as challenge tests to determine an individual's sensitivity to allergens like food, dust, pollen, and pet hair, which aid in the development of treatment strategies. Hospitals and labs devote more money to multiplex testing platforms and automated analyzers which allow the simultaneous screening of many allergens, increasing flexibility and effectiveness. Digital health platforms facilitate this technological integration by facilitating predictive analytics, improved data management, and electronic reporting. These tests allow for the assessment of drug hypersensitivity using beta-lactam-specific IgE measurement, respiratory allergy profiling using pHL p 1 and pHL p 5 for grass pollen immunotherapy selection, venom allergy confirmation using Api m 1 for bee sting reactions, and food allergy risk stratification using Ara h 2 quantification in peanut challenges. The market for allergy diagnostics is driven by the rising incidence of allergic illnesses, as medical professionals look for accurate instruments to recognise patterns of sensitisation and effectively direct immunotherapy or avoidance tactics.
Around the world, a significant percentage of individuals suffer from allergic disorders. Approximately 22% of individuals globally have an allergic condition, according to a global survey conducted in 33 nations. This means that almost 1 in 5 people have an allergy.
According to extensive epidemiological studies, at least 30–40% of people worldwide are afflicted by allergic disorders as a group, which include rhinitis, eczema, food allergies, and asthma.
More than 160 allergy and immunology medications are under development, with a record number of late-stage clinical studies in progress, according to the most recent biopharma update from investment banking firm Stifel.
In 2025, allergy biotechs' average enterprise value increased by 72% to $1.35 billion, making them the second-highest among the illness categories the company examined. This increase surpassed obesity, one of the most popular drug development fields, and came in second only to RNA treatments.
Report Coverage
This research report categorizes the allergy diagnostics market based on various segments and regions, forecasts revenue growth, and analyzes trends in each submarket. The report analyses the key growth drivers, opportunities, and challenges influencing the allergy diagnostics market. Recent market developments and competitive strategies, such as expansion, product launch, development, partnership, merger, and acquisition, have been included to draw the competitive landscape in the market. The report strategically identifies and profiles the key market players and analyzes their core competencies in each sub-segment of the allergy diagnostics market.
Driving Factors
Market growth is influenced by primary factors, which include increasing allergy rates, advanced diagnostic technologies and the expansion of healthcare systems. The diagnostic landscape transforms these factors, which enable faster and more precise detection and create new business opportunities for existing companies and startup ventures. The global rise in allergy cases, which includes respiratory and food-related allergies, stands as the most powerful force driving market expansion. Urban areas, together with changes in food consumption and higher levels of pollution exposure, serve as the main factors behind this situation. Enhanced healthcare access across both developed nations and developing nations has resulted in greater test numbers and expanded patient reach for diagnostic testing companies.
The businesses are gathering money and making acquisitions in the field in addition to clinical research. This spring, Swiss biotech Mabylon raised $34 million to expand an antibody program that targets food allergies, while Bigger companies are also acting: Sanofi paid $9.5 billion to purchase Blueprint Medicines in order to broaden its immunology portfolio to include disorders that are similar to those treated for allergies.
bioMérieux acquired a stake in Allergen Alert, a startup pioneering rapid food allergen detection. The company is developing a portable, reliable solution that identifies allergens and gluten within minutes, enhancing food safety for individuals with allergies or celiac disease and supporting professionals in delivering safer dietary options.
Restraining Factors
The financial difficulties facing healthcare providers are driving up the cost of allergy diagnostic tools. Return on investment takes longer to recover since high-end devices like chromatography equipment, hemodialyzers, and chemical analysers are quite expensive. Careful planning and resource allocation techniques are required in light of these rising costs.
Market Segmentation
The allergy diagnostics market share is classified into product type, allergen type, and test type.
- The fungal allergy diagnostic segment accounted for the largest share in 2024 and is anticipated to grow at a significant CAGR during the forecast period.
Based on the product type, the allergy diagnostics market is divided into fungal allergy diagnostics and neutral lactase enzymes. Among these, the fungal allergy diagnostic segment accounted for the largest share in 2024 and is anticipated to grow at a significant CAGR during the forecast period. This is because higher testing volumes are a result of increased exposure to mould spores in indoor environments. Children's and adults' respiratory sensitivity is increased by climate change and urban pollution, which increases the need for diagnostics. Doctors are increasingly assessing fungal allergen responses in the treatment of chronic sinusitis and asthma.
For instance, Dupixent (dupilumab) been approved in the U.S. as the first and only treatment for Allergic Fungal Rhinosinusitis (AFRS), marking a breakthrough in care for patients with this chronic and difficult-to-treat condition.
- The inhaled allergens segment accounted for the largest share in 2024 and is anticipated to grow at a significant CAGR during the forecast period.
Based on the allergen type, the allergy diagnostics market is divided into inhaled allergens, drug allergens, food allergens and others. Among these, the inhaled allergens segment accounted for the largest share in 2024 and is anticipated to grow at a significant CAGR during the forecast period. This is because dust mites, pollen, pet dander, and mould are associated with a high incidence of allergic rhinitis and asthma. For a quicker diagnosis and more focused immunotherapy prescriptions, medical professionals give priority to inhaled allergen testing.
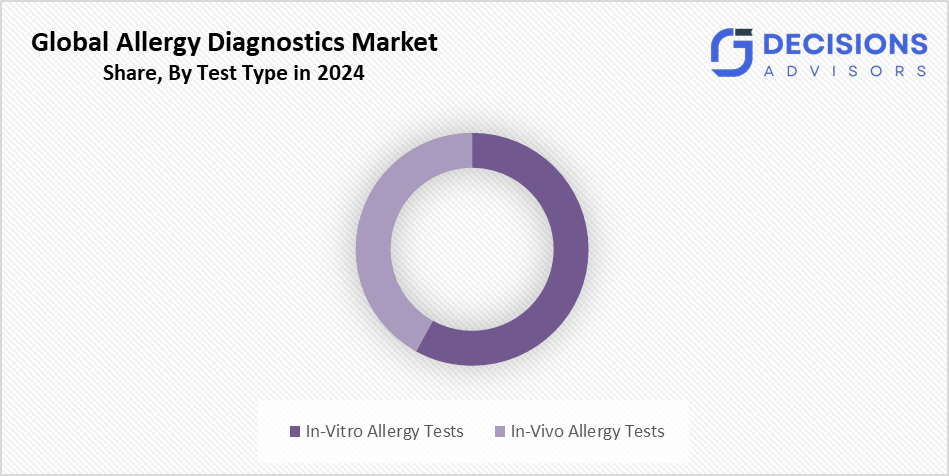
- The in-vitro allergy tests segment accounted for the highest market revenue in 2024 and is anticipated to grow at a significant CAGR during the forecast period.
Based on the test type, the allergy diagnostics market is divided into in-vitro allergy tests and in-vivo allergy tests. Among these, the in-vitro allergy tests segment accounted for the highest market revenue in 2024 and is anticipated to grow at a significant CAGR during the forecast period. This is because they provide quantitative results that help long-term illness surveillance and safer testing conditions for individuals at risk of severe reactions. Automated immunoassay analysers and other technological advancements improve sensitivity and shorten turnaround times.
For instance, the FDA 510(k) Clearance Tracker for In Vitro Diagnostic (IVD) Tests and Instruments is essentially a monitoring tool that records which diagnostic devices and assays have received clearance under the FDA’s 510(k) pathway.
Regional Segment Analysis of the Allergy Diagnostics Market
- North America (U.S., Canada, Mexico)
- Europe (Germany, France, U.K., Italy, Spain, Rest of Europe)
- Asia-Pacific (China, Japan, India, Rest of APAC)
- South America (Brazil and the Rest of South America)
- The Middle East and Africa (UAE, South Africa, Rest of MEA)
Asia Pacific is anticipated to hold the largest share of the allergy diagnostics market over the predicted timeframe.
Asia Pacific is anticipated to hold the largest share of the allergy diagnostics market over the predicted timeframe. The regional market is propelled by elements including growing infrastructure and healthcare reforms. Moreover, local key firm entries, rising allergy rates, and population growth all contribute to market expansion. Furthermore, China, India, and Japan are important markets in this area that are expected to increase regional market share. Strategic activities are being implemented by major players in these nations to increase their market position.
Toray (Japan) introduced a biological crystal chip for advanced multi-allergen testing. It can measure multiple allergen-specific IgG antibodies from just 20 µL of blood, using 3D gene microarray technology for high sensitivity and precision, while a low-contamination polymer prevents interference.
North America is expected to grow at a rapid CAGR in the allergy diagnostics market during the forecast period. Asthma and Allergy Foundation of America data indicates that allergies are the sixth most prevalent chronic illness in the United States, affecting around 50 million people each year. The need for quick and accurate allergy diagnostics is expanding as drug-related deaths increase. The high incidence of allergic rhinitis and the prevalence of skin allergies, especially eczema and skin inflammation, among Black Americans will drive the growth of the allergy diagnostics market in the United States.
DBV Technologies collected almost $300 million to support its efforts to market its peanut allergy patch in the United States
Competitive Analysis:
The report offers the appropriate analysis of the key organizations/companies involved within the allergy diagnostics market, along with a comparative evaluation primarily based on their product offering, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborative analysis focusing on the current news and developments of the companies, which includes product development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
List of Key Companies
- Agnitiost
- Biomérieux
- Danaher Corporation (Beckman Coulter)
- Eurofins Scientific
- Euroimmun
- Excelsior bio-system
- Hollisterstier allergy
- Hycor Biomedical
- Lincoln diagnostics
- Med Phar Enterprise
- Neogen Corporation
- Omega diagnostics
- Rega biotechnology
- Sgs Taiwan
- Others
Key Target Audience
- Market Players
- Investors
- End-users
- Government Authorities
- Consulting and Research Firm
- Venture capitalists
- Value-Added Resellers (VARs)
Recent Development
- In July 2025, InBio and Beckman Coulter Life Sciences announced a strategic partnership to advance food allergy research. The collaboration aims to combine expertise in allergen detection and diagnostic innovation, driving new insights and solutions to improve patient safety and outcomes in the growing field of food allergy management.
- In April 2025, Beckman Coulter Life Sciences introduced a next-generation allergy test designed to improve diagnostic accuracy and reduce patient risk. The innovative solution aims to streamline allergy detection, offering clinicians enhanced tools to support safer, faster, and more reliable patient care.
- In September 2024, Mitsubishi partnered with Alerje to transform allergy care, combining innovative technology with patient-centred solutions. The collaboration aims to enhance allergy management through connected devices and digital health tools, offering improved monitoring, treatment adherence, and quality of life for patients living with allergic conditions.
- In July 2024, AliveDx received the IVDR CE Mark for its groundbreaking microarray immunoassay in allergy diagnostics. This approval marks a significant advancement in precision testing, enabling more accurate detection and improved patient outcomes across Europe.
Market Segment
This study forecasts revenue at global, regional, and country levels from 2020 to 2035. Decisions Advisors has segmented the allergy diagnostics market based on the below-mentioned segments:
Global Allergy Diagnostics Market, By Product Type
- Fungal Allergy Diagnostic
- Neutral Lactase Enzymes
Global Allergy Diagnostics Market, By Allergen Type
- Inhaled Allergens
- Drug Allergens
- Food Allergens and Others
Global Allergy Diagnostics Market, By Test Type
- In-Vitro Allergy Tests
- In-Vivo Allergy Tests
Global Allergy Diagnostics Market, By Regional Analysis
- North America
- US
- Canada
- Mexico
- Europe
- Germany
- UK
- France
- Italy
- Spain
- Russia
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- Australia
- Rest of Asia Pacific
- South America
- Brazil
- Argentina
- Rest of South America
- Middle East & Africa
- UAE
- Saudi Arabia
- Qatar
- South Africa
- Rest of the Middle East & Africa
Frequently Asked Questions (FAQ)
- How is molecular allergy diagnostics transforming traditional testing methods?
Molecular allergy diagnostics enables the identification of specific allergenic protein components rather than whole allergen extracts. This improves diagnostic precision, supports personalised immunotherapy decisions, and reduces cross-reactivity misinterpretation, making treatment planning more effective.
- What role does artificial intelligence (AI) play in allergy diagnostics?
AI supports allergy diagnostics through predictive analytics, automated result interpretation, digital reporting systems, and improved laboratory workflow efficiency. AI-driven algorithms can identify sensitisation patterns and assist clinicians in selecting targeted treatment strategies.
- How is home-based or point-of-care allergy testing influencing market growth?
The emergence of rapid test kits and portable diagnostic platforms allows allergy testing outside traditional hospital settings. This improves accessibility, reduces diagnostic delays, and supports early detection, particularly in remote or underserved regions.
- Which patient demographics are contributing most significantly to the demand for allergy diagnostics?
Pediatric and geriatric populations are key contributors. Rising childhood food allergies and increasing respiratory allergies among the ageing population are driving higher testing volumes globally.
- What regulatory factors impact the commercialisation of allergy diagnostic products?
Allergy diagnostic devices must comply with regulatory frameworks such as FDA 510(k) clearance in the U.S. and IVDR certification in Europe. Strict validation requirements, clinical performance data, and post-market surveillance obligations influence product launch timelines and costs.
- How do multiplex testing platforms benefit clinical laboratories?
Multiplex platforms allow simultaneous detection of multiple allergens from a single patient sample. This reduces testing time, enhances laboratory efficiency, lowers operational costs per test, and improves patient convenience.
- What are the emerging investment opportunities in the allergy diagnostics market?
Investment opportunities are growing in digital allergy management platforms, biomarker-based diagnostics, wearable allergy monitoring devices, and AI-integrated laboratory systems. Startups focusing on rapid allergen detection and personalised immunotherapy support tools are attracting venture capital interest.
Check Licence
Choose the plan that fits you best: Single User, Multi-User, or Enterprise solutions tailored for your needs.
We Have You Covered
- 24/7 Analyst Support
- Clients Across the Globe
- Tailored Insights
- Technology Tracking
- Competitive Intelligence
- Custom Research
- Syndicated Market Studies
- Market Overview
- Market Segmentation
- Growth Drivers
- Market Opportunities
- Regulatory Insights
- Innovation & Sustainability
Report Details
| Scope | Global |
| Pages | 240 |
| Delivery | PDF & Excel via Email |
| Language | English |
| Release | Mar 2026 |
| Access | Download from this page |
