Global Anti Cathepsin B Market
Global Anti Cathepsin B Market Size, Share, By Product (Primary Antibodies, Proteins & Peptides, Lysates), By Application (Cancer, Traumatic Brain Injury, Ebola Infection, Fertility Treatment), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), and By Region (North America, Europe, Asia-Pacific, Latin America, Middle East & Africa), Analysis and Forecast 2026-2035
Report Overview
Table of Contents
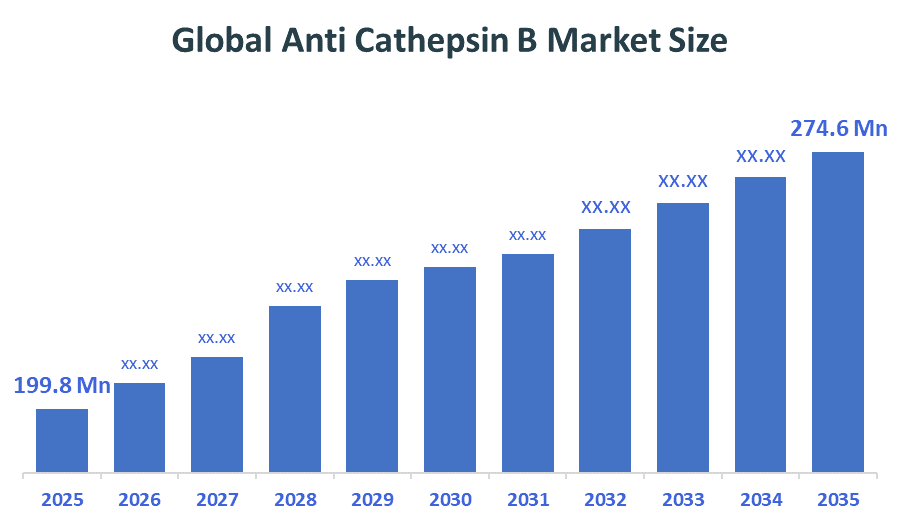
The Global Anti Cathepsin B Market Size is forecast to grow from USD 199.8Million in 2025 to approximately USD 274.6 Million by 2035. According to Decision Advisors, a detailed research report on the anti cathepsin b market indicates that the increasing use of cathepsin B–targeted antibodies in oncology research trend dominates the market, accounting for approximately 46.5-47.5% of the total share globally. Thermo Fisher Scientific leads the market with an estimated 12-18% global market share. The company reported a 2025 revenue of approximately USD 43-45 Billion, making it one of the most influential forces shaping industry trends and overall market growth.
Market Snapshot
- Global Anti Cathepsin B Market Size (2025): USD 199.8Million
- Projected Global Anti Cathepsin B Market Size (2035): USD 274.6Million
- Global Anti Cathepsin B Market Compound Annual Growth Rate (CAGR): 3.23%
- Largest Regional Market: North America
- Fastest Growing Region: Asia-Pacific
- Market 3rd Largest Region: Europe
- Base Year: 2025
- Historical Period: 2021–2024
- Forecast Period: 2026–2035
Market Overview/ Introduction
The Global Anti-Cathepsin B Market refers to a sector that develops, produces, and sells products that include inhibitors and antibodies, and diagnostic tools. These products specifically target Cathepsin B, which represents a lysosomal cysteine protease that scientists associate with cancer development, neurodegenerative disorders, and inflammatory diseases. The Anti-Cathepsin B market includes all therapeutic products and research materials that serve to block or control this enzyme for enhanced clinical diagnosis and treatment options. The market will expand through new uses that scientists will establish for precision oncology and neurodegenerative disorder treatment and biomarker-based diagnostic systems. The market expands because more people develop chronic illnesses and more people want targeted treatments, biotechnology progresses, and research and development investments increase. The development of drug delivery systems, personalized medicine, and AI-based drug discovery solutions drives the worldwide growth and market entry of Anti-Cathepsin B products.
- The National Cancer Institute continued funding multiple schemes, such as Innovative Research in Cancer Nanotechnology (IRCN) and Toward Translation of Nanotechnology Cancer Interventions (TTNCI), to support the development of advanced nanoparticle-based cancer therapies and targeted inhibitors. These programs promote translational research for improving tumor selectivity and therapeutic efficacy.
- The National Institutes of Health Common Fund supports large-scale nanomedicine research programs aimed at developing molecular-level therapies for diseases, including cancer. The initiative funds interdisciplinary research to design nanoscale drug delivery systems, which are critical for next-generation inhibitors such as Cathepsin B-targeted nanoparticles.
Notable Insights: -
- North America is anticipated to hold the largest share of approximately 45% in the anti cathepsin b market over the forecast period.
- Asia Pacific is expected to grow at a rapid CAGR of approximately 11% in the anti cathepsin b market during the forecast period.
- The primary antibodies segment dominated the market in 2025, approximately 50 %, and is projected to grow at a substantial CAGR during the forecast period.
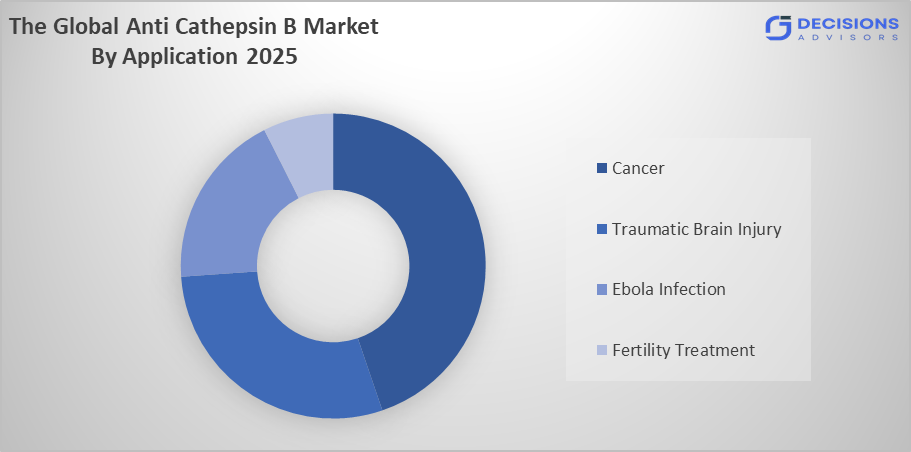
- The cancer segment dominated the market in 2025, approximately 60%, and is projected to grow at a substantial CAGR during the forecast period.
- The hospital pharmacies segment dominated the market in 2025, approximately 45%, and is projected to grow at a substantial CAGR during the forecast period.
- The compound annual growth rate of the Global Anti Cathepsin B Market is 3.23%.
- The market is likely to achieve a valuation of USD 274.6 Million by 2035.
What is role of technology in grooming the market?
Technology plays a crucial role in grooming the Global Anti-Cathepsin B Market by enhancing drug discovery, diagnostic accuracy, and therapeutic development. Advanced tools such as high-throughput screening and computational drug design enable faster identification of potent Cathepsin B inhibitors. Artificial intelligence and machine learning are increasingly used to analyze biological datasets, predict enzyme behavior, and optimize drug candidates, reducing development time and cost. Additionally, CRISPR and gene-editing technologies support deeper understanding of Cathepsin B’s role in disease pathways. Innovations in molecular diagnostics and biomarker detection improve early disease identification and patient stratification. Nanotechnology-based drug delivery systems further enhance targeted therapy efficiency and reduce side effects. Overall, technological advancements are accelerating research, improving clinical outcomes, and expanding commercial opportunities in the Anti-Cathepsin B market globally.
Market Drivers
The Global Anti-Cathepsin B Market is expanding because many patients suffer from cancer, neurodegenerative diseases, and inflammatory diseases, and Cathepsin B is a big part of how these diseases get worse. The demand for targeted therapies together with precision medicine drives the development of selective inhibitors and diagnostic instruments. More funding in research and development through biotechnology and pharmaceutical industries creates a bigger pipeline of upcoming drug products. Better biomarker discovery and molecular diagnostics enable earlier disease detection, which results in improved treatment outcomes. Also, more uses for drug delivery systems, especially enzyme-activated therapies, and more partnerships between research institutes and businesses are helping the market grow even more around the world.
Restrain
The global anti cathepsin b market faces restraints due the high cost of development and production of specialized inhibitors, antibodies, and assay kits, which limits accessibility for smaller research institutions. Additionally, stringent regulatory requirements, limited clinical validation, and variability in enzyme behaviour across diseases further challenge widespread adoption and commercialization.
Competitive Analysis:
The report offers the appropriate analysis of the key organizations/companies involved within the global Anti cathepsin B market, along with a comparative evaluation primarily based on their product offerings, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborative analysis focusing on the current news and developments of the companies, which includes product development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
Top Companies in Global Anti Cathepsin B Market
- Merck KGaA
- Bio-Techne
- BioVision Inc.
- Santa Cruz Biotechnology, Inc.
- MedChemExpress
- BioCat GmbH
- ApexBio Technology
- Cayman Chemical
- Selleck Chemicals
- BOC Sciences
- AG Scientific
- Abcam Plc
Government Initiatives
|
Country |
Key Government Initiatives |
|
USA |
The FDA and NIH award grants through the Rare Neurodegenerative Disease Grants Program to identify molecular targets like Cathepsin B for ALS and Alzheimer's. The agency also promotes drug repurposing to fast-track existing molecules as potential protease inhibitors. |
|
India |
The government proposed Biopharma SHAKTI (2026-27) with an outlay of ?10,000 crores to boost biologics production. Additionally, the CDSCO recently exempted orphan drugs from mandatory port testing to accelerate patient access to rare disease therapies. |
|
China |
The NMPA has optimized the review process, allowing eligible innovative drugs to be approved for clinical trials within 30 working days. Under the Healthy China 2030 plan, the government uses new Commercial Health Insurance Innovative Drug Lists to cover high-value treatments for Alzheimer’s and rare diseases. |
Study on the Supply, Demand, Distribution, and Market Environment of the Anti Cathepsin B Market
The Global Anti-Cathepsin B Market demonstrates a balanced interplay between supply, demand, distribution, and market environment, supported by growing research and therapeutic applications. Demand is primarily driven by pharmaceutical and biotechnology companies focusing on oncology and neurodegenerative diseases, while academic institutes contribute to steady research-based consumption. Supply depends on advanced biomanufacturing processes involving critical raw materials such as recombinant proteins, cell culture media, enzymes, and high-purity reagents, which require stringent quality control and specialized sourcing. Distribution networks are increasingly shifting toward direct sales, online platforms, and global distributors to ensure timely delivery and product availability. The market environment is influenced by regulatory standards, technological advancements, and funding for life sciences research. Additionally, strategic partnerships and outsourcing of production are helping companies maintain supply chain efficiency and meet rising global demand.
Price Analysis and Consumer Behaviour Analysis
Price analysis in the Global Anti-Cathepsin B Market indicates a premium pricing structure for high-quality recombinant proteins, monoclonal antibodies, and validated assay kits, driven by stringent quality requirements and complex production processes. Pharmaceutical companies often pay 2–3 times higher prices for reliable, reproducible reagents, while academic institutions prefer cost-effective alternatives due to budget constraints. However, increasing competition and e-commerce-based procurement are creating pricing pressures, especially from low-cost suppliers in Asia-Pacific. From a consumer behavior perspective, buyers prioritize product specificity, reproducibility, and validated data over price, particularly in regulated research environments. Pharmaceutical and biotech firms dominate demand, focusing on premium, high-performance products, whereas academic and smaller labs show price sensitivity. Additionally, purchasing decisions are increasingly influenced by supplier reputation, technical support, and digital accessibility, reflecting a shift toward value-based and convenience-driven procurement strategies.
Market Segmentation
The Anti Cathepsin B Market share is classified into product, application, and distribution channel.
- The primary antibodies segment dominated the market in 2025, approximately 50%, and is projected to grow at a substantial CAGR during the forecast period.
Based on the product, the anti cathepsin b market is divided into primary antibodies, proteins & peptides, and lysates. Among these, the primary antibodies segment dominated the market in 2025, approximately 50%, and is projected to grow at a substantial CAGR during the forecast period. The growth of the primary antibodies segment is growing due to their extensive use in research, diagnostics, and protein detection assays. Their high specificity and reliability in identifying Cathepsin B expression make them essential in cancer and neurodegenerative studies. Growing demand for reproducible laboratory results and expanding biomedical research activities further strengthen their leading market position globally.
- The cancer segment dominated the market in 2025, approximately 60%, and is projected to grow at a substantial CAGR during the forecast period.
Based on the application, the anti cathepsin b market is divided into cancer, traumatic brain injury, ebola infection, and fertility treatment. Among these, the cancer segment dominated the market in 2025, approximately 60%, and is projected to grow at a substantial CAGR during the forecast period. The cancer segment dominated due to the strong involvement of Cathepsin B in tumor invasion, metastasis, and progression. Increasing global cancer prevalence and rising adoption of targeted therapies significantly boost demand for Anti-Cathepsin B solutions. Continuous oncology research and biomarker development further reinforce this segment’s dominance across pharmaceutical and clinical research sectors.
- The hospital pharmacies drugs segment dominated the market in 2025, approximately 45%, and is projected to grow at a substantial CAGR during the forecast period.
Based on the distribution channel, the anti cathepsin b market is divided into hospital pharmacies, retail pharmacies, and online pharmacies. Among these, the hospital pharmacies segment dominated the market in 2025, approximately 45%, and is projected to grow at a substantial CAGR during the forecast period. The growth of the hospital pharmacies segment is due to high usage of diagnostic kits, antibodies, and research reagents in clinical and hospital-based laboratories. Strong procurement systems, direct supplier contracts, and reliable access to advanced therapeutic tools support this dominance. Growing hospital-based research and diagnostic activities further enhance demand through this distribution channel globally.
Strategies to Implement for Growth of the Market in Non-Leading Regions
To drive growth of the Global Anti-Cathepsin B Market in non-leading regions, companies should focus on expanding research collaborations with local academic institutions and healthcare organizations to strengthen clinical development and awareness. Enhancing access through cost-effective production and tiered pricing strategies can improve adoption in price-sensitive markets. Investing in regional clinical trials helps address population-specific disease profiles and regulatory requirements, accelerating approvals. Building distribution networks and partnerships with local pharmaceutical firms can improve market penetration. Additionally, increasing awareness through physician education and training programs will support the adoption of Anti-Cathepsin B-based diagnostics and therapies. Leveraging digital health technologies and telemedicine can further extend reach in underserved areas. Overall, a combination of localized strategies, affordability, and strong partnerships will be key to unlocking growth potential in emerging and underpenetrated markets.
Regional Segment Analysis of the Anti Cathepsin B Market
- North America (U.S., Canada, Mexico)
- Europe (Germany, France, U.K., Italy, Spain, Rest of Europe)
- Asia-Pacific (China, Japan, India, Rest of APAC)
- South America (Brazil and the Rest of South America)
- The Middle East and Africa (UAE, South Africa, Rest of MEA)
North America is anticipated to hold the largest share of approximately 45% the anti cathepsin b market over the forecast period.
North America is anticipated to hold the largest share of approximately 45% the anti cathepsin b market over the forecast period. The dominance of this region is primarily driven by advanced healthcare infrastructure, high diagnosis rates, strong reimbursement policies, and early adoption of complement inhibitors. Presence of leading pharmaceutical companies and robust clinical trial activity further supports regional dominance.
Asia Pacific is expected to grow at a rapid CAGR of approximately 11% in the anti cathepsin b market during the forecast period.
Asia Pacific is expected to grow at a rapid CAGR of approximately 11% in the anti cathepsin b market during the forecast period. This growth is driven by strong biotechnology infrastructure, high R&D investment, and advanced healthcare systems. The presence of major pharmaceutical companies and leading research institutions accelerates innovation in targeted therapies and diagnostics. High prevalence of cancer and neurodegenerative diseases further boosts demand, supported by well-established regulatory frameworks and early adoption of advanced technologies.
Europe is the 3rd largest region to grow in the anti cathepsin b market during the period.
Europe is the third largest region in the Global Paroxysmal Nocturnal Hemoglobinuria (PNH) Therapeutics Market, driven by expanding biotechnology research, increasing healthcare expenditure, and rising awareness of precision medicine. Growing cancer burden and improving diagnostic infrastructure in countries like China and India are fueling demand. Government support for life sciences, cost-effective clinical trials, and increasing presence of global pharma companies are further accelerating market expansion in this region.
Future Market Trends in Global Anti Cathepsin B Market: -
1. Growth in Targeted Cancer Therapies
The increasing focus on precision medicine is driving the adoption of Cathepsin B–responsive drug delivery systems in oncology. These therapies enable site-specific drug release within tumor microenvironments, improving treatment efficacy while minimizing systemic toxicity. Rising cancer prevalence and ongoing research into protease-activated therapies are expected to accelerate demand for Anti-Cathepsin B inhibitors in targeted cancer treatment applications globally.
2. Expansion in Neurodegenerative Disease Applications
A Growing evidence linking Cathepsin B activity to neurodegenerative disorders such as Alzheimer’s and Parkinson’s disease is expanding research interest. Pharmaceutical companies are investing in inhibitors as potential therapeutic and diagnostic tools. With the global burden of neurological diseases rising and limited effective treatments available, Anti-Cathepsin B solutions are expected to gain traction in neurology-focused drug development pipelines.
3. Advancements in Biomarkers and Diagnostic Technologies
Continuous improvements in molecular diagnostics and biomarker discovery are enhancing early disease detection capabilities. Cathepsin B is increasingly being explored as a reliable biomarker for various chronic and inflammatory conditions. Integration of advanced technologies such as AI-based screening and high-throughput assays is improving accuracy and efficiency, thereby boosting the adoption of Anti-Cathepsin B products in diagnostic and research settings.
Recent Development
- In February 2026, Researchers developed next-generation cathepsin B-activatable nanoparticle inhibitors (CatB-NPs) designed to enhance tumor selectivity and overcome systemic toxicity limitations. The study demonstrated that these nanoparticles selectively activated within tumor tissues, improving antitumor efficacy while minimizing off-target effects, addressing key challenges of conventional inhibitors.
- In December 2025, a study on self-assembling, cathepsin B-cleavable prodrug nanoparticles highlighted their potential as advanced anticancer inhibitors, showing improved pharmacokinetics, targeted delivery, and reduced toxicity compared to traditional therapies.
- In August 2024, advanced proteomics and oncology studies emphasized Cathepsin B as a critical biomarker in tumor progression, accelerating the development of high-affinity antibodies and targeted therapeutic strategies for precision cancer treatment.
- In April 2023, Researchers developed a novel self-assembling peptide-based nano-inhibitor (RR–BA conjugate) targeting Cathepsin B, demonstrating significant anticancer efficacy and improved selectivity in tumor models, highlighting its potential as a next-generation therapeutic approach in oncology.
How is Recent Developments Helping the Market?
Recent developments are significantly accelerating the growth of the Global Anti-Cathepsin B Market by advancing both research capabilities and therapeutic applications. Innovations in drug discovery, particularly the development of highly selective Cathepsin B inhibitors, are improving treatment outcomes in oncology, neurodegenerative disorders, and inflammatory diseases. Increased investment by biotechnology and pharmaceutical companies is supporting clinical trials and expanding the pipeline of targeted therapies. Additionally, progress in biomarker identification and molecular diagnostics is enabling earlier disease detection and personalized treatment strategies. Collaborations between research institutions and industry players are also enhancing technology transfer and speeding up commercialization. Furthermore, the integration of advanced technologies such as AI-driven drug design and high-throughput screening is optimizing compound identification. Together, these advancements are strengthening market potential by increasing efficacy, reducing side effects, and broadening the scope of Anti-Cathepsin B applications.
Market Segment
This study forecasts revenue at global, regional, and country levels from 2020 to 2035. Decision Advisors has segmented the Anti cathepsin b market based on the below-mentioned segments:
Global Anti Cathepsin B Market, By Product
- Primary Antibodies
- Proteins & Peptides
- Lysates
Global Anti Cathepsin B Market, By Application
- Cancer
- Traumatic Brain Injury
- Ebola Infection
- Fertility Treatment
Global Anti Cathepsin B Market, By Distribution Channel
- Hospital Pharmacies
- Retail Pharmacies
- Online Pharmacies
Global Anti Cathepsin B Market, By Regional Analysis
- North America
- US
- Canada
- Mexico
- Europe
- Germany
- UK
- France
- Italy
- Spain
- Russia
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- Australia
- Rest of Asia Pacific
- South America
- Brazil
- Argentina
- Rest of South America
- Middle East & Africa
- UAE
- Saudi Arabia
- Qatar
- South Africa
- Rest of the Middle East & Africa
Frequently Asked Questions (FAQ)
Q What are the emerging technological innovations shaping the Anti-Cathepsin B market?
A. The market is being shaped by AI-driven drug discovery, CRISPR-based gene editing, and advanced proteomics platforms that help identify Cathepsin B activity more precisely. These innovations are accelerating inhibitor design, improving target validation, and reducing R&D timelines. Additionally, nanocarrier-based delivery systems are enhancing drug stability and targeted release, making therapies more effective and safer for oncology and neurological applications.
Q. How does the regulatory environment influence market development?
A. Strict regulatory frameworks ensure the safety and efficacy of Cathepsin B inhibitors and diagnostic tools, but they also increase development timelines and costs. Agencies such as the FDA and EMA require extensive clinical validation, which can slow commercialization. However, harmonization of global guidelines and fast-track approvals for oncology drugs are gradually improving market entry opportunities for innovative therapies.
Q. What role do academic and research institutes play in this market?
A. Academic and research institutes play a crucial role by conducting foundational studies on Cathepsin B’s biological functions and disease associations. They contribute to early-stage discovery, biomarker identification, and preclinical testing. Their collaborations with pharmaceutical companies help translate lab findings into clinical applications, making them key drivers of innovation and pipeline development in the market.
Q. What are the key challenges in supply chain management for this market?
A. The supply chain faces challenges such as dependence on high-purity biological raw materials, temperature-sensitive storage requirements, and complex manufacturing processes for antibodies and enzymes. Disruptions in global logistics and limited availability of specialized reagents can delay production. Additionally, ensuring batch-to-batch consistency and maintaining quality standards across global distribution networks remains a significant operational challenge.
Check Licence
Choose the plan that fits you best: Single User, Multi-User, or Enterprise solutions tailored for your needs.
We Have You Covered
- 24/7 Analyst Support
- Clients Across the Globe
- Tailored Insights
- Technology Tracking
- Competitive Intelligence
- Custom Research
- Syndicated Market Studies
- Market Overview
- Market Segmentation
- Growth Drivers
- Market Opportunities
- Regulatory Insights
- Innovation & Sustainability
Report Details
| Scope | Global |
| Pages | 240 |
| Delivery | PDF & Excel via Email |
| Language | English |
| Release | Apr 2026 |
| Access | Download from this page |
