Global Asthma Spacers Market
Global Asthma Spacers Market Size, Share, and COVID-19 Impact Analysis, By Spacer Design (Valved Holding Chamber, Non-Valved Holding Chamber, Pocket-Sized Spacers, Reusable Spacers, Disposable Spacers), By Material Type (Plastic, Metal, Silicone, and Glass), By Distribution Channel (Hospital pharmacies, Retail pharmacies, and Online pharmacies), and By Region (North America, Europe, Asia-Pacific, Latin America, Middle East, and Africa), Analysis and Forecast 2025-2035
Report Overview
Table of Contents
Global Asthma Spacers Market Size Insights Forecasts to 2035
- The Global Asthma Spacers Market Size Was Estimated at USD 2.21 Billion in 2024
- The Market Size is Expected to Grow at a CAGR of around 6.39 % from 2025 to 2035
- The Worldwide Asthma Spacers Market Size is Expected to Reach USD 4.37 Billion by 2035
- Europe is expected to grow the fastest during the forecast period.
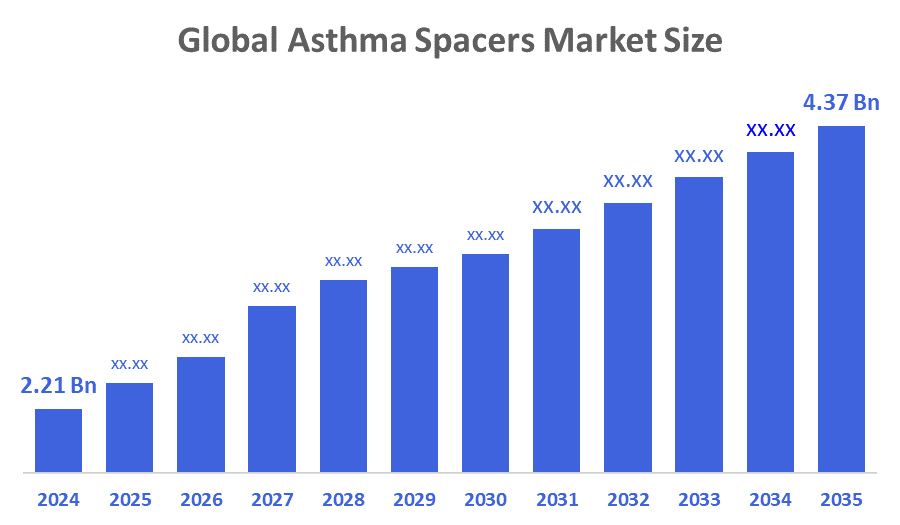
According to a research report published by Decisions Advisors and Consulting, The Global Asthma Spacers Market Size Was Worth Around USD 2.21 Billion In 2024 And Is Predicted To Grow To Around USD 4.37 Billion By 2035 With A Compound Annual Growth Rate (CAGR) Of 6.39 % From 2025 To 2035. The market's expansion is mostly driven by the increasing use of inhalation therapy devices and advancements in technology in respiratory care, which have led to more product innovation and digitisation in both clinical and at-home asthma management. The market for asthma spacers has grown significantly in recent years, thanks to the participation of top businesses and creative new advancements.
Market Overview
Inhalation chambers, sometimes referred to as asthma spacers, are crucial accessories for metered-dose inhalers (MDIs) that maximise drug delivery straight to the lungs. medical devices that work in tandem with metered-dose inhalers (MDIs) to enhance the way that drugs are inhaled into the lungs of people who have asthma or other respiratory disorders. They are extensively used in clinical and homecare settings because they can improve drug efficacy, decrease inhalation mistakes, and increase patient compliance, especially for older and pediatric patients. They have become essential accessories for managing asthma. The condition shows different rates of occurrence because it exists in different areas, with developed nations and urban regions reporting higher rates. The causes of asthma develop through various pathways that research shows depend on both genetic traits and environmental factors, which include air pollution, together with allergen exposure. The rising asthma rates make control efforts plus burden reduction work essential for the international public health system. Inhalers function as standard asthma treatment devices, but asthma spacers have become essential tools that enhance their treatment capabilities. The spacers enable better medication delivery to the lungs, which results in better control of asthma symptoms. The global market for asthma spacers creates new growth opportunities because market conditions change, and leading companies develop new products.
With an expected 480 million cases in 2020 and 592 million by 2050, the prevalence of COPD is 10.3%. COPD is expected to become more common as the population ages and smoking rates rise in low-income economies. With 262 million cases worldwide, asthma is a common chronic illness that ranks 34th in terms of the world's primary causes of disease burden and lived with disability.
Report Coverage
This research report categorizes the asthma spacers market based on various segments and regions, forecasts revenue growth, and analyzes trends in each submarket. The report analyses the key growth drivers, opportunities, and challenges influencing the asthma spacers market. Recent market developments and competitive strategies, such as expansion, product launch, development, partnership, merger, and acquisition, have been included to draw the competitive landscape in the market. The report strategically identifies and profiles the key market players and analyzes their core competencies in each sub-segment of the asthma spacers market.
Driving Factors
The combination of growing public knowledge about respiratory health and medical technology improvements drives this field forward. Healthcare professionals support spacer usage because they believe it helps patients experience better health results. The worldwide rise of asthma cases drives market growth because more patients need dependable ways to control their condition. The Asthma Spacer Market will gain advantage from product design and material innovations that will make devices easier to use and improve patient compliance. Manufacturers are developing spacers that provide effective performance while enabling users across different age ranges and asthma severity levels to use them. The market future will be affected by digital health technology implementation because patients want systems that help them monitor their health and manage their conditions.
NICE indeed published draft guidance (January 2026) recommending eight digital platforms to support people with asthma in managing their condition. These platforms are being made available in the NHS while further evidence is collected over the next three years.
Restraining Factors
Despite demonstrated clinical efficacy in enhancing drug deposition and asthma management outcomes, widespread adoption is hindered by a high dependence on inhaler-only therapy, low patient awareness of spacer benefits, and cost sensitivity in emerging markets.
Market Segmentation
The asthma spacers market share is classified into spacer design, material type, and distribution channel.
- The valved holding chamber segment accounted for the highest share in 2024 and is anticipated to grow at a significant CAGR during the forecast period.
Based on the spacer design, the asthma spacers market is differentiated into valved holding chambers, non-valved holding chambers, pocket-sized spacers, reusable spacers, and disposable spacers. Among these, the valved holding chamber segment accounted for the highest share in 2024 and is anticipated to grow at a significant CAGR during the forecast period. Their easy-to-use design that improves the effectiveness of medication distribution. They are the preferred tool for many patients and healthcare professionals since they reduce medicine waste and enhance breathing techniques.
- The plastic segment accounted for the largest market share in 2024 and is anticipated to grow at a remarkable CAGR during the forecast period.
Based on the material type, the asthma spacers market is divided into plastic, metal, silicone, and glass. Among these, the plastic segment accounted for the largest market share in 2024 and is anticipated to grow at a remarkable CAGR during the forecast period. This is because of their ability to manage patients on a massive scale and their access to state-of-the-art medical technology. They offer asthmatics a safe setting in which they can get prompt assistance and instruction on how to utilise spacers.
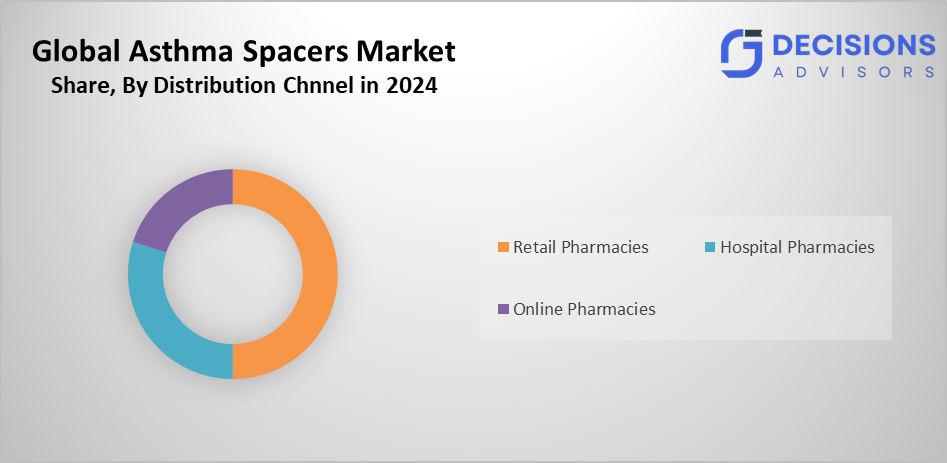
- The retail pharmacies segment accounted for the highest market share in 2024 and is anticipated to grow at a substantial CAGR over the forecast period.
Based on the distribution channel, the asthma spacers market is segmented into hospital pharmacies, retail pharmacies, and online pharmacies. Among these, the retail pharmacies segment accounted for the highest market share in 2024 and is anticipated to grow at a substantial CAGR over the forecast period. This is because of their wide network and the individualised service they provide, allowing clients to easily obtain asthma spacers and expert guidance. These pharmacies increase customer loyalty by offering a variety of connected health items and are frequently placed strategically.
Regional Segment Analysis of the Asthma Spacers Market
- North America (U.S., Canada, Mexico)
- Europe (Germany, France, U.K., Italy, Spain, Rest of Europe)
- Asia-Pacific (China, Japan, India, Rest of APAC)
- South America (Brazil and the Rest of South America)
- The Middle East and Africa (UAE, South Africa, Rest of MEA
Asia Pacific is anticipated to hold the largest share of the asthma spacers market over the predicted timeframe.
Asia Pacific is anticipated to hold the largest share of the asthma spacers market over the predicted timeframe. The region's growth is driven by rising air pollution levels, increasing asthma prevalence, and a growing middle class with better access to healthcare. China and India lead the global stage through their substantial healthcare infrastructure investments, which support respiratory health awareness programs. The regulatory bodies are improving their product safety guidelines by creating more comprehensive standards, which will benefit market growth. Multinational corporations and local manufacturers in the Asia-Pacific region compete against each other to gain a bigger portion of the market. The rising use of telemedicine, together with digital health solutions, will create more demand for asthma spacers because these tools help patients manage their condition while following treatment guidelines.
Alkem Laboratories launched “Reliever Free India”, an asthma awareness campaign timed with World Asthma Day 2023. The initiative is designed to educate both physicians and patients about the risks of overusing reliever medications and to promote SMART therapy (Single Maintenance and Reliever Therapy), which uses just one inhaler for both daily management and symptom relief.
The National Asthma Council Australia has launched a comprehensive suite of resources to promote the use of spacers with pressurised metered-dose inhalers (pMDIs). The initiative includes posters for clinics and pharmacies, information sheets for healthcare professionals, and a step-by-step “how-to” video. Clinical experts note that nearly three-quarters of patients make errors when using inhalers, but spacer use can significantly improve medication delivery to the lungs, enhancing treatment effectiveness for both adults and children.
Europe is expected to grow at a rapid CAGR in the asthma spacers market during the forecast period. The region benefits from its strict regulatory standards, together with the increasing focus on managing respiratory health problems. The market in Germany and the UK shows expansion because of rising asthma rates and government programs that improve healthcare access. The European Medicines Agency (EMA) establishes essential regulations for medical devices that guarantee safety and effectiveness to promote industry expansion. The market demonstrates strong local manufacturer and distributor presence, which improves product availability to customers. The partnerships between healthcare providers and pharmaceutical companies create new treatment methods that lead to better patient results and higher treatment compliance.
Munich-based CAELIA Health has announced plans to transform respiratory care with its next-generation inhalation system, combining the SereneAir smart spacer and AI-powered SereneCompanion app. Accepted into the Fraunhofer AHEAD program in March 2025, the venture aims to tackle poor adherence and inhalation errors affecting over 90% of asthma patients worldwide. By offering real-time measurement of inhalation technique and personalised feedback, CAELIA Health seeks to improve outcomes for vulnerable patients, starting with pediatric asthma.
Competitive Analysis:
The report offers the appropriate analysis of the key organizations/companies involved within the asthma spacers market, along with a comparative evaluation primarily based on their product offering, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborative analysis focusing on the current news and developments of the companies, which includes product development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
List of Key Companies
- Cipla Inc.
- GlaxoSmithKline plc
- Koninklijke Philips N.V.
- Koo Medical Equipment
- Laboratoire ProtecSom
- Lupin Limited
- Mediacare Equipments (India) Pvt. Ltd.
- Monaghan Medical Corporation
- PARI GmbH
- Rossmax International Ltd.
- Teleflex Incorporated
- Others
Key Target Audience
- Market Players
- Investors
- End-users
- Government Authorities
- Consulting and Research Firm
- Venture capitalists
- Value-Added Resellers (VARs)
Recent Development
- In August 2025, CalRx, in partnership with the California Department of Public Health’s Office of School Health, announced the launch of the School Albuterol Access Initiative. Beginning Spring 2026, the program will provide TK–12 schools across California with albuterol inhalers and single-use disposable spacers at no cost for three years, under CDPH’s standing order. CalRx is currently selecting a vendor to develop a reliable distribution platform to ensure seamless delivery of these critical respiratory care tools.
- In July 2025, the BTech Class of 2026 at Ahmedabad University won InventX 2025 at IIT Jammu for a pocket-friendly asthma spacer. Her innovative design, developed with IIT Bhubaneswar teammates, earned unanimous jury acclaim and the top prize.
Market Segment
This study forecasts revenue at global, regional, and country levels from 2020 to 2035. Decisions Advisors has segmented the asthma spacers market based on the below-mentioned segments:
Global Asthma Spacers Market, By Spacer Design
- Valved Holding Chamber
- Non-Valved Holding Chamber
- Pocket-Sized Spacers
- Reusable Spacers
- Disposable Spacers
Global Asthma Spacers Market, By Material Type
- Plastic
- Metal
- Silicone
- Glass
Global Asthma Spacers Market, By Distribution Channel
- Hospital pharmacies
- Retail pharmacies
- Online pharmacies
Global Asthma Spacers Market, By Regional Analysis
- North America
- US
- Canada
- Mexico
- Europe
- Germany
- UK
- France
- Italy
- Spain
- Russia
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- Australia
- Rest of Asia Pacific
- South America
- Brazil
- Argentina
- Rest of South America
- Middle East & Africa
- UAE
- Saudi Arabia
- Qatar
- South Africa
- Rest of the Middle East & Africa
Frequently Asked Questions (FAQ)
1. How does reimbursement policy impact the adoption of asthma spacers globally?
Reimbursement frameworks significantly influence spacer adoption, particularly in developed economies. Markets with strong insurance coverage or government-funded healthcare programs demonstrate higher spacer usage rates, while limited reimbursement in emerging economies can restrict patient access.
2. What role does pediatric asthma management play in shaping market demand?
Pediatric patients represent a major demand driver, as children are more prone to inhaler technique errors. Spacers improve drug delivery efficiency and safety in younger populations, making them a preferred accessory in pediatric respiratory care protocols.
3. How are environmental sustainability trends influencing spacer manufacturing?
Manufacturers are increasingly focusing on recyclable materials, biodegradable plastics, and reduced carbon footprint production methods. Environmental regulations and eco-conscious consumers are encouraging sustainable design innovations in spacer products.
4. What technological advancements are expected to redefine asthma spacer products over the next decade?
Future developments may include smart spacers integrated with sensors, Bluetooth connectivity, dose tracking systems, and AI-powered adherence monitoring tools, enhancing patient engagement and treatment outcomes.
5. How does the competitive landscape differ between multinational and regional manufacturers?
Multinational companies typically emphasise advanced R&D and global distribution networks, while regional manufacturers compete on cost efficiency and localised production strategies tailored to specific healthcare systems.
6. What are the major barriers to spacer adoption in low- and middle-income countries?
Key barriers include affordability concerns, limited awareness among patients and caregivers, lack of standardised prescribing practices, and inconsistent supply chain infrastructure.
7. How could telehealth and remote patient monitoring influence future spacer demand?
The expansion of telemedicine and digital respiratory care platforms enables physicians to monitor inhaler technique remotely, which may increase recommendations for spacer use as part of integrated asthma management programs.
Check Licence
Choose the plan that fits you best: Single User, Multi-User, or Enterprise solutions tailored for your needs.
We Have You Covered
- 24/7 Analyst Support
- Clients Across the Globe
- Tailored Insights
- Technology Tracking
- Competitive Intelligence
- Custom Research
- Syndicated Market Studies
- Market Overview
- Market Segmentation
- Growth Drivers
- Market Opportunities
- Regulatory Insights
- Innovation & Sustainability
Report Details
| Scope | Global |
| Pages | 250 |
| Delivery | PDF & Excel via Email |
| Language | English |
| Release | Mar 2026 |
| Access | Download from this page |
