Global Biopharmaceutical and Biomedicine Market
Global Biopharmaceutical and Biomedicine Market Size, Share, By Product Type (Monoclonal Antibodies, Vaccines, Recombinant Human Insulin, Human Growth Hormone, Erythropoietin), By Application (Oncology, Cardiovascular Diseases, Neurological Disorders, Infectious Diseases), By Route of Administration (Oral, Intravenous, Subcutaneous), and By Region (North America, Europe, Asia-Pacific, Latin America, Middle East & Africa), Analysis and Forecast 2026?2035
Report Overview
Table of Contents
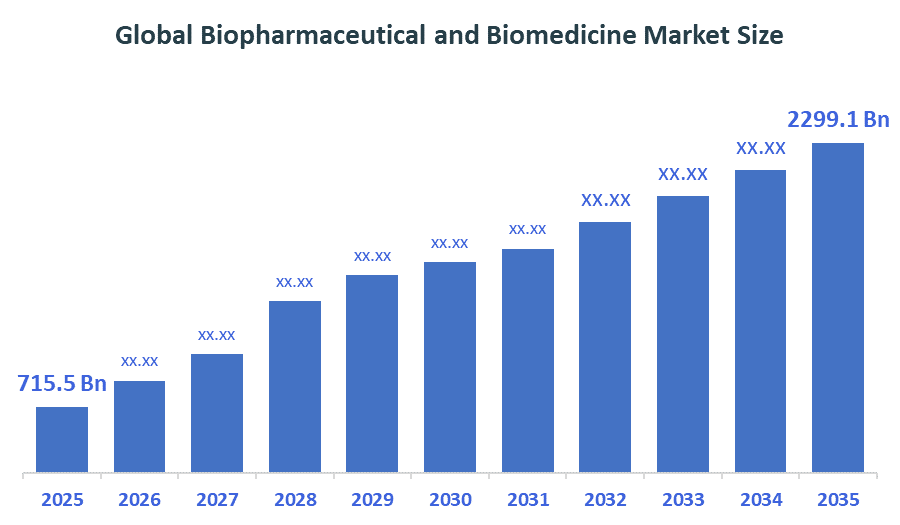
The Global Biopharmaceutical and Biomedicine Market Size is forecast to grow from USD 715.5 Billion in 2025 to approximately USD 2299.1 Billion by 2035. According to Decision Advisors, a detailed research report on the Biopharmaceutical and biomedicine market indicates that monoclonal antibodies dominate the market, accounting for approximately 63% of the total share globally. F. Hoffmann-La Roche Ltd the market with an estimated 7.5% global market share. The company reported a 2025 revenue of approximately USD 74 billion, making it one of the most influential forces shaping industry trends and overall market growth.
Market Snapshot
- Global Biopharmaceutical and Biomedicine Market Size (2025): USD 715.5 Billion
- Projected Global Biopharmaceutical and Biomedicine Market Size (2035): USD 2299.1 Billion
- Global Biopharmaceutical and Biomedicine Market Compound Annual Growth Rate (CAGR): 12.38%
- Largest Regional Market: North America
- Fastest Growing Region: Asia-Pacific
- 2nd Largest Region: Europe
- Base Year: 2025
- Historical Period: 2021–2024
- Forecast Period: 2026–2035
Market Overview/ Introduction
The Global Biopharmaceutical and Biomedicine Market refers to the worldwide industry focused on the development, production, and commercialization of advanced therapeutic products derived from biological sources to treat, prevent, and diagnose diseases. Biopharmaceuticals and biomedicine include innovative therapies such as monoclonal antibodies, vaccines, gene therapies, and cell-based treatments that leverage biotechnology for targeted and effective healthcare solutions. The market is expanding rapidly due to rising chronic disease prevalence, increasing demand for personalized medicine, and continuous advancements in technologies like gene editing and biologics manufacturing. Future opportunities lie in biosimilars, precision medicine, and emerging markets, while strong R&D investments, regulatory support, and growing adoption of advanced therapies are key factors driving sustained market growth globally.
- India’s National Biopharma Mission, supported by the World Bank, and was launched to accelerate biopharmaceutical innovation by strengthening industry–academia collaboration, funding startups, and improving clinical research infrastructure. It enabled vaccine development and affordable biologics, positioning India as a global biotech hub while enhancing innovation, accessibility, and commercialization of advanced therapies.
- The National Institutes of Health funds foundational biomedical research, supporting breakthroughs such as mRNA vaccines, gene editing, and cancer therapies. Its initiatives bridge early-stage research and commercialization, driving innovation, job creation, and global competitiveness in the biopharmaceutical market through strong public–private collaboration.
- The World Health Organization collaborates with global partners to fund programs targeting non-communicable diseases, antimicrobial resistance, and health system strengthening. These initiatives promote equitable access to advanced therapies, enhance global research collaboration, and support sustainable growth of the biopharmaceutical and biomedicine market worldwide.
Notable Insights: -
- North America is anticipated to hold the largest share of approximately 41% in the Biopharmaceutical and biomedicine market over the forecast period.
- Asia Pacific is expected to grow at a rapid CAGR of approximately 11.8% in the Biopharmaceutical and biomedicine market during the forecast period.
- The monoclonal antibodies treatment segment dominated the market in 2025, approximately 42%, and is projected to grow at a substantial CAGR during the forecast period.
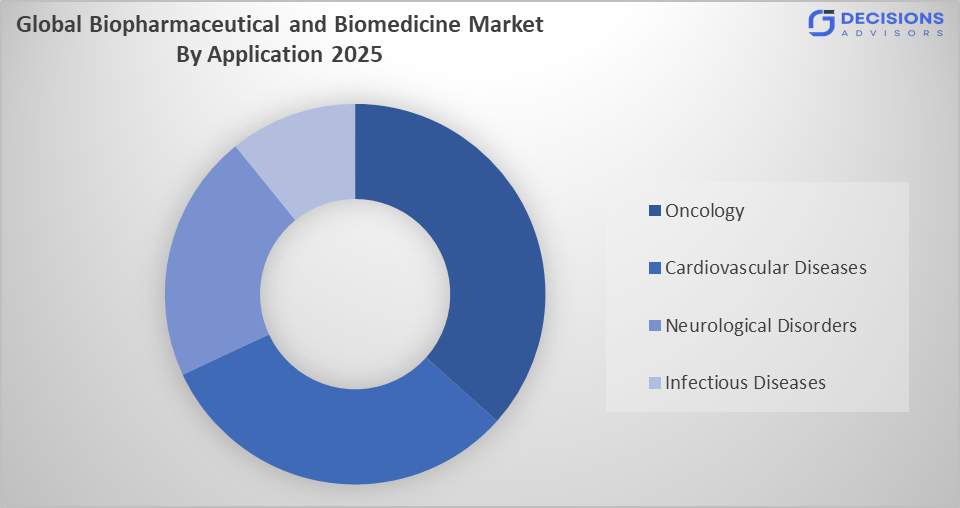
- The oncology segment dominated the market in 2025, approximately 38%, and is projected to grow at a substantial CAGR during the forecast period.
- The intravenous segment dominated the market in 2025, approximately 47%, and is projected to grow at a substantial CAGR during the forecast period.
- The compound annual growth rate of the global biopharmaceutical and biomedicine market is 12.38%.
- The market is likely to achieve a valuation of USD 2299.1 Billion by 2035.
What is role of technology in grooming the market?
Technology plays a transformative role in shaping the global biopharmaceutical and biomedicine market by accelerating drug discovery, improving precision, and reducing development costs. Advanced tools like artificial intelligence, machine learning, and high-throughput screening enable faster identification of drug candidates, while gene-editing technologies such as CRISPR gene editing support development of targeted therapies. Innovations in biologics manufacturing, including single-use systems and continuous processing, enhance scalability and efficiency. Additionally, digital health platforms and real-world data analytics improve clinical trial design and patient outcomes. Organizations like IBM Watson Health have contributed to data-driven insights in healthcare research. These technological advancements not only streamline operations but also foster personalized medicine, biosimilar development, and global accessibility, ultimately driving sustained growth and innovation in the market.
Market Drivers
The Global biopharmaceutical and biomedicine market is driven by the rising prevalence of chronic diseases such as cancer, diabetes, and autoimmune disorders, increasing demand for targeted and personalized therapies, and rapid advancements in biotechnology. Innovations like monoclonal antibodies and gene therapy are transforming treatment outcomes. Strong R&D investments by major companies such as Johnson & Johnson and Pfizer further accelerate product development. Additionally, supportive regulatory frameworks, growing healthcare expenditure, and increasing adoption of biosimilars contribute to market expansion. Expanding access to advanced treatments in emerging economies also plays a crucial role in sustaining global market growth.
Restrain
The Global biopharmaceutical and biomedicine market faces restraints such as high R&D costs, complex regulatory approvals, and lengthy clinical trial processes. Additionally, stringent compliance requirements, patent expirations, and biosimilar competition limit profitability. Supply chain disruptions and limited accessibility in developing regions further challenge overall market expansion and adoption rates globally.
Competitive Analysis:
The report offers the appropriate analysis of the key organizations/companies involved within the global biopharmaceutical and biomedicine market, along with a comparative evaluation primarily based on their product offerings, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborative analysis focusing on the current news and developments of the companies, which includes product development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
Top Companies in Global Biopharmaceutical and Biomedicine Market
- Pfizer Inc.
- Johnson & Johnson
- Roche Holding AG
- Merck & Co. Inc.
- AbbVie Inc.
- Novartis AG
- Amgen Inc.
- AstraZeneca PLC
- Bristol-Myers Squibb
- Sanofi
- Gilead Sciences Inc.
- Eli Lilly and Company
Government Initiatives
|
Country |
Key Government Initiatives |
|
US |
The U.S. government strengthened the biopharmaceutical market through initiatives led by the U.S. Food and Drug Administration, accelerating biologics approvals and supporting innovation via programs like Breakthrough Therapy designation. Increased federal funding for biotech R&D and pandemic preparedness further boosted the development of advanced therapies. |
|
UK |
Under the Life Sciences Vision, the UK government promoted biomedicine innovation by funding genomic research and speeding up clinical trials through the National Health Service. Regulatory flexibility and investment in cell and gene therapy manufacturing enhanced competitiveness and encouraged global biopharma companies to expand operations. |
|
Europe |
European governments supported the market through the European Medicines Agency and Horizon Europe funding programs, promoting biologics research and cross-border collaboration. The EU Pharmaceutical Strategy improved access to innovative medicines while ensuring safety and affordability, strengthening regional biopharmaceutical growth and innovation capacity. |
Study on the Supply, Demand, Distribution, and Market Environment of the Biopharmaceutical and Biomedicine Market
The Global Biopharmaceutical and Biomedicine Market is characterized by a complex balance of supply, demand, distribution, and market environment. Supply is driven by advanced biologics manufacturing, recombinant technologies, and strong investments from leading pharmaceutical companies, though it remains constrained by high production costs and strict regulatory standards. Demand is rising rapidly due to increasing prevalence of chronic diseases, cancer, autoimmune disorders, and aging populations worldwide. Distribution networks rely on specialized cold-chain logistics and partnerships between manufacturers, hospitals, and biotech distributors to ensure product integrity. The market environment is highly competitive and innovation-focused, supported by strong R&D pipelines, mergers, and strategic collaborations. However, pricing pressures, patent expirations, and regulatory hurdles influence market dynamics. Overall, technological advancements and personalized medicine trends are reshaping global supply-demand equilibrium and improving accessibility of biopharmaceutical therapies.
Price Analysis and Consumer Behaviour Analysis
The Global Biopharmaceutical and Biomedicine Market shows complex price dynamics driven by high R&D expenditure, advanced biologics production, and strict regulatory compliance. Pricing is generally premium due to innovation-intensive therapies, but increasing biosimilar entry and patent expirations are gradually improving affordability. Value-based pricing models and government reimbursement policies also influence final treatment costs across regions. Consumer behaviour is highly dependent on physician recommendations, hospital procurement systems, and insurance coverage. Patients are increasingly aware of advanced biologic therapies and prefer personalized and targeted treatments with better outcomes. Demand sensitivity remains higher in emerging economies due to cost constraints, while developed markets show stronger acceptance of high-cost innovative drugs. Overall pricing strategies and evolving consumer preferences are shaping competitive positioning and market accessibility globally over the forecast period and beyond worldwide demand.
Market Segmentation
The Biopharmaceutical and Biomedicine Market share is classified into product type, application, and route of administration.
- The monoclonal antibodies segment dominated the market in 2025, approximately 42%, and is projected to grow at a substantial CAGR during the forecast period.
Based on the product type, the biopharmaceutical and biomedicine market is divided into monoclonal antibodies, vaccines, recombinant human insulin, human growth hormone, and erythropoietin. Among these, the monoclonal antibodies segment dominated the market in 2025, with approximately 42%, and is projected to grow at a substantial CAGR during the forecast period. The growth of the monoclonal antibodies segment is driven by their high specificity, strong clinical efficacy, and widespread use in treating chronic and complex diseases such as cancer and autoimmune disorders. Continuous approvals, expanding therapeutic indications, and strong R&D pipelines further drive their adoption. Additionally, increasing demand for targeted therapies and personalized medicine significantly supports this segment’s leading position.
- The oncology segment dominated the market in 2025, approximately 38%, and is projected to grow at a substantial CAGR during the forecast period.
Based on the application, the biopharmaceutical and biomedicine market is divided into oncology, cardiovascular diseases, neurological disorders, and infectious diseases. Among these, the oncology segment dominated the market in 2025, approximately 38%, and is projected to grow at a substantial CAGR during the forecast period. The oncology segment dominated due to the rising global cancer burden and increasing demand for advanced biologic treatments. Biopharmaceutical innovations, including immunotherapies and targeted therapies, have significantly improved survival rates. Strong investment in cancer research, growing awareness, and favorable regulatory support for oncology drugs continue to boost this segment’s dominance in the global market.
- The intravenous segment dominated the market in 2025, approximately 47%, and is projected to grow at a substantial CAGR during the forecast period.
Based on the route of administration, the biopharmaceutical and biomedicine market is divided into oral, intravenous, and subcutaneous. Among these, the intravenous segment dominated the market in 2025, approximately 47%, and is projected to grow at a substantial CAGR during the forecast period. The growth of the intravenous segment is due to its rapid drug delivery, high bioavailability, and effectiveness in administering biologics, especially in critical care and oncology treatments. Hospitals primarily prefer this route for precise dosing and immediate therapeutic effects. Moreover, the increasing number of hospital-based treatments and complex biologic drugs further strengthens intravenous administration’s market dominance.
Strategies to Implement for Growth of the Market in Non-Leading Regions
To expand the Global Biopharmaceutical and Biomedicine Market in non-leading regions, companies should adopt localized manufacturing to reduce dependency on imports and lower production costs. Strengthening healthcare infrastructure and cold-chain logistics is essential to ensure safe distribution of biologics. Strategic partnerships with local pharmaceutical firms and government bodies can improve regulatory approvals and market penetration. Increasing investments in clinical trials within emerging economies will enhance accessibility and trust in advanced therapies. Affordable pricing models, along with biosimilar adoption, can boost patient affordability and demand. Capacity-building initiatives such as training healthcare professionals and improving diagnostic capabilities are also crucial. Additionally, digital health technologies and telemedicine can improve treatment reach in remote areas. These combined strategies will accelerate market penetration, improve accessibility, and support sustainable growth in underdeveloped and developing regions globally.
Regional Segment Analysis of the Biopharmaceutical and Biomedicine Market
- North America (U.S., Canada, Mexico)
- Europe (Germany, France, U.K., Italy, Spain, Rest of Europe)
- Asia-Pacific (China, Japan, India, Rest of APAC)
- South America (Brazil and the Rest of South America)
- The Middle East and Africa (UAE, South Africa, Rest of MEA)
North America is anticipated to hold the largest share of approximately 41% the biopharmaceutical and biomedicine market over the forecast period.
North America is anticipated to hold the largest share of approximately 41% the biopharmaceutical and biomedicine market over the forecast period. The dominance of this region is driven by advanced healthcare infrastructure, high diagnosis rates, and strong demand for antihistamines and anxiolytics. Favorable reimbursement policies, robust pharmaceutical R&D, and the presence of leading drug manufacturers further support growth. Additionally, increasing awareness of mental health and allergy treatments boosts market consumption across the region.
Asia Pacific is expected to grow at a rapid CAGR of approximately 11.8% in the biopharmaceutical and biomedicine market during the forecast period.
Asia Pacific is expected to grow at a rapid CAGR of approximately 11.8% in the biopharmaceutical and biomedicine market during the forecast period. This growth is fueled by expanding healthcare infrastructure, rising patient population, and increasing government investments in biotechnology. Countries like China and India are boosting domestic production and clinical research. Growing demand for affordable biologics and biosimilars further accelerates regional market expansion.
Europe is the 2nd largest region to grow in the biopharmaceutical and biomedicine market during the period.
Europe is the second-largest region in the global biopharmaceutical and biomedicine market due to its strong regulatory framework led by the European Medicines Agency, well-established healthcare systems, and robust R&D funding. The presence of major companies like Roche and Novartis, along with rising demand for advanced biologics, supports sustained regional growth.
Future Market Trends in Global Biopharmaceutical and Biomedicine Market: -
1. Personalized Medicine
Personalized medicine is emerging as a key trend due to advancements in genomics and biomarker research. It enables targeted therapies tailored to individual patients, improving treatment effectiveness and reducing side effects. Rising demand for precision healthcare in oncology and rare diseases is strongly driving this market shift globally.
2. Cell and Gene Therapy Expansion
Cell and gene therapy is rapidly growing due to its potential to cure previously untreatable genetic and chronic diseases. Increased clinical approvals, strong R&D investments, and breakthrough innovations are accelerating adoption. This trend is transforming treatment approaches and significantly boosting long-term market growth worldwide.
3. AI-Driven Drug Development
AI-driven drug development is reshaping the biopharmaceutical industry by reducing discovery time and improving accuracy in molecule identification. Machine learning algorithms enhance clinical trial success rates and lower costs. Growing digital transformation in healthcare is fueling adoption of AI technologies across global biomedicine research and production.
Recent Development
- In April 2026, Eli Lilly advanced its oral GLP-1 drug orforglipron (Foundayo), receiving regulatory approval and preparing for broader global rollout. These developments highlighted growing innovation and competition in metabolic disease treatments.
- In February 2026, Novo Nordisk announced positive late-stage trial results for its next-generation obesity and diabetes therapy CagriSema, a dual GLP-1/amylin drug showing superior weight loss and glucose control, positioning it for potential future launch.
- In February 2026, Gilead Sciences announced the acquisition of Arcellx for approximately $7.8 billion to strengthen its oncology pipeline, particularly focusing on the CAR-T therapy anito-cel targeting multiple myeloma. The therapy was under regulatory review with expectations for potential approval and commercial launch in late 2026, positioning it as a key innovation in cancer treatment.
How is Recent Developments Helping the Market?
Recent developments are significantly accelerating growth in the Global Biopharmaceutical and Biomedicine Market. Advances in gene therapy, monoclonal antibodies, and mRNA technology have improved treatment precision and effectiveness for complex diseases such as cancer and rare genetic disorders. Increased adoption of AI and machine learning in drug discovery has reduced development timelines and improved success rates. Expansion of biosimilars has enhanced affordability and widened patient access globally. Strategic mergers, acquisitions, and collaborations between biotech firms and pharmaceutical giants have strengthened R&D capabilities and product pipelines. Regulatory agencies are also fast-tracking approvals for breakthrough therapies, supporting quicker market entry. Additionally, improvements in biomanufacturing technologies and cold-chain logistics have enhanced supply efficiency. Together, these developments are driving innovation, reducing costs, and expanding global accessibility of advanced biopharmaceutical and biomedicine solutions significantly.
Market Segment
This study forecasts revenue at global, regional, and country levels from 2020 to 2035. Decision Advisors has segmented the biopharmaceutical and biomedicine market based on the below-mentioned segments:
Global Biopharmaceutical and Biomedicine Market, By Product Type
- Monoclonal Antibodies
- Vaccines
- Recombinant Human Insulin
- Human Growth Hormone
- Erythropoietin
Global Biopharmaceutical and Biomedicine Market, By Application
- Oncology
- Cardiovascular Diseases
- Neurological Disorders
- Infectious Diseases
Global Biopharmaceutical and Biomedicine Market, By Route of Administration
- Oral
- Intravenous
- Subcutaneous
Global Biopharmaceutical and Biomedicine Market, By Regional Analysis
- North America
- US
- Canada
- Mexico
- Europe
- Germany
- UK
- France
- Italy
- Spain
- Russia
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- Australia
- Rest of Asia Pacific
- South America
- Brazil
- Argentina
- Rest of South America
- Middle East & Africa
- UAE
- Saudi Arabia
- Qatar
- South Africa
- Rest of the Middle East & Africa
Frequently Asked Questions (FAQ)
Q. What are the key challenges affecting the scalability of the Global Biopharmaceutical and Biomedicine Market?
A. The market faces scalability challenges due to high capital investment requirements, complex biologics manufacturing processes, and strict global regulatory compliance. Limited skilled workforce in advanced biotechnology and dependency on cold-chain logistics further restrict large-scale production and distribution. These factors collectively slow down rapid global expansion despite rising demand for biologic therapies.
Q. How do biosimilars influence competition in the Global Biopharmaceutical and Biomedicine Market?
A. Biosimilars significantly intensify competition by offering cost-effective alternatives to expensive biologic drugs. They help reduce treatment costs and increase patient accessibility, especially in emerging economies. Their growing approvals across major regulatory bodies also pressure innovator companies to innovate continuously, thereby reshaping pricing strategies and improving overall market affordability.
Q. What role do clinical trials play in shaping market growth and innovation?
A. Clinical trials are essential for validating the safety and efficacy of new biologic therapies. They accelerate innovation by enabling the development of targeted treatments such as immunotherapies and gene therapies. Expansion of multinational clinical trial networks also helps companies access diverse patient populations, improving drug effectiveness and speeding up global product approvals.
Q. How is patient awareness influencing the Global Biopharmaceutical and Biomedicine Market?
A. Rising patient awareness about advanced treatment options is increasing demand for biologics and personalized medicine. Patients are more informed through digital health platforms and are actively seeking targeted therapies with fewer side effects. This shift in awareness is encouraging healthcare providers and companies to focus more on innovative, patient-centric treatment solutions.
Check Licence
Choose the plan that fits you best: Single User, Multi-User, or Enterprise solutions tailored for your needs.
We Have You Covered
- 24/7 Analyst Support
- Clients Across the Globe
- Tailored Insights
- Technology Tracking
- Competitive Intelligence
- Custom Research
- Syndicated Market Studies
- Market Overview
- Market Segmentation
- Growth Drivers
- Market Opportunities
- Regulatory Insights
- Innovation & Sustainability
Report Details
| Scope | Global |
| Pages | 210 |
| Delivery | PDF & Excel via Email |
| Language | English |
| Release | Apr 2026 |
| Access | Download from this page |
