Global Bispecific Antibodies Market
Global Bispecific Antibodies Market Size, Share, and COVID-19 Impact Analysis, By Type (IgG-based Bispecific Antibodies, ScFv-based Bispecific Antibodies, Bispecific T-cell Engager Antibodies), By Mechanism of Action (Direct Cell-killing, Immune Modulation, Cytokine Release), By Application (Cancer Treatment, Autoimmune Disorders, Infectious Diseases, Transplant Rejection), By End Use (Pharmaceutical Companies, Research Institutions, Clinical Research Organizations), and By Region (North America, Europe, Asia-Pacific, Latin America, Middle East, and Africa), Analysis and Forecast 2025 - 2035.
Report Overview
Table of Contents
Global Bispecific Antibodies Market Insights Forecasts to 2035
- The Global Bispecific Antibodies Market Size Was Estimated at USD 5.65 billion in 2024
- The Market Size is Expected to Grow at a CAGR of around 44.54 % from 2025 to 2035
- The Worldwide Bispecific Antibodies Market Size is Expected to Reach USD 325.11 billion by 2035
- North America is expected to grow the fastest during the forecast period.
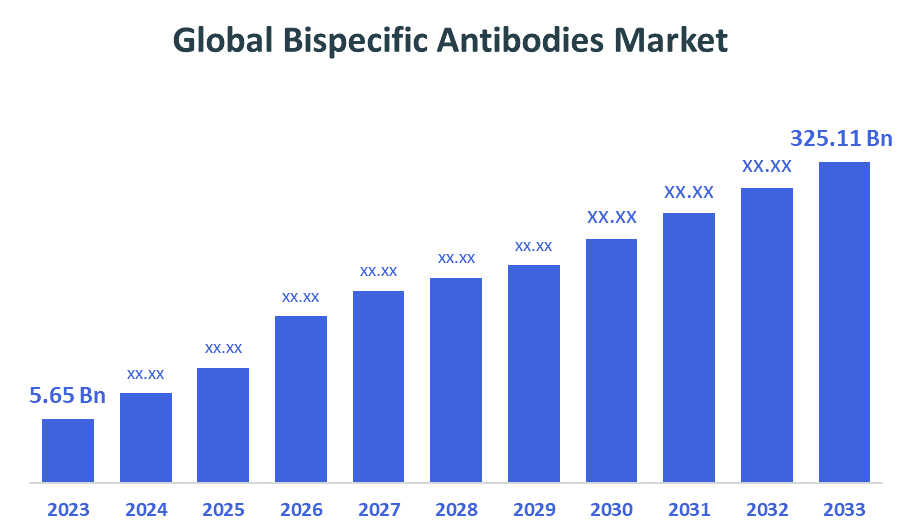
According to a research report published by Decisions Advisors and Consulting, The Global Bispecific Antibodies Market Size was worth around USD 5.65 billion in 2024 and is predicted to grow to around USD 325.11 billion by 2035 with a compound annual growth rate (CAGR) of 44.54 % from 2025 to 2035. The market for bispecific antibodies is expanding due in large part to the increase in cancer incidence. Therapeutic proteins known as bispecific antibodies (BsAbs) bind to two different antigens or epitopes. Bispecific antibodies, in contrast to conventional monoclonal antibodies, which typically target a single antigen, have special capacities by engaging several targets, enabling novel therapeutic modes of action and increased efficacy in treating a variety of illnesses.
Market Overview
The bispecific antibodies market includes a novel class of synthetic proteins designed to tackle difficult illnesses and treatment issues. Moreover, their dual specificity allows these antibodies to attach to two different targets in the biological system at the same time, such as antigens or receptors. Two or more antigen-binding components from different parental antibodies are fused to create bispecific antibodies. A single construct that can engage several targets at once is produced by this fusion. Different formats, such as full-length immunoglobulins, antibody fragments, or other designed protein scaffolds, are possible due to the design flexibility. There are numerous therapeutic benefits to bispecific antibodies' special capacity to connect different cellular or molecular targets. For example, bispecific antibodies can target both immune cells and malignant cells in cancer treatment. Bispecific antibodies' adaptability and specificity have sparked a great deal of curiosity and creativity in medication creation. Numerous illnesses, such as cancer, autoimmune diseases, viral diseases, and inflammatory ailments, may be treated by them. Bispecific antibodies have the potential to significantly influence precision medicine and tailored therapies in the future as long as research and development efforts continue.
Report Coverage
This research report categorizes the bispecific antibodies market based on various segments and regions, forecasts revenue growth, and analyzes trends in each submarket. The report analyses the key growth drivers, opportunities, and challenges influencing the bispecific antibodies market. Recent market developments and competitive strategies, such as expansion, product launch, development, partnership, merger, and acquisition, have been included to draw the competitive landscape in the market. The report strategically identifies and profiles the key market players and analyzes their core competencies in each sub-segment of the bispecific antibodies market.
Driving Factors
The development of bispecific antibodies is being fueled by pharmaceutical and biotechnology companies' increasing investments in R&D. These expenditures are intended to increase the effectiveness of current therapies and investigate novel therapeutic targets. The market for bispecific antibodies is growing as a result of the increasing use of these antibodies in many therapeutic fields. The development of bispecific antibodies with enhanced safety and efficacy profiles has been made possible by continuous improvements in antibody engineering and technology. Antibody-drug conjugates and Fc engineering are examples of novel technologies that are expanding the therapeutic potential of bispecific antibodies. Innovative treatment is becoming more and more necessary as chronic illnesses like cancer and autoimmune disorders become more common.
UCB signed a collaboration worth up to $1.1 billion with Chinese biotech Antengene to develop ATG-201, a CD19/CD3 bispecific T-cell engager (TCE) for autoimmune diseases. Antengene will conduct initial Phase 1 trials in China and Australia before transferring development rights to UCB, which will then take the candidate forward globally.
Restraining Factors
Bispecific antibody treatments must fulfil stringent regulatory requirements, particularly those about safety, efficacy, and manufacturing quality, before being licensed. Moreover, bispecific antibodies are unique, and the regulatory environment is always shifting; navigating the regulatory path for these molecules can be challenging.
Market Segmentation
The bispecific antibodies market share is classified into type, mechanism of action, application, and end use.
- The IgG-based bispecific antibodies segment accounted for the largest share in 2024 and is anticipated to grow at a remarkable CAGR during the forecast period.
Based on the type, the bispecific antibodies market is segmented into IgG-based bispecific antibodies, scFv-based bispecific antibodies, and bispecific T-cell engager antibodies. Among these, the IgG-based bispecific antibodies segment accounted for the largest share in 2024 and is anticipated to grow at a remarkable CAGR during the forecast period. Their strong performance and capacity to stimulate the immune system, antibodies are often employed in autoimmune disorders and oncology. A large body of clinical evidence demonstrating their efficacy and safety supports their supremacy.
Zenas Pharma reported positive Phase III data in IgG4-related disease (IgG4-RD), paving the way for a Biologics License Application (BLA). Despite the strong clinical results, the company’s stock price dropped sharply, reflecting investor concerns about commercialisation timelines and competitive dynamics.
- The immune modulation segment accounted for the largest share in 2024 and is anticipated to grow at a significant CAGR during the forecast period.
Based on the mechanism of action, the bispecific antibodies market is differentiated into direct cell-killing, immune modulation, and cytokine release. Among these, the immune modulation segment accounted for the largest share in 2024 and is anticipated to grow at a significant CAGR during the forecast period. The segment share is currently ruled by immune modulation, which offers effective therapeutic approaches that take advantage of the immune system's natural capacities. This section demonstrates remarkable adaptability across several malignancy side effects by facilitating the activation of T-cells and other immune responses
EpiBiologics has secured $107 million in Series B financing to advance its precision protein degradation platform for cancer and immune-related diseases.
- The cancer treatment segment accounted for the highest revenue in 2024 and is anticipated to grow at a substantial CAGR over the forecast period.
Based on the application, the bispecific antibodies market is divided into cancer treatment, autoimmune disorders, infectious diseases, and transplant rejection. Among these, the cancer treatment segment accounted for the highest revenue in 2024 and is anticipated to grow at a substantial CAGR over the forecast period. With a strong reliance on targeted medicines and customised medicine techniques, cancer therapy continues to hold a dominant market share. Bispecific antibodies offer novel therapy approaches that improve efficacy and lessen negative effects as the field of oncology develops.
InduPro Therapeutics and Eli Lilly announced a global strategic collaboration and licensing agreement to develop first-in-class bispecific and multispecific oncology therapeutics using InduPro’s proprietary proximity-guided platform. The deal also includes an equity investment from Lilly, underscoring its confidence in InduPro’s novel approach.
- The pharmaceutical companies segment accounted for the highest market revenue in 2024 and is expected to grow at a substantial CAGR over the forecast period.
Based on the end use, the bispecific antibodies market is divided into pharmaceutical companies, research institutions, and clinical research organisations. Among these, the pharmaceutical companies segment accounted for the highest market revenue in 2024 and is expected to grow at a substantial CAGR over the forecast period. Their strong market access tactics and in-depth understanding of medication development. With substantial financing for R&D, they frequently take the lead in introducing novel treatments to the market. Their well-established networks enable effective marketing and distribution of bispecific antibodies.
Asahi Kasei announced that it will acquire German biotech AiCuris Anti-infective Cures AG for approximately €780 million (~$920 million), strengthening its global pharmaceutical platform with a focus on severe infectious diseases. The deal is expected to close in Q1 FY2026 and contribute positively to operating income from fiscal 2028 onward.
Regional Segment Analysis of the Bispecific Antibodies Market
- North America (U.S., Canada, Mexico)
- Europe (Germany, France, U.K., Italy, Spain, Rest of Europe)
- Asia-Pacific (China, Japan, India, Rest of APAC)
- South America (Brazil and the Rest of South America)
- The Middle East and Africa (UAE, South Africa, Rest of MEA)
Asia Pacific is anticipated to hold the largest share of the bispecific antibodies market over the predicted timeframe.
Asia Pacific is anticipated to hold the largest share of the bispecific antibodies market over the predicted timeframe. The Asia-Pacific area has experienced remarkable expansion and has enormous potential in the global market. Countries like China, Japan, and South Korea made substantial contributions to manufacturing, research, and development. The region's booming biotechnology sector, burgeoning patient population, and rising healthcare spending all contributed to the Asia Pacific's market domination.
In China, ImmuneCare, a subsidiary of ImmuneOnco, has filed a patent for IMC-015, a bispecific antibody designed with a differentiated “fat-loss & muscle-gain” strategy aimed at countering GLP-1-induced muscle loss.
North America is expected to grow at a rapid CAGR in the bispecific antibodies market during the forecast period. The region's high prevalence of chronic diseases, substantial R&D investments, and sophisticated healthcare infrastructure are the main drivers of its growth. Innovative medications have been approved more quickly thanks to regulatory support from organisations like the FDA, which has improved market dynamics. The competitive environment is defined by important players' strategic partnerships and ongoing innovation. The USA and Canada contribute to the market as well, concentrating on R&D projects. The existence of these industry titans guarantees a strong pipeline of bispecific antibody treatments, serving a variety of therapeutic domains.
Alligator Bioscience has secured a U.S. patent for its bispecific antibody platform, reinforcing its position in next-generation immuno-oncology. Next-generation immunotherapies that target immune modulators and tumour-associated antigens across a range of cancer indications and combination methods are intended to be powered by the protected architecture.
Competitive Analysis:
The report offers the appropriate analysis of the key organizations/companies involved within the bispecific antibodies market, along with a comparative evaluation primarily based on their product offering, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborative analysis focusing on the current news and developments of the companies, which includes product development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
List of Key Companies
- Amgen Inc
- AstraZeneca
- Eli Lilly
- Emergent BioSolutions Inc
- F. Hoffmann-La Roche Ltd
- Genmab A/S
- MacroGenics, Inc
- Mereo BioPharma Group plc
- Merus
- Pieris Pharmaceuticals, Inc
- Regeneron Pharmaceuticals Inc
- Sanofi
- Sobi, TG Therapeutics Inc
- Xencor
- Others
Key Target Audience
- Market Players
- Investors
- End-users
- Government Authorities
- Consulting and Research Firm
- Venture capitalists
- Value-Added Resellers (VARs)
Recent Development
- In March 2026, Earendil Labs and WuXi Biologics announced a strategic collaboration to accelerate the development and manufacturing of bispecific/multispecific antibodies and antibody-drug conjugates (ADCs), targeting autoimmune diseases, cancer, and other conditions. The partnership combines Earendil’s AI-driven biologics R&D with WuXi Biologics’ global CRDMO capabilities.
- In January 2026, AbbVie and RemeGen announced an exclusive licensing agreement to co-develop a novel bispecific antibody for advanced solid tumours. This deal positions AbbVie to expand its oncology pipeline with cutting-edge bispecific technology, while RemeGen secures a powerful global partner to bring its innovation to market.
Market Segment
This study forecasts revenue at global, regional, and country levels from 2020 to 2035. Decisions Advisors has segmented the bispecific antibodies market based on the below-mentioned segments:
Global Bispecific Antibodies Market, By Type
- IgG-based Bispecific Antibodies
- ScFv-based Bispecific Antibodies
- Bispecific T-cell Engager Antibodies
Global Bispecific Antibodies Market, By Mechanism of Action
- Direct Cell-killing
- Immune Modulation
- Cytokine Release
Global Bispecific Antibodies Market, By Application
- Cancer Treatment
- Autoimmune Disorders
- Infectious Diseases
- Transplant Rejection
Global Bispecific Antibodies Market, By End Use
- Pharmaceutical Companies
- Research Institutions
- Clinical Research Organisations
Global Bispecific Antibodies Market, By Regional Analysis
- North America
- US
- Canada
- Mexico
- Europe
- Germany
- UK
- France
- Italy
- Spain
- Russia
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- Australia
- Rest of Asia Pacific
- South America
- Brazil
- Argentina
- Rest of South America
- Middle East & Africa
- UAE
- Saudi Arabia
- Qatar
- South Africa
- Rest of the Middle East & Africa
Frequently Asked Questions (FAQ)
1. What are the major challenges in manufacturing bispecific antibodies at a commercial scale?
Manufacturing bispecific antibodies is more complex than producing traditional monoclonal antibodies because they require the precise assembly of two different antigen-binding sites. Ensuring correct pairing of heavy and light chains, maintaining structural stability, and achieving high production yields remain key technical challenges. Advanced protein engineering techniques and improved bioprocessing platforms are helping companies address these issues, but manufacturing scalability and cost efficiency continue to be significant concerns for large-scale commercialisation.
2. How do bispecific antibodies compare with monoclonal antibodies in terms of therapeutic effectiveness?
Bispecific antibodies have the advantage of targeting two different antigens simultaneously, which can enhance therapeutic effectiveness in certain diseases. For example, they can redirect immune cells directly toward tumour cells, potentially increasing tumour-killing efficiency. However, the clinical benefits depend on the specific disease, antibody design, and mechanism of action. In some cases, monoclonal antibodies remain effective and easier to produce, so treatment choice often depends on the clinical scenario.
3. What role does artificial intelligence play in the development of bispecific antibodies?
Artificial intelligence is increasingly used in antibody discovery and engineering. AI algorithms can analyse biological data to identify potential antigen targets, predict protein structures, and optimise antibody design. This can significantly shorten the development timeline for new bispecific antibodies and improve their safety and efficacy profiles by predicting potential off-target interactions and stability issues during early research stages.
4. Which emerging therapeutic areas could drive future demand for bispecific antibodies beyond oncology?
Although oncology is currently the primary application, emerging areas such as neurodegenerative diseases, metabolic disorders, and rare genetic diseases are being explored. Researchers are investigating whether bispecific antibodies can cross biological barriers, modulate immune responses in neurological conditions, or target multiple metabolic pathways simultaneously. These potential applications could expand the market significantly in the long term.
5. How do pricing and reimbursement policies influence the adoption of bispecific antibody therapies?
Bispecific antibody treatments can be expensive due to complex development and manufacturing processes. Pricing strategies and reimbursement policies from governments and insurance providers play a crucial role in determining patient access. In markets where reimbursement frameworks support advanced biologics, adoption tends to be higher. Conversely, limited reimbursement coverage can slow market uptake even when clinical benefits are significant.
6. What regulatory considerations are unique to bispecific antibody therapies?
Regulators often require extensive preclinical and clinical data to evaluate the safety of bispecific antibodies because they interact with multiple biological targets simultaneously. Potential risks such as excessive immune activation, cytokine release syndrome, and off-target effects must be carefully assessed. As a result, regulatory agencies may require additional safety monitoring and complex clinical trial designs compared with traditional biologics.
7. How could combination therapies influence the future market growth of bispecific antibodies?
Combination therapies involving bispecific antibodies and other treatment modalities—such as immune checkpoint inhibitors, targeted therapies, or chemotherapy—are an important area of research. Combining different therapeutic approaches may improve treatment outcomes by addressing multiple disease mechanisms simultaneously. If clinical trials demonstrate improved survival rates or reduced resistance, combination therapies could significantly expand the commercial potential of bispecific antibodies.
Check Licence
Choose the plan that fits you best: Single User, Multi-User, or Enterprise solutions tailored for your needs.
We Have You Covered
- 24/7 Analyst Support
- Clients Across the Globe
- Tailored Insights
- Technology Tracking
- Competitive Intelligence
- Custom Research
- Syndicated Market Studies
- Market Overview
- Market Segmentation
- Growth Drivers
- Market Opportunities
- Regulatory Insights
- Innovation & Sustainability
Report Details
| Scope | Global |
| Pages | 240 |
| Delivery | PDF & Excel via Email |
| Language | English |
| Release | Mar 2026 |
| Access | Download from this page |
