Global Cardiogenic Shock Market
Global Cardiogenic Shock Market Size, Share, and COVID-19 Impact Analysis, By Treatment Type (In-Vitro Test Kits, Drugs and Devices), By End User (Cardiac Catheterization Labs, Hospitals, Ambulatory Surgical Centres and Others), and By Region (North America, Europe, Asia-Pacific, Latin America, Middle East, and Africa), Analysis and Forecast 2025-2035
Report Overview
Table of Contents
Global Cardiogenic Shock Market Size Insights Forecasts to 2035
- The Global Cardiogenic Shock Market Size Was Estimated at USD 3.89 Billion in 2024
- The Market Size is Expected to Grow at a CAGR of around 6.48 % from 2025 to 2035
- The Worldwide Cardiogenic Shock Market Size is Expected to Reach USD 7.76 Billion by 2035
- North America is expected to grow the fastest during the forecast period.
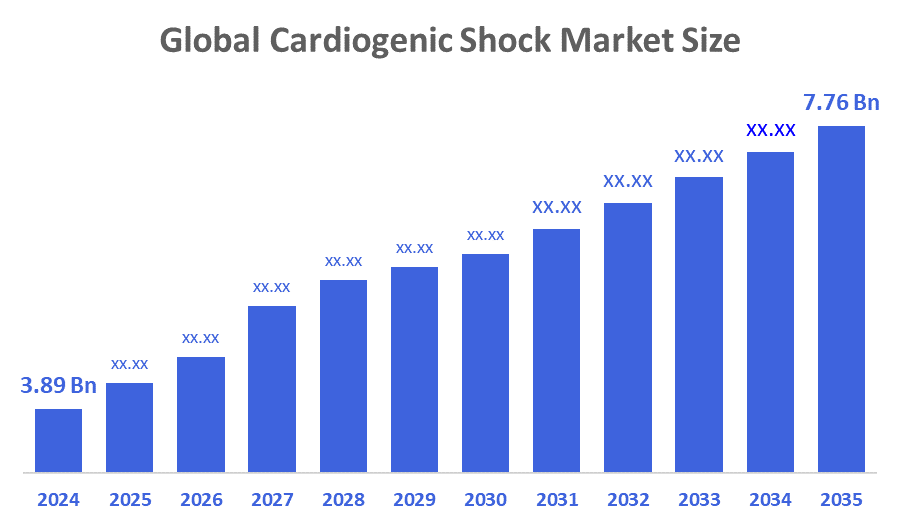
According to a research report published by Decisions Advisors and Consulting, The Global Cardiogenic Shock Market Size Was Worth Around USD 3.89 Million In 2024 And Is Predicted To Grow To Around USD 7.76 Million By 2035 With A Compound Annual Growth Rate (CAGR) Of 6.48 % From 2025 To 2035. The industry is expected to see innovation in minimally invasive hemodynamic smart technologies by 2035. AI predictive analytics for the early diagnosis of cardiogenic shock is also gaining momentum. The industry will benefit from improved access to healthcare in developing countries, and the current clinical trials for the next-generation vasodilators and gene therapies have the potential to change the treatment landscape in the coming decade.
Market Overview
The cardiogenic shock treatment market refers to the global healthcare sector focused on therapies, devices, and interventions used to manage cardiogenic shock a life-threatening condition where the heart fails to pump enough blood to meet the body’s needs. Cardiogenic shock is a serious form of heart failure mainly caused by major damage to the heart muscle, often from a large heart attack. This damage weakens the heart's ability to pump blood, resulting in low cardiac output and reduced blood flow to the body. The body initially tries to maintain blood flow through mechanisms like blood vessel constriction and fluid retention, but these can worsen tissue oxygen deprivation, lung swelling, and failure of multiple organs. Inflammation and problems in the small blood vessels also worsen cardiogenic shock, creating a cycle of declining heart function and decreased blood flow. Diagnosing cardiogenic shock focuses on recognising lasting low cardiac output along with signs of poor blood flow to the tissues. These signs may include confusion, cold hands and feet, low urine output, and high lactate levels. Additional lab tests and invasive monitoring may help confirm the diagnosis and distinguish cardiogenic shock from other types of shock and worsening chronic heart failure.
4TEEN4 Pharmaceuticals GmbH, a German biotech company, secured $64 million in Series C financing to advance research on a new cardiogenic shock drug called procizumab. The therapy works by capturing circulating dipeptidyl peptidase 3 (cDPP3), a key molecular driver of cardiogenic shock, aiming to reduce the condition’s extremely high mortality rate.
Report Coverage
This research report categorizes the cardiogenic shock market based on various segments and regions, forecasts revenue growth, and analyzes trends in each submarket. The report analyses the key growth drivers, opportunities, and challenges influencing the cardiogenic shock market. Recent market developments and competitive strategies, such as expansion, product launch, development, partnership, merger, and acquisition, have been included to draw the competitive landscape in the market. The report strategically identifies and profiles the key market players and analyzes their core competencies in each sub-segment of the cardiogenic shock market.
Driving Factors
The market for treating cardiogenic shock is expanding rapidly due to advancements in mechanical circulatory support technology, artificial intelligence (AI) assisted diagnoses, and an increase in access to critical care in developing nations. There are numerous opportunities for large pharmaceutical and medical technology companies to participate in this newly developing area by acquiring or developing new technologies or therapies to meet the growing demands of this evolving field. If not properly upgraded, their existing systems may become obsolete. Cardiogenic shock management is expanding rapidly, driven by rising acute cardiovascular events, ageing populations, and improved ICU practices. Adoption of mechanical circulatory support devices, novel pharmacological therapies, driven monitoring, and early detection initiatives, alongside growing R&D investments and wider access to advanced cardiac care, are transforming outcomes and fueling market growth.
Restraining Factors
The growth of the Global Cardiogenic Shock Market Size is significantly limited by the few clinical practices and guidelines for managing the condition. Even though cardiovascular diseases are becoming more common, the absence of standardised and effective treatment protocols for cardiogenic shock creates major challenges. This shortfall in clinical practices results in inconsistent treatment outcomes and slows down the development and use of new therapies.
Market Segmentation
The cardiogenic shock market share is classified into treatment type and end user.
- The drugs segment accounted for the largest share in 2024 and is anticipated to grow at a significant CAGR during the forecast period.
Based on the treatment type, the cardiogenic shock market is differentiated into in-vitro test kits, drugs and devices. Among these, the drugs segment accounted for the largest share in 2024 and is anticipated to grow at a significant CAGR during the forecast period. Even as the approved cardiovascular medications expand their indications and give credence to the industry growth, drug-based treatment is still the front-line therapy approach. The inotropes segment accounts for a significant majority of the global market, as inotropes are the first line of treatment of hypotension and end-organ hypoperfusion owing to cardiogenic shock.
- The hospitals segment accounted for the highest market revenue in 2024 and is anticipated to grow at a significant CAGR during the forecast period.
Based on the end user, the cardiogenic shock market is divided into cardiac catheterisation labs, hospitals, ambulatory surgical centres and others. Among these, the hospitals segment accounted for the highest market revenue in 2024 and is anticipated to grow at a significant CAGR during the forecast period. This segment growth reflects device miniaturisation and operator familiarity. It help stabilize shock in complex PCI cases and reduces procedure-to-support times to less than 45 minutes and shortens ICU stay.
Regional Segment Analysis of the Cardiogenic Shock Market
- North America (U.S., Canada, Mexico)
- Europe (Germany, France, U.K., Italy, Spain, Rest of Europe)
- Asia-Pacific (China, Japan, India, Rest of APAC)
- South America (Brazil and the Rest of South America)
- The Middle East and Africa (UAE, South Africa, Rest of MEA)
Asia Pacific is anticipated to hold the largest share of the cardiogenic shock market over the predicted timeframe.
Asia Pacific is anticipated to hold the largest share of the cardiogenic shock market over the predicted timeframe. Asia Pacific is projected to grow rapidly due to an increase in raw material production for drugs and the provision of manufacturing facilities to take care of clinical needs. In addition, the increasing incidence of cardiovascular disease, cardiogenic shock, economic burdens and the ageing population all point toward continued market growth as well. The health sector has experienced vast improvements in recent years, such as the optimisation of devices that are used in cardiovascular devices, the advent of minimally invasive procedures, and the ability to develop clinical diagnostics, clinical advances and provide management of very high-risk patients, which will contribute to the development of this market in the future.
The Japan Critical Care Cardiology Committee (J4CS) published its 2025 expert consensus statement on cardiogenic shock management, developed using the Delphi method. The guidance emphasises the rapid progression challenges of cardiogenic shock.
North America is expected to grow at a rapid CAGR in the cardiogenic shock market during the forecast period. The cardiogenic shock market is expanding as awareness continues to rise about having a healthy heart, as well as the number of cases of cardiovascular disease in North America. Rapid advancements in, and ongoing research into, the methods and technology for treating cardiac disease also support this trend. The FDA's Breakthrough Device Program is helping provide accelerated pathways for new innovative treatments (e.g., new generations of mechanical cardiac support devices – such as ECMO and pVADs). The increased use of AI to detect early shock in patients will continue to improve patient outcomes and drive new growth in the industry. The reimbursement rates set by Medicare and Medicaid will continue to play an important role in the acceptance of therapy, particularly for high-value therapies. Additionally, supply chains and health care costs will continue to drive growth.
Competitive Analysis:
The report offers the appropriate analysis of the key organizations/companies involved within the cardiogenic shock market, along with a comparative evaluation primarily based on their product offering, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborative analysis focusing on the current news and developments of the companies, which includes product development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
List of Key Companies
- Abiomed (Johnson & Johnson)
- Berlin Heart
- Biotronik
- Boston Scientific
- Cardiac Assist
- CardiacAssist/TandemLife
- Edwards Lifesciences
- Getinge AB
- LivaNova
- Maquet (Getinge Group)
- Medtronic
- Teleflex
- Terumo Corporation
- Thoratec (part of Abbott Laboratories)
- ZOLL Medical (Asahi Kasei Group)
- Others
Key Target Audience
- Market Players
- Investors
- End-users
- Government Authorities
- Consulting and Research Firm
- Venture capitalists
- Value-Added Resellers (VARs)
Recent Development
- In November 2025, the Society of Cardiovascular Angiography and Interventions (SCAI) launched the Door to Lactate Clearance (DLC) Cardiogenic Shock Initiative, focusing on measuring and rapidly lowering lactate levels to improve survival. The initiative is based on both new evidence and experience and seeks to integrate time-critical metabolic targets into the mainstream of shock management, irrespective of the devices that the hospital possesses.
- In September 2025, the Society for Cardiovascular Angiography and Interventions (SCAI) launched the Door to Lactate Clearance (DLC) Cardiogenic Shock Initiative. It was aiming to make lactate clearance a central marker for early diagnosis and management of cardiogenic shock.
- In March 2025, Johnson & Johnson Medtech showcased its newest generation of the Impella heart pump and presented long-term survival data at the TCT 2025 conference in San Francisco. Now, robust RCT evidence supports the survival benefit of Impella CP in cardiogenic shock, thanks to the danger shock trial published in the New England Journal of Medicine.
Market Segment
This study forecasts revenue at global, regional, and country levels from 2020 to 2035. Decisions Advisors has segmented the cardiogenic shock market based on the below-mentioned segments:
Global Cardiogenic Shock Market, By Treatment Type
- In-Vitro Test Kits
- Drugs
- Devices
Global Cardiogenic Shock Market, By End User
- Cardiac Catheterisation Labs
- Hospitals
- Ambulatory Surgical Centres
- Others
Global Cardiogenic Shock Market, By Regional Analysis
- North America
- US
- Canada
- Mexico
- Europe
- Germany
- UK
- France
- Italy
- Spain
- Russia
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- Australia
- Rest of Asia Pacific
- South America
- Brazil
- Argentina
- Rest of South America
- Middle East & Africa
- UAE
- Saudi Arabia
- Qatar
- South Africa
- Rest of the Middle East & Africa
Frequently Asked Questions (FAQ)
- What is the market size and growth rate?
The market was valued at USD 3.89 billion in 2024 and is expected to reach USD 7.76 billion by 2035, growing at a CAGR of 6.48% from 2025 to 2035.
- What is cardiogenic shock?
It's a life-threatening condition where the heart fails to pump enough blood, often due to a major heart attack, causing low output, organ failure, cold extremities, confusion, and high lactate levels.
- Which treatment type leads the market?
Drugs hold the largest share in 2024 and are projected to grow at a significant CAGR, with inotropes as frontline therapy for hypotension and hypoperfusion.
- What end-user segment dominates revenue?
Hospitals accounted for the highest revenue in 2024 and are expected to grow at a significant CAGR, due to device miniaturisation and efficient shock stabilisation in complex cases.
- Which regions are leading?
Asia-Pacific holds the largest share due to rising cardiovascular cases, ageing populations, and healthcare improvements in China, Japan, and India; North America grows fastest with advanced tech.
- What drives market growth?
Advancements in mechanical support, AI diagnostics, rising acute heart events, ageing demographics, R&D in vasodilators/gene therapies, and better ICU access fuel expansion.
- What restrains the market?
Lack of standardised clinical guidelines and practices leads to inconsistent outcomes, slowing new therapy adoption despite rising cardiovascular prevalence.
- What are the key recent developments?
4TEEN4 raised $64M for procizumab (cDPP3 therapy); SCAI's Door to Lactate Clearance Initiative (Sep/Nov 2025); J&J's Impella data at TCT 2025; Japan J4CS consensus (2025).
Check Licence
Choose the plan that fits you best: Single User, Multi-User, or Enterprise solutions tailored for your needs.
We Have You Covered
- 24/7 Analyst Support
- Clients Across the Globe
- Tailored Insights
- Technology Tracking
- Competitive Intelligence
- Custom Research
- Syndicated Market Studies
- Market Overview
- Market Segmentation
- Growth Drivers
- Market Opportunities
- Regulatory Insights
- Innovation & Sustainability
Report Details
| Scope | Global |
| Pages | 240 |
| Delivery | PDF & Excel via Email |
| Language | English |
| Release | Feb 2026 |
| Access | Download from this page |
