Global Cell and Gene Therapies in Rare Disorders Market
Global Cell and Gene Therapies in Rare Disorders Market Size, Share, By Therapy Type (Gene Therapy, Cell Therapy, Gene-Modified Cell Therapy), By Vector Type (Viral Vectors and Non-Viral Vectors), By Application (Rare Genetic Disorders, Rare Cancers, Neurological Disorders, Metabolic Disorders), By Region (North America, Europe, Asia-Pacific, Latin America, Middle East, and Africa), Analysis and Forecast 2025 ? 2035.
Report Overview
Table of Contents
Market Snapshot
- Market Size (2025): USD 3.27 Billion
- Projected Market Size (2035): USD 42.8 Billion
- Compound Annual Growth Rate (CAGR): 26.34%
- Largest Regional Market: North America
- Fastest Growing Region: Asia Pacific
- 2nd Largest Region: Europe
- Base Year: 2025
- Historical Period: 2021–2024
- Forecast Period: 2025–2035
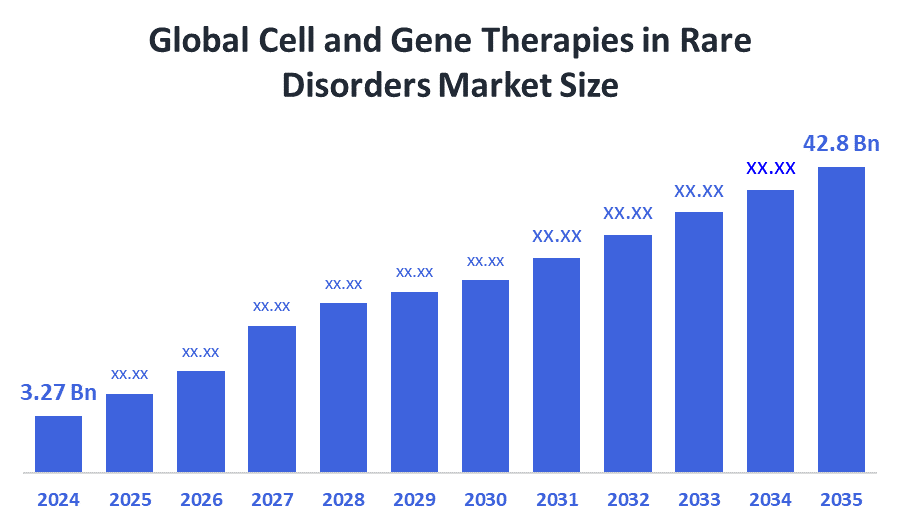
According to Decision Advisors, the Global Cell and Gene Therapies in Rare Disorders Market Size is expected to grow from USD 3.27 Billion in 2025 to USD 42.8 Billion by 2035, at a CAGR of 26.34% during the forecast period 2025-2035. The global cell and gene therapies in rare disorders market is projected to grow significantly over the next decade due to the increasing investigational new drug applications, rising regulatory approvals in the US and Europe, and expanding global clinical studies. Japan leads in rapid development and approvals.
Market Overview/ Introduction
The cell and gene therapies (CGT) market for rare disorders is experiencing rapid growth because of new genetic engineering developments and increasing rare disease cases and strong regulatory backing. The therapies treat genetic disorders by targeting their fundamental genetic origins which results in either permanent cures or extended treatment benefits.
Indian vaccine manufacturer Bharat Biotech announced on Thursday that it invested $75 million into its inaugural cell and gene therapy center located in Telangana, which will produce new therapeutic products within three years. In 2025, approximately 5,500 rare diseases are registered in the WHO’s International Classification of Diseases 11th Revision, and it is estimated that there are about 8,000 rare diseases, including cases with reported specific symptoms.
eyeDNA Therapeutics, a clinical-stage gene therapy company that develops transformative therapies for inherited retinal disorders, has received Rare Pediatric Disease Designation (RPDD) from the US Food and Drug Administration (FDA) for HORA-PDE6b, its innovative gene therapy that treats inherited retinal dystrophy (IRD) caused by PDE6b gene mutations.
- In Europe, between 6,000 and 8,000 distinct rare diseases affect millions, despite each condition being individually rare
- The U.S. Food and Drug Administration released draft guidelines in February 2026 to help sponsors who want to obtain approval of targeted individualized therapies through demonstration of treatment effectiveness and safety using evidence when randomized controlled trials cannot be conducted because of limited patient availability.
Notable Insights: -
- North America holds the largest regional market share, approximately 48.3% in the global Cell and Gene Therapies in Rare Disorders market.
- Asia Pacific is the fastest-growing region in the global cell and gene therapies in rare disorders market.
- By therapy type, the gene-modified cell therapy segment held a dominant position in terms of market share in 2025.
- By application, the rare genetic disorders segment is the dominating one, accounting for over 35% of the global market share in 2025.
- The compound annual growth rate of the global cell and gene therapies in rare disorders market is 26.34%.
- The market is likely to achieve a valuation of USD 42.8 billion by 2035.
What is role of technology in grooming the market?
Technology is playing a critical role in shaping the global cell and gene therapies in rare disorders market by enabling precision, scalability, and innovation. Advanced gene-editing tools such as CRISPR and next-generation sequencing (NGS) allow accurate identification and correction of genetic mutations, improving treatment effectiveness. Improvements in viral vector engineering, particularly AAV and lentiviral systems, have enhanced gene delivery efficiency and safety profiles. Automation and AI-based analytical systems complete drug discovery work and clinical trial design work and patient selection work to create shorter development periods. Biomanufacturing innovations through modular production systems and closed-system production methods enable therapy scaling while maintaining product quality. Digital health technologies, with real-world data, enable monitoring of patients after treatment as well as tracking of their outcomes. The current technological developments prepare CGTs for faster approval processes while decreasing expenses and widening patient access, which establishes CGTs as both a groundbreaking treatment method and an effective solution for rare disease conditions.
How is Recent Developments Helping the Market?
Recent developments, such as improving treatment outcomes, expanding therapeutic options, and increasing patient access, are supporting the growth of the global cell and gene therapies in rare disorders market. The use of non-viral vectors such as lipid nanoparticles and engineered AAVs results in lower immune rejection while enabling more accurate redosing procedures, which expand the number of patients who can receive treatment. The FDA’s START pilot program and the EMA’s PRIME scheme accelerate approvals for ultra-rare diseases, which provide developers with frequent communication and shorter review cycles. AI now serves two purposes because it identifies the best genetic targets and shows how patients will respond to treatment, which enables researchers to complete their work faster while reducing the high expenses of failed clinical trials. Leading companies create automated closed-loop systems that decrease human mistakes and eliminate production delays that previously restricted product availability in the market.
Market Drivers
The global cell and gene therapies in rare disorders market is propelled by the cell therapies, which treat rare disorders keeps expanding as more clinical studies are being conducted around the world. The fastest growth and regulatory approval process in the 7 million market exists within Japan. More than 55% funding increase for rare disease therapies occurred because venture capital and pharmaceutical companies such as Novartis and Pfizer made significant investments. The majority of rare diseases 80% have genetic origins, which currently lack effective long-term treatment options. CGTs offer a shift from symptom management to one-time curative solutions by targeting root genetic causes. The FDA and EMA agencies create fast-track approval systems through their Orphan Drug Designation Fast Track and Breakthrough Therapy designations, which provide market protection and lower costs.
Restrain
The main restraints on the global cell and gene therapies in rare disorders market include the One-time therapies often carry multimillion-dollar price tags, which include Hemgenix, priced at USD 3.5 million and Zolgensma. The elevated costs impose extreme financial obstacles that affect payers, healthcare systems and individual patients. The regulators need between 5 and 15 years of extended monitoring data to detect late-occurring negative effects, which include tumor development and immune system responses.
Competitive Analysis:
The report offers the appropriate analysis of the key organizations/companies involved within the cell and gene therapies in rare disorders market, along with a comparative evaluation primarily based on their product of offering, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborative analysis focusing on the current news and developments of the companies, which includes product development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
Top Companies in Global Cell and Gene Therapies in Rare Disorders Market
- Novartis AG
- Gilead Sciences, Inc.
- Bristol Myers Squibb
- F. Hoffmann-La Roche Ltd
- Vertex Pharmaceuticals Incorporated
- CRISPR Therapeutics AG
- Biogen Inc.
- Sarepta Therapeutics, Inc.
- BioMarin Pharmaceutical Inc.
- Pfizer Inc.
Government Initiatives
|
Country |
Key Government Initiatives |
|
India |
The Indian government issued draft amendments to include gene therapeutic and cell-derived products under formal drug licensing rules to better regulate and promote homegrown therapies. |
|
US |
The US FDA proposed a new "plausible-mechanism" pathway to speed up approvals for personalized treatments for rare, life-threatening genetic diseases. This allows drugmakers to rely on early efficacy signals and biological rationale rather than full-scale randomized trials when patient groups are too small. |
Market Segmentation
The cell and gene therapies in rare disorders market share is classified into therapy type, vector type, and application.
- The gene-modified cell therapy segment dominated the market in 2025, and is projected to grow at a substantial CAGR during the forecast period.
Based on the therapy type, the cell and gene therapies in the rare disorders market are divided into gene therapy, cell therapy, gene-modified cell therapy. Among these, the gene-modified cell therapy segment dominated the market in 2025 and is projected to grow at a substantial CAGR during the forecast period. It is because the company establishes market dominance through its extensive commercial adoption while maintaining a strong development pipeline of approved treatments for rare blood cancers. The system attracts maximum research and development funding because it combines exact gene therapy targeting with scalable therapeutic capabilities of cellular biology.
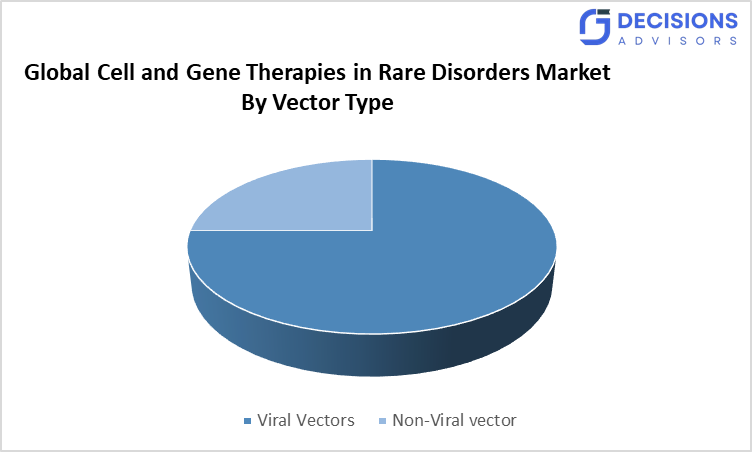
- The viral vectors segment accounted for the largest share in 2025, and is anticipated to grow at a significant CAGR during the forecast period.
Based on the vector type, the cell and gene therapies in rare disorders market are divided into viral vectors and non-viral vectors. Among these, the viral vectors segment accounted for the largest share in 2025, and is anticipated to grow at a significant CAGR during the forecast period. This dominance is It is mainly because AAVs exhibit high transduction efficiency and innate ability to integrate genetic material into host cells, resulting in durable, long-term gene expression for monogenic rare diseases.
- The rare genetic disorders segment dominated the market in 2025, and is projected to grow at a substantial CAGR during the forecast period.
Based on the application, the cell and gene therapies in rare disorders market are divided into rare genetic disorders, rare cancers, neurological disorders, metabolic disorders. Among these, the rare genetic disorders segment dominated the market in 2025, and is projected to grow at a substantial CAGR during the forecast period. Due to the fact that approximately 80% of rare diseases have a single-gene (monogenic) origin, they are ideal targets for curative gene replacement or editing therapies that address the underlying cause rather than just symptoms.
What is the Reason for the Region Dominance?
The dominance of certain regions in the global cell and gene therapies in rare disorders market is primarily driven by a robust regulatory and financial ecosystem. The U.S. FDA’s Orphan Drug Act and Breakthrough Therapy designations provide critical incentives, including tax credits, fee waivers, and seven years of market exclusivity, which significantly de-risk high-cost R&D. Furthermore, the region possesses the world’s most advanced healthcare infrastructure, featuring a dense network of Authorized Treatment Centers (ATCs) capable of handling complex cell processing. Massive capital infusion from venture capital and government grants (exceeding $10 billion annually) fuels a dominant clinical pipeline. Finally, the early adoption of innovative reimbursement models, such as value-based and annuity payments, ensures faster commercial patient access compared to other regions.
Strategies to Implement for Growth of the Market in Non-Leading Regions
Growth in non-leading regions can be supported through strategic investments, product innovation, and expanding distribution networks. The operation of Regional CDMO facilities enables companies to save money and decrease their operational risks, which would otherwise occur during extended cold-chain transportation of time-sensitive cellular products. The establishment of reliance pathways enables local agencies to recognize FDA and EMA approvals, which permits them to eliminate the need for extra clinical testing. The combination of "Orphan Drug" legislation with this system creates financial benefits through tax deductions and exclusive market rights, which international developers find appealing. Local Phase II/III trial partnerships with international companies develop medical facilities while educating expert teams who will manage advanced therapy procedures. Resource-limited healthcare systems can finance curative treatments through annuity-based payments and outcome-based rebates, which distribute treatment expenses throughout multiple years according to patient recovery progress.
Regional Segment Analysis of the Cell and Gene Therapies in Rare Disorders Market
- North America (U.S., Canada, Mexico)
- Europe (Germany, France, U.K., Italy, Spain, Rest of Europe)
- Asia-Pacific (China, Japan, India, Rest of APAC)
- South America (Brazil and the Rest of South America)
- The Middle East and Africa (UAE, South Africa, Rest of MEA)
North America is anticipated to hold the largest share of the Cell and Gene Therapies in Rare Disorders market over the predicted timeframe.
North America is anticipated to hold the largest share of the Cell and Gene Therapies in Rare Disorders market over the predicted timeframe. This market is accounting about approximately 48.3% of the total market share. The dominance of this market is driven by the U.S. FDA’s progressive policies, including the Orphan Drug Act and Breakthrough Therapy designations, which provide developers with tax credits, fee waivers, and extended market exclusivity. The region hosts the world's highest density of authorized treatment centres (ATCs) and specialised hospitals equipped for complex therapy administration. Mature insurance and reimbursement systems, including outcomes-based contracts, accelerate the commercial adoption of high-cost curative treatments.
Asia Pacific is expected to grow at a rapid CAGR in the Cell and Gene Therapies in Rare Disorders market during the forecast period. Asia Pacific is poised to be the fastest-growing region in the global Cell and Gene Therapies in Rare Disorders market with a remarkable CAGR of 19.2% during the forecast period of 2025 to 2035. Countries like Japan and China have established expedited approval frameworks for regenerative medicines. For instance, China's National Medical Products Administration (NMPA) has shortened certain approval timelines to just 60 working days. APAC offers a massive and diverse patient population, which is critical for rare disease research. Median recruitment rates in the region are up to four times faster than in the U.S. and Europe, significantly reducing clinical trial timelines.
Europe is the 2nd largest region to grow in the Cell and Gene Therapies in Rare Disorders market during the period. Europe is the second-largest region in the market, holding approximately 25–28% of the global share. The EMA’s PRIME (Priority Medicines) scheme and Orphan Medicinal Product designations provide accelerated pathways and 10-year market exclusivity. Massive R&D hubs in Germany, France, and the UK (such as the Cell and Gene Therapy Catapult) drive continuous innovation.
Future Market Trends in Global Cell and Gene Therapies in Rare Disorders Market: -
- Shift Toward Curative, One-Time Therapies
Gene therapies, particularly those using viral vectors like AAV, have demonstrated long-term efficacy with a single administration. Over 20+ gene therapies have been approved globally, with many targeting rare diseases. Clinical data show sustained benefits lasting 5–10 years or more in some cases. Additionally, more than 1,000 active clinical trials are ongoing, reflecting strong pipeline growth and increasing investor confidence in durable, one-time therapeutic solutions.
- Rising Number of Gene Therapy Approvals
The number of gene therapy approvals is steadily increasing, reflecting strong regulatory support and clinical success. Globally, around 36 gene therapies have been approved as of 2025, with over 46 cell and gene therapies approved in the U.S. alone. Additionally, regulatory agencies approved 8 new CGT products in 2024, highlighting accelerating innovation and faster approval pathways.
- Advancements in Gene Editing Technologies
Advancements in gene editing technologies, particularly CRISPR-Cas systems, are significantly transforming rare disease treatment. The global CRISPR market was valued at ~USD 4.0 billion in 2024 and is projected to exceed USD 13.5 billion by 2033. Over 300 gene-editing clinical trials are ongoing globally, with CRISPR accounting for over 50% of approaches. These innovations enable precise, efficient, and potentially curative therapies.
Recent Development
In February 2024, REGENXBIO Inc. announced topline results from the Phase I/II/III CAMPSIITE® trial of RGX-121 for the treatment of patients up to 5 years old diagnosed with Mucopolysaccharidosis Type II (MPS II), also known as Hunter syndrome, demonstrating that the pivotal phase of the trial met its primary endpoint with statistical significance.
Market Segment
This study forecasts revenue at global, regional, and country levels from 2020 to 2035. Decision Advisors has segmented the cell and gene therapies in rare disorders market based on the below-mentioned segments:
Global Cell and Gene Therapies in Rare Disorders Market, By Treatment Type
- Gene Therapy
- Cell Therapy
- Gene-Modified Cell Therapy
Global Cell and Gene Therapies in Rare Disorders Market, By Route of Administration
- Viral Vectors
- Non-Viral Vectors
Global Cell and Gene Therapies in Rare Disorders Market, By Distribution Channel
- Rare Genetic Disorders
- Rare Cancers
- Neurological Disorders
- Metabolic Disorders
Global Cell and Gene Therapies in Rare Disorders Market, By Regional Analysis
- North America
- US
- Canada
- Mexico
- Europe
- Germany
- UK
- France
- Italy
- Spain
- Russia
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- Australia
- Rest of Asia Pacific
- South America
- Brazil
- Argentina
- Rest of South America
- Middle East & Africa
- UAE
- Saudi Arabia
- Qatar
- South Africa
- Rest of the Middle East & Africa
Frequently Asked Questions (FAQ)
Q: Which technologies are commonly used?
A: Common technologies include CRISPR gene editing, viral vectors like AAV and lentivirus for gene delivery, and stem cell-based therapies. Next-generation sequencing is used for diagnosis and patient selection. These technologies enhance precision, safety, and effectiveness, enabling targeted treatment approaches for various rare genetic disorders.
Q: What is the future outlook of the market?
A: The market is expected to grow significantly due to continuous innovation, increasing approvals, and expanding clinical pipelines. Advancements in personalized medicine and gene-editing technologies will improve outcomes. As manufacturing scales and costs decrease, accessibility will improve, making these therapies more widely available globally.
Q: How are manufacturing challenges being addressed?
A: Manufacturing challenges are being tackled through automation, closed-system production, and improved vector design. Companies are investing in scalable facilities to meet growing demand. Technological advancements are helping reduce production time and costs, while ensuring consistency, quality, and compliance with strict regulatory standards
Q: What is the impact of high treatment costs?
A: High costs, often exceeding USD 1–3 million per treatment, limit patient access and strain healthcare systems. To address this, companies and payers are adopting innovative pricing models such as outcome-based payments and instalment plans, improving affordability and enabling broader adoption of these advanced therapies
Q: How do regulatory policies impact the market?
A: Regulatory policies play a crucial role by providing fast-track approvals, orphan drug designations, and financial incentives. These measures encourage innovation and reduce development timelines. However, strict safety and efficacy requirements can also pose challenges, impacting the speed at which new therapies reach the market.
Check Licence
Choose the plan that fits you best: Single User, Multi-User, or Enterprise solutions tailored for your needs.
We Have You Covered
- 24/7 Analyst Support
- Clients Across the Globe
- Tailored Insights
- Technology Tracking
- Competitive Intelligence
- Custom Research
- Syndicated Market Studies
- Market Overview
- Market Segmentation
- Growth Drivers
- Market Opportunities
- Regulatory Insights
- Innovation & Sustainability
Report Details
| Scope | Global |
| Pages | 210 |
| Delivery | PDF & Excel via Email |
| Language | English |
| Release | Mar 2026 |
| Access | Download from this page |
