Global Connected Drug Delivery Devices Market
Global Connected Drug Delivery Devices Market Size, Share, and COVID-19 Impact Analysis, By Type (Injectable Devices and Inhalation Devices & Others), By Technology (Bluetooth, NFC, and Others), By End User (Homecare Settings and Hospitals), and By Region (North America, Europe, Asia-Pacific, Latin America, Middle East, and Africa), Analysis and Forecast 2025-2035
Report Overview
Table of Contents
Global Connected Drug Delivery Devices Market Size Insights Forecasts to 2035
- The Global Connected Drug Delivery Devices Market Size Was Estimated at USD 7.34 Billion in 2024
- The Market Size is Expected to Grow at a CAGR of around 23.15 % from 2025 to 2035
- The Worldwide Connected Drug Delivery Devices Market Size is Expected to Reach USD 72.54 Billion by 2035
- North America is expected to grow the fastest during the forecast period.
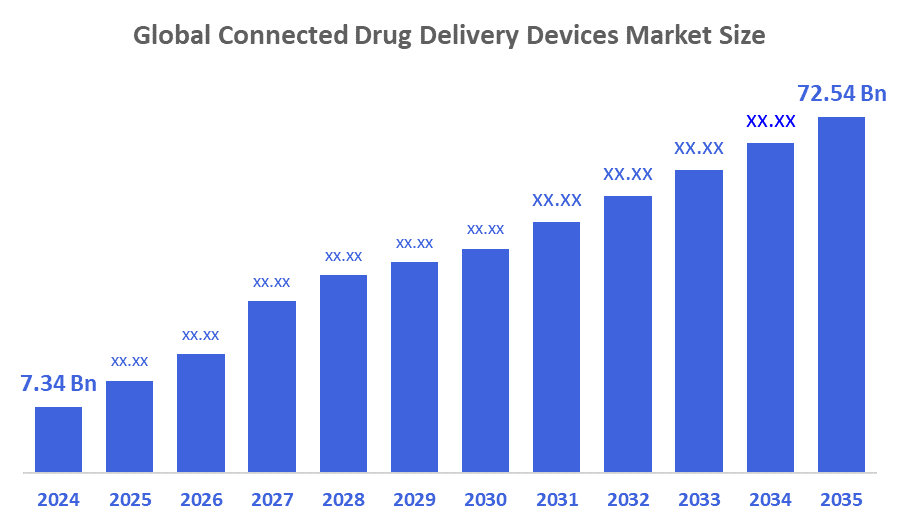
According to a research report published by Decisions Advisors and Consulting, the Global Connected Drug Delivery Devices Market Size was worth around USD 7.34 billion in 2024 and is predicted to grow to around USD 72.54 billion by 2035 with a compound annual growth rate (CAGR) of 23.15 % from 2025 to 2035. Its market dominance for associated medication delivery devices is driven by improvements in healthcare technology, widespread use of connected devices, rising rates of chronic illness, and favourable regulatory environments. The demand to boost patient medication adherence, R&D efforts, and the acceleration of remote digital monitoring are some of the reasons driving the market.
Market Overview
The medical devices with digital connectivity and communication capabilities that allow for tracking, monitoring, and managing medication administration are referred to as connected drug delivery devices. Connectivity to a variety of digital services is one of the fundamental features of connected medication delivery devices. In addition, the technology that improves patient adherence, improves treatment outcomes, and gives medical personnel precise data for individualised care is provided by these medical devices. Many market players are working on these devices in a bid to provide better monitoring of patients and counselling. Many functions are included in connected drug delivery systems, including tools for patient education, dosage reminders, adherence trackers, and patient diaries to gather health data. Asthma, diabetes, chronic obstructive pulmonary disease (COPD), and other cardiovascular conditions are frequently treated with these devices.
AbbVie entered into a definitive agreement to acquire West Pharmaceutical Services’ drug delivery device manufacturing facility in Tempe, Arizona, along with associated intellectual property. This move is part of AbbVie’s broader $10 billion U.S. manufacturing investment plan over the next decade. The facility includes multiple production lines and advanced on-body injector technology. The transaction is expected to close in mid-2026, strengthening AbbVie’s U.S.-based manufacturing footprint and innovation capabilities.
Enable Injections secured a $30 million investment from Sanofi to accelerate the expansion of its enFuse On-Body Delivery System (OBDS). The funding will support manufacturing build-out, strengthen supply chain capabilities, and drive commercial growth. The enFuse system enables large-volume subcutaneous drug delivery at home, reducing hospital visits and improving patient convenience, particularly for biologics and oncology treatments.
Report Coverage
This research report categorizes the connected drug delivery devices market based on various segments and regions, forecasts revenue growth, and analyzes trends in each submarket. The report analyses the key growth drivers, opportunities, and challenges influencing the connected drug delivery devices market. Recent market developments and competitive strategies, such as expansion, product launch, development, partnership, merger, and acquisition, have been included to draw the competitive landscape in the market. The report strategically identifies and profiles the key market players and analyzes their core competencies in each sub-segment of the connected drug delivery devices market.
Driving Factors
In the healthcare industry, economic revenue associated with pharmaceutical delivery devices, the worldwide epidemic accelerated the widespread use of remote medical services. Devices that allowed for medication self-management and self-monitoring at home were in high demand. The industry expansion is stimulated through innovation in digital health technologies, and the expansion of connected medicine delivery devices was facilitated by changes in telehealth reimbursement policies. The rise in chronic disease, the development of digital health technology, and the demand for individualised medication are all factors contributing to the expansion of globally networked drug delivery devices. Furthermore, real-time monitoring, dose tracking, reminders, and data sharing with healthcare practitioners are options provided by linked drug delivery systems, which enhance patient engagement and medication adherence.
OncoNano Medicine announced a research collaboration with Gilead Sciences to apply its proprietary ON-BOARD drug delivery technology to one of Gilead’s drug candidates. The partnership aims to enhance precision drug delivery, improve therapeutic outcomes, and accelerate innovation in oncology and other therapeutic areas. By leveraging OncoNano’s advanced delivery platform, the collaboration seeks to optimise drug performance and patient benefit.
Restraining Factors
The implementation of networked medicine delivery devices is hampered by high costs and strict regulatory compliance. Expensive technology restricts access, and long approval procedures impede new ideas. Moreover, such obstacles limit market expansion with postpone patient benefits and necessitate cost-cutting measures and favourable legislative changes.
Market Segmentation
The connected drug delivery devices market share is classified into type, technology, and end user.
- The injectable devices segment accounted for the highest share in 2024 and is anticipated to grow at a remarkable CAGR over the forecast period.
Based on the type, the connected drug delivery devices market is segmented into injectable devices, inhalation devices, and others. Among these, the injectable devices segment accounted for the highest share in 2024 and is anticipated to grow at a remarkable CAGR over the forecast period. This is because these devices may combine sophisticated monitoring capabilities with medicine delivery, greatly improving patient care. By measuring medicine usage in real time, these devices assist patients in following their recommended regimens and enhance treatment results overall.
For instance, QuiO, a digital health startup, raised $1 million in seed funding to advance development of its Smartinjector™ connected injector technology. The device is designed to improve medication adherence for chronic disease patients by using IoT-enabled monitoring of self-injections.
- The Bluetooth segment accounted for the highest revenue share in 2024 and is expected to grow at a remarkable CAGR over the forecast period.
Based on the technology, the connected drug delivery devices market is differentiated into Bluetooth, NFC, and others. Among these, the Bluetooth segment accounted for the highest revenue share in 2024 and is expected to grow at a remarkable CAGR over the forecast period. Bluetooth is the industry leader because of its many benefits for medical applications. It's low power consumption, which enables devices like wearable injectors, insulin pens, and inhalers to function well over extended periods of time without requiring frequent battery replacements. This improves patient compliance and lowers operating expenses. Real-time insights into medicine usage, dosage, and adherence are made possible by Bluetooth's wireless communication capabilities, which facilitate smooth data transmission between drug delivery devices and mobile applications or healthcare systems.
For instance, MOBI launched its Smart WiFi Pill Dispenser, designed to simplify medication management and support remote caregiving. The device features dual-band WiFi connectivity, caregiver tracking, and integration with other MOBI smart health devices, all accessible through a single app.
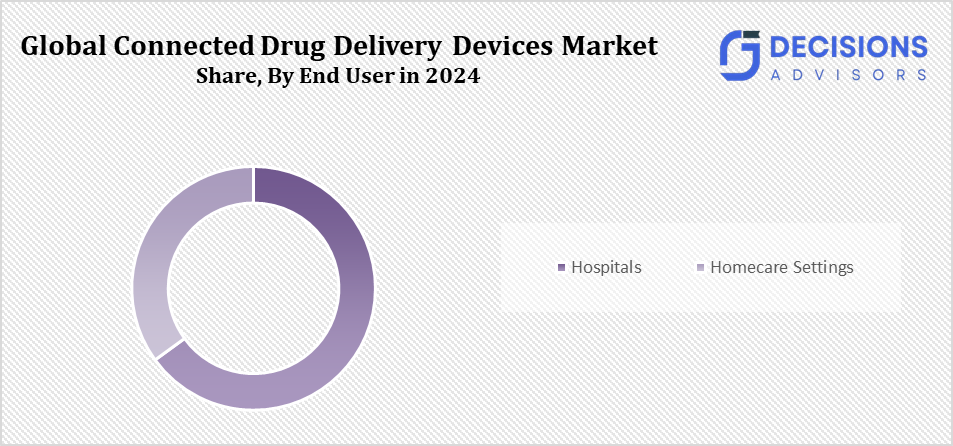
- The hospitals segment accounted for the largest market share in 2024 and is projected to grow at a notable CAGR over the forecast period.
Based on the end user, the connected drug delivery devices market is divided into homecare settings and hospitals. Among these, the hospitals segment accounted for the largest market share in 2024 and is projected to grow at a notable CAGR over the forecast period. This is due to their pressing need for effective patient care, greater drug adherence, and better treatment outcomes; hospitals and healthcare providers dominate the industry. Further, its individualised therapies track patient progress and expedite medication management; these facilities rely on connected devices.
Regional Segment Analysis of the Connected Drug Delivery Devices Market
- North America (U.S., Canada, Mexico)
- Europe (Germany, France, U.K., Italy, Spain, Rest of Europe)
- Asia-Pacific (China, Japan, India, Rest of APAC)
- South America (Brazil and the Rest of South America)
- The Middle East and Africa (UAE, South Africa, Rest of MEA)
Asia Pacific is anticipated to hold the largest share of the connected drug delivery devices market over the predicted timeframe.
Asia Pacific is anticipated to hold the largest share of the connected drug delivery devices market over the predicted timeframe. The Asia Pacific market industry is driven by the growing prevalence of chronic diseases and the availability of a large patient population base. It is also anticipated that the growing number of businesses and projects they have implemented would support regional expansion. The market in the Asia Pacific is expanding rapidly due to factors such as China, India, and other countries' quick embrace of medical technology, cost advantages, and growing awareness of connected drug delivery devices. Some of the major drivers boosting the market revenue are the adoption of aggressive marketing techniques by industry participants, rising healthcare spending, and the anticipated growth of the distribution network of major companies.
For example, Adherium is a public firm situated in New Zealand that specialises in creating solutions for patient adherence, remote monitoring, and data management in the respiratory field.
North America is expected to grow at a rapid CAGR in the connected drug delivery devices market during the forecast period. The increased expenditure on healthcare suggests a greater emphasis on improving patient outcomes and reducing overall healthcare costs through the use of cutting-edge technologies. In addition, the area is home to a sizable number of pharmaceutical firms and producers of medical equipment that make significant investments in connected drug delivery device research and development (R&D). These businesses are using the Internet of Things and digital connectivity to create cutting-edge gadgets that improve patient monitoring, medication compliance, and treatment results in general. The rapid growth of this market segment is further facilitated by favourable reimbursement policies and supportive regulatory frameworks in the region.
The FDA recently raised expectations for drug delivery device evidence, issuing draft guidance that emphasises detailed performance data for devices and combination products. The June 2024 draft guidance highlights the need for robust clinical and technical validation of essential drug delivery outputs, including accuracy, reliability, and safety of delivery mechanisms. In parallel, the FDA’s 2026 guidance agenda signals a stronger focus on real-world evidence, patient preference data, and quality management systems, meaning companies will need to demonstrate not only technical performance but also patient-centric outcomes and compliance with evolving standards.
Competitive Analysis:
The report offers the appropriate analysis of the key organizations/companies involved within the connected drug delivery devices market, along with a comparative evaluation primarily based on their product offering, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborative analysis focusing on the current news and developments of the companies, which includes product development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
List of Key Companies
- Adherium
- Aptar Pharma
- AstraZeneca
- Becton, Dickinson, and Company
- Bigfoot Biomedical
- Cohero Health
- Eli Lilly and Company
- Insulet Corporation
- Novartis
- Novo Nordisk
- Phillips Medisize
- Propeller Health
- Teva Pharmaceuticals
- West Pharmaceutical Services
- Others
Key Target Audience
- Market Players
- Investors
- End-users
- Government Authorities
- Consulting and Research Firm
- Venture capitalists
- Value-Added Resellers (VARs)
Recent Development
- In February 2026, Medtronic officially launched the MiniMed Go Smart MDI system with the Simplera sensor across Europe. This marks the first integrated solution that combines data from the InPen smart insulin pen and the Simplera™ continuous glucose sensor into a single mobile app. The system provides real-time, actionable insights for people managing diabetes with multiple daily injections (MDI), helping improve glucose control and treatment decisions.
- In October 2025, BD (Becton, Dickinson and Company) launched new AI-enabled solutions to strengthen connectivity across healthcare settings. At the core is the BD Incada Connected Care Platform, a scalable, cloud-based ecosystem that unifies data from BD devices, including infusion pumps, patient monitors, and pharmacy robotics, into one intelligent network. The platform debuted alongside the next-generation BD Pyxis Pro Automated Medication Dispensing Solution, enabling enterprise-wide visibility and actionable insights.
Market Segment
This study forecasts revenue at global, regional, and country levels from 2020 to 2035. Decisions Advisors has segmented the connected drug delivery devices market based on the below-mentioned segments:
Global Connected Drug Delivery Devices Market, By Type
- Injectable Devices
- Inhalation Devices
- Others
Global Connected Drug Delivery Devices Market, By Technology
- Bluetooth
- NFC
- Others
Global Connected Drug Delivery Devices Market, By End User
- Homecare Settings
- Hospitals
Global Connected Drug Delivery Devices Market, By Regional Analysis
- North America
- US
- Canada
- Mexico
- Europe
- Germany
- UK
- France
- Italy
- Spain
- Russia
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- Australia
- Rest of Asia Pacific
- South America
- Brazil
- Argentina
- Rest of South America
- Middle East & Africa
- UAE
- Saudi Arabia
- Qatar
- South Africa
- Rest of the Middle East & Africa
Frequently Asked Questions (FAQ)
1. How is artificial intelligence (AI) transforming connected drug delivery devices?
AI integration enables predictive analytics, automated dose adjustments, and personalised therapy recommendations. Advanced algorithms analyse patient adherence patterns and physiological data to optimise treatment regimens. Companies like Medtronic are increasingly embedding AI-driven insights into diabetes management ecosystems to improve clinical decision-making.
2. What cybersecurity challenges are associated with connected drug delivery devices?
As these devices transmit sensitive health data via cloud platforms and mobile applications, cybersecurity risks such as data breaches and unauthorised access are significant concerns. Regulatory authorities, including the U.S. Food and Drug Administration, are strengthening cybersecurity compliance requirements for connected medical devices to ensure patient data protection and system integrity.
3. How are interoperability standards influencing market adoption?
Interoperability allows connected drug delivery devices to integrate seamlessly with electronic health records (EHRs), hospital information systems, and remote monitoring platforms. Adoption of standardised communication protocols enhances scalability, supports coordinated care, and improves clinical workflow efficiency, making devices more attractive to healthcare providers.
4. What is the impact of value-based healthcare models on this market?
Value-based healthcare frameworks emphasise measurable patient outcomes and cost efficiency. Connected drug delivery devices provide real-time adherence and outcome data, enabling payers and providers to track performance metrics. This aligns well with reimbursement models that reward improved therapeutic effectiveness and reduced hospital readmissions.
5. How are wearable technologies shaping future product development?
The convergence of wearable biosensors with connected injectors and inhalers is driving innovation. Continuous monitoring devices paired with smart delivery systems create closed-loop therapeutic ecosystems, particularly in chronic disease management such as diabetes and respiratory disorders.
6. What role do pharmaceutical–technology partnerships play in market expansion?
Strategic collaborations between pharma companies and digital health firms accelerate product innovation, improve device–drug compatibility, and expand commercialisation capabilities. Partnerships similar to those involving Sanofi in connected delivery investments illustrate how co-development models enhance ecosystem integration and speed market entry.
7. How is patient engagement influencing purchasing decisions in this market?
Modern patients increasingly prefer user-friendly, app-integrated medical devices that provide reminders, progress tracking, and real-time feedback. Devices offering intuitive interfaces and caregiver connectivity features are gaining preference, particularly in home care environments, thereby influencing product differentiation strategies among manufacturers.
Check Licence
Choose the plan that fits you best: Single User, Multi-User, or Enterprise solutions tailored for your needs.
We Have You Covered
- 24/7 Analyst Support
- Clients Across the Globe
- Tailored Insights
- Technology Tracking
- Competitive Intelligence
- Custom Research
- Syndicated Market Studies
- Market Overview
- Market Segmentation
- Growth Drivers
- Market Opportunities
- Regulatory Insights
- Innovation & Sustainability
Report Details
| Scope | Global |
| Pages | 250 |
| Delivery | PDF & Excel via Email |
| Language | English |
| Release | Mar 2026 |
| Access | Download from this page |
