Global Cytotoxic Drugs Contract Manufacturing Market
Global Cytotoxic Drugs Contract Manufacturing Market Size, Share, By Product Type (Active Pharmaceutical Ingredients (APIs), Finished Dosage Forms (FDFs)), By Route of Administration (Injectable, Oral, Topical), By End User (Pharmaceutical Companies, Biotechnology Companies, Research Organizations), and By Region (North America, Europe, Asia-Pacific, Latin America, Middle East & Africa), Analysis and Forecast 2026-2035
Report Overview
Table of Contents
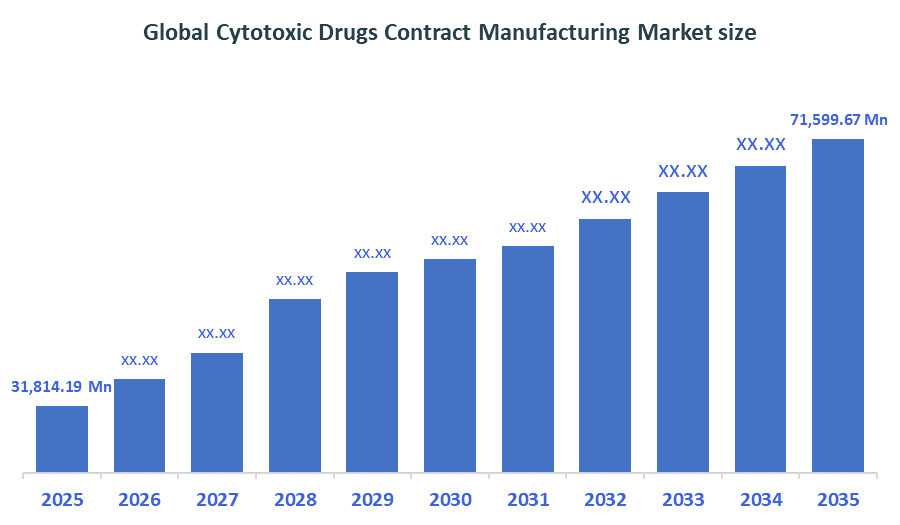
The Global Cytotoxic Drugs Contract Manufacturing Market size is forecast to grow from USD 31,814.19 Million in 2025 to approximately USD 71,599.67 Million by 2035. According to Decision Advisors, a detailed research report on the cytotoxic drugs contract manufacturing market indicates that the industrial-scale manufacturing dominates the market, accounting for approximately 65% of the total share globally. Lonza Group leads the market with an estimated 10-15% global market share. The company reported a 2025 revenue of approximately USD 8.5 billion, making it one of the most influential forces shaping industry trends and overall market growth.
Market Snapshot
- Global Cytotoxic Drugs Contract Manufacturing Market Size (2025): USD 31,814.19 Million
- Projected Global Cytotoxic Drugs Contract Manufacturing Market Size (2035): USD 71,599.67 Million
- Global Cytotoxic Drugs Contract Manufacturing Market Compound Annual Growth Rate (CAGR): 8.45%
- Largest Regional Market: North America
- Fastest Growing Region: Asia-Pacific
- Market 3rd Largest Region: Europe
- Base Year: 2025
- Historical Period: 2021–2024
- Forecast Period: 2026–2035
Market Overview/ Introduction
The term Global Cytotoxic Drugs Contract Manufacturing Market encompasses the process where the production services for cytotoxic drugs very strong pharmaceutical compounds mainly used in the treatment of cancer diseases, are outsourced to specialized manufacturing companies. Cytotoxic drugs contract manufacturing entails the involvement of third parties in the process of developing, formulating, and mass-producing such dangerous pharmaceuticals. Factors propelling growth within the market include increased prevalence of cancer across the globe, demand for effective manufacturing processes, and compliance issues. Emerging trends in the market include advancements in containment technology, penetration of new markets, and increasing investments in the oncology pipeline.
- In January 2026, India introduced amendments to the New Drugs and Clinical Trials Rules to accelerate drug approvals, reduce timelines, and adopt risk-based regulation. While easing processes for low-risk drugs, strict oversight remains for high-risk categories like cytotoxic drugs, strengthening manufacturing compliance and global outsourcing attractiveness.
- The U.S. government strengthened its Strategic Active Pharmaceutical Ingredients Reserve through a 2025 executive order to secure domestic drug supply chains. This initiative supports local manufacturing of high-potency APIs, including cytotoxic compounds, reducing reliance on imports and encouraging contract manufacturing expansion.
Notable Insights: -
- North America is anticipated to hold the largest share of approximately 40% in the cytotoxic drugs contract manufacturing market over the forecast period.
- Asia Pacific is expected to grow at a rapid CAGR of approximately 9.5% in the cytotoxic drugs contract manufacturing market during the forecast period.
- The active pharmaceutical ingredients (APIs) segment dominated the market in 2025, approximately 60%, and is projected to grow at a substantial CAGR during the forecast period.
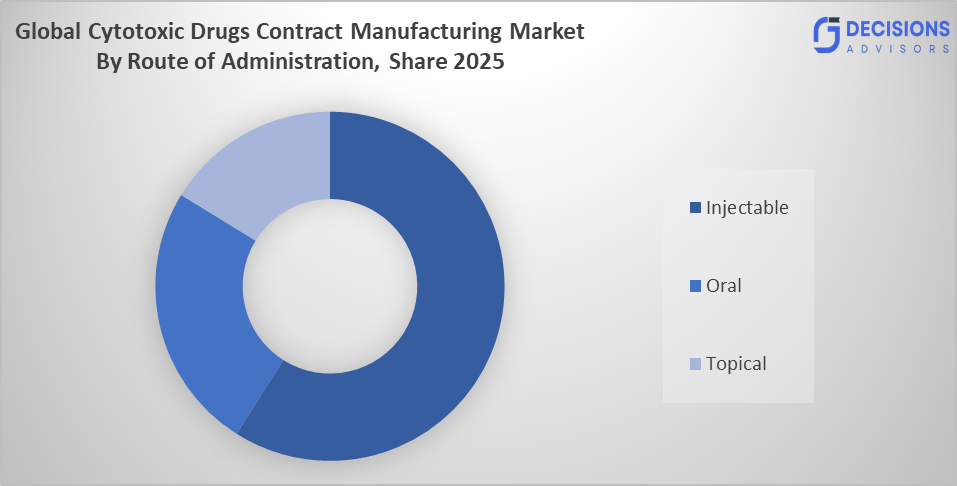
- The injectable segment dominated the market in 2025, approximately 70%, and is projected to grow at a substantial CAGR during the forecast period.
- The pharmaceutical companies segment dominated the market in 2025, approximately 65%, and is projected to grow at a substantial CAGR during the forecast period.
- The compound annual growth rate of the Global Cytotoxic Drugs Contract Manufacturing Market is 8.45%.
- The market is likely to achieve a valuation of USD 71,599.67 Million by 2035.
What is role of technology in grooming the market?
Technology plays a critical role in shaping the Global Cytotoxic Drugs Contract Manufacturing Market by improving safety, efficiency, and product quality. Advanced high-containment systems such as isolators and closed processing equipment enable safe handling of highly potent cytotoxic compounds while meeting strict regulatory standards. Automation and digital manufacturing solutions enhance precision, reduce human error, and optimize production timelines. Innovations in formulation and drug delivery, particularly for sterile injectables and targeted therapies, are expanding manufacturing capabilities. Additionally, the adoption of data analytics and real-time monitoring systems supports better quality control and compliance with global GMP regulations. Continuous process optimization and scalable manufacturing technologies further reduce costs and increase output. Overall, technological advancements are enabling contract manufacturers to meet growing oncology demand, accelerate time-to-market, and maintain high safety and quality standards.
Market Drivers
The Global Cytotoxic Drugs Contract Manufacturing Market is driven by the growing incidence of cancers that call for improved cancer treatment. Increasingly, pharmaceutical and biotechnology organizations outsource their manufacturing processes to CDMOs in order to cut down on costs and dedicate themselves to research. In the pharmaceutical industry, there is an increase in demand for modern manufacturing equipment owing to the escalating need for potent APIs and personalized therapies such as ADCs. Partnerships between organizations and expert manufacturers are established due to regulatory demands that assist them in fulfilling compliance and product quality standards. There are three primary contributors to the market’s global growth.
Restrain
The Global Cytotoxic Drugs Contract Manufacturing Market faces restraints due to high capital investment requirements for containment facilities and strict regulatory compliance. Complex handling procedures and safety risks limit operational flexibility. Additionally, a shortage of skilled professionals and stringent approval processes increases costs and timelines, restricting market expansion, especially in emerging regions.
Competitive Analysis:
The report offers the appropriate analysis of the key organizations/companies involved within the global cytotoxic drugs contract manufacturing market, along with a comparative evaluation primarily based on their product offerings, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborative analysis focusing on the current news and developments of the companies, which includes product development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
Top Companies in Global Cytotoxic Drugs Contract Manufacturing Market
- Lonza Group
- Piramal Pharma Solutions
- Merck KGaA (via SAFC Pharma)
- Baxter BioPharma Solutions
- BSP Pharmaceuticals S.p.A.
- Catalent Inc.
- Thermo Fisher Scientific
- WuXi AppTec
- Siegfried Holding AG
- Recipharm AB
Government Initiatives
|
Country |
Key Government Initiatives |
|
USA |
The U.S. Food and Drug Administration (FDA) October 2025 launched a pilot program to fast-track approvals of domestically manufactured generic drugs and APIs, aiming to strengthen local pharmaceutical production and reduce foreign dependence. In parallel, regulatory efforts in 2025 focused on accelerating biosimilar approvals to lower costs and expand access to oncology and biologic treatments. These initiatives support increased domestic manufacturing capacity for cytotoxic and high-potency drugs. |
|
India |
The Government of India in February 2026 (Union Budget 2026) announced the ?10,000 crore Biopharma Shakti initiative to boost biopharmaceutical manufacturing and innovation. Alongside this, continued implementation of PLI schemes and bulk drug parks (2025–2026) has strengthened domestic API and high-potency drug manufacturing while reducing import dependence. These policies directly enhance contract manufacturing capabilities for cytotoxic drugs and ADCs. |
|
China |
China revised its drug regulatory framework in December 2025, accelerating approval pathways for innovative drugs and encouraging advanced biologics manufacturing. The government has also supported localized production and innovation in complex therapies, including antibody-drug conjugates (ADCs) and high-potency compounds, to modernize pharmaceutical manufacturing infrastructure and attract outsourcing partnerships |
Study on the Supply, Demand, Distribution, and Market Environment of the Cytotoxic Drugs Contract Manufacturing Market
The Global Cytotoxic Drugs Contract Manufacturing Market is shaped by a tightly controlled balance of supply, demand, distribution, and regulatory environment. Supply is dominated by specialized CDMOs with high-containment facilities and technical expertise, while high capital requirements and strict safety standards limit new entrants. Demand is rising due to the increasing global cancer burden and expanding oncology drug pipelines, driving pharmaceutical companies to outsource manufacturing. Distribution operates indirectly, with CDMOs supplying drug manufacturers, and final products reaching patients through hospitals and specialized healthcare centers, supported by robust cold-chain logistics. The market environment is highly regulated, requiring compliance with stringent GMP and safety norms. Additionally, growing technological advancements, strategic partnerships, and increased investments in emerging regions are enhancing manufacturing capabilities, improving accessibility, and supporting the overall expansion of the global cytotoxic drugs contract manufacturing market.
Price Analysis and Consumer Behaviour Analysis
Pricing in the global cytotoxic drugs contract manufacturing market is influenced by the high cost of containment infrastructure, regulatory compliance, and specialized workforce requirements. Manufacturing cytotoxic drugs involves stringent safety protocols, which significantly increase operational expenses compared to conventional drugs. Prices also vary based on product type, with sterile injectables and high-potency APIs commanding premium rates due to complex production processes. Additionally, long-term contracts and bulk manufacturing agreements often provide cost advantages to large pharmaceutical companies, while smaller firms face relatively higher per-unit costs. Regional pricing differences are evident, as emerging markets offer cost-competitive manufacturing due to lower labor and facility costs, making them attractive outsourcing destinations. In this market, the primary “consumers” are pharmaceutical and biotechnology companies rather than end patients. These clients prioritize quality, regulatory compliance, and reliability over cost alone when selecting contract manufacturers. There is a growing preference for CDMOs offering end-to-end services, including development, manufacturing, and packaging, to streamline operations. Demand is also shaped by the rising focus on oncology therapies, pushing companies to seek partners with expertise in handling high-potency compounds.
Market Segmentation
The Cytotoxic Drugs Contract Manufacturing Market share is classified into product type, route of administration, and end user.
- The active pharmaceutical ingredients (APIs) segment dominated the market in 2025, approximately 60%, and is projected to grow at a substantial CAGR during the forecast period.
Based on the product type, the cytotoxic drugs contract manufacturing market is divided into active pharmaceutical ingredients (APIs) and finished dosage forms (FDFs). Among these, the active pharmaceutical ingredients (APIs) segment dominated the market in 2025, approximately 60%, and is projected to grow at a substantial CAGR during the forecast period. The growth of the active pharmaceutical ingredients (APIs) segment is growing due to the complexity and high containment requirements of cytotoxic and HPAPI manufacturing, which pharmaceutical companies prefer to outsource to specialized CDMOs. APIs form the core of oncology drugs, and rising demand for targeted therapies further strengthens this segment’s leadership globally.
- The injectable segment dominated the market in 2025, approximately 70%, and is projected to grow at a substantial CAGR during the forecast period.
Based on the route of administration, the cytotoxic drugs contract manufacturing market is divided into injectable, oral, and topical. Among thes the injectable segment dominated the market in 2025, approximately 70%, and is projected to grow at a substantial CAGR during the forecast period. The injectable segment dominated due to cytotoxic drugs, which are primarily administered intravenously to ensure rapid and controlled delivery in cancer treatment. The need for sterile manufacturing environments, precise dosing, and hospital-based administration makes injectable the dominant choice, increasing outsourcing to contract manufacturers with advanced aseptic and containment capabilities.
- The pharmaceutical companies segment dominated the market in 2025, approximately 65%, and is projected to grow at a substantial CAGR during the forecast period.
Based on the end user, the cytotoxic drugs contract manufacturing market is divided into pharmaceutical companies, biotechnology companies, and research organizations. Among these, the pharmaceutical companies segment dominated the market in 2025, approximately 65%, and is projected to grow at a substantial CAGR during the forecast period. The growth of the pharmaceutical companies segment is due to the large pharma firms increasingly outsourcing cytotoxic drug manufacturing to reduce costs and focus on core R&D activities. The growing oncology pipeline, regulatory complexities, and need for specialized HPAPI facilities drive reliance on CDMOs, making pharmaceutical companies the primary contributors to market demand.
Strategies to Implement for Growth of the Market in Non-Leading Regions
Expanding the Global Cytotoxic Drugs Contract Manufacturing Market in non-leading regions requires a mix of capacity building, partnerships, and regulatory alignment. Companies should invest in cost-efficient, high-containment manufacturing facilities to meet safety standards while leveraging lower operational costs in emerging markets. Forming strategic collaborations with local pharmaceutical firms and governments can ease market entry and improve supply chain networks. Strengthening workforce capabilities through technical training in handling highly potent compounds is also essential. Additionally, aligning with international regulatory frameworks such as GMP and adopting advanced technologies like isolator-based systems can boost credibility and attract global clients. Incentives such as tax benefits and public-private funding can further encourage investments. These strategies collectively enhance regional competitiveness, expand manufacturing footprints, and improve access to oncology therapeutics in underserved markets.
Regional Segment Analysis of the Cytotoxic Drugs Contract Manufacturing Market
- North America (U.S., Canada, Mexico)
- Europe (Germany, France, U.K., Italy, Spain, Rest of Europe)
- Asia-Pacific (China, Japan, India, Rest of APAC)
- South America (Brazil and the Rest of South America)
- The Middle East and Africa (UAE, South Africa, Rest of MEA)
North America is anticipated to hold the largest share of approximately 40% the cytotoxic drugs contract manufacturing market over the forecast period.
North America is anticipated to hold the largest share of approximately 40% the cytotoxic drugs contract manufacturing market over the forecast period. The dominance of this region is primarily driven by advanced pharmaceutical infrastructure, a strong presence of major CDMOs, and high oncology drug demand. Additionally, robust regulatory frameworks, significant R&D investments, and extensive outsourcing by pharmaceutical companies support large-scale cytotoxic manufacturing capabilities in the region.
Asia Pacific is expected to grow at a rapid CAGR of approximately 9.5% in the cytotoxic drugs contract manufacturing market during the forecast period.
Asia Pacific is expected to grow at a rapid CAGR of approximately 9.5% in the cytotoxic drugs contract manufacturing market during the forecast period. This growth is driven by cost-effective manufacturing, expanding pharmaceutical industries in China and India, and increasing cancer prevalence. Rising investments in high-potency drug facilities, skilled workforce availability, and global companies shifting outsourcing to reduce costs further accelerate regional expansion.
Europe is the 3rd largest region to grow in the cytotoxic drugs contract manufacturing market during the period.
Europe is the third-largest region in the global cytotoxic drugs contract manufacturing market, supported by strong pharmaceutical hubs in countries like Germany, Switzerland, and the United Kingdom. Growth is driven by increasing oncology drug demand, the presence of specialized HPAPI manufacturing facilities, and strict regulatory standards ensuring high-quality production.
Future Market Trends in Global Cytotoxic Drugs Contract Manufacturing Market: -
1. Increasing Outsourcing to Specialized CDMOs
A major future trend is the growing reliance on contract development and manufacturing organizations (CDMOs) for cytotoxic drugs. Due to complex handling requirements and high infrastructure costs, over half of pharma companies are outsourcing production. This shift allows firms to focus on R&D while leveraging specialized expertise, scalability, and regulatory compliance.
2. Rising Demand for High-Potency APIs and Targeted Therapies
The market is witnessing strong growth in highly potent APIs (HPAPIs) and targeted oncology therapies such as antibody-drug conjugates. As cancer incidence rises, innovation in precision medicines is expanding pipelines, with many drugs in development relying on contract manufacturers. This trend is increasing demand for advanced containment and specialized manufacturing capabilities.
3. Expansion of Advanced Manufacturing Facilities and Technologies
Future growth is driven by investments in high-containment facilities, sterile injectables, and modular production systems. CDMOs are expanding capabilities in aseptic fill-finish, bioconjugation, and scalable production to meet global demand. These technological advancements improve efficiency, ensure regulatory compliance, and enable faster commercialization of complex cytotoxic therapies.
Recent Development
- In March 2026, Novartis announced that it had agreed to acquire SNV4818, a pan-mutant-selective PI3Kα inhibitor, from Synnovation Therapeutics in a deal valued at up to USD 3 billion (USD 2 billion upfront plus milestones). The acquisition strengthened Novartis’ breast cancer pipeline by adding a next-generation targeted therapy currently in early clinical development.
- In June 2024, Veranova announced that it had initiated a USD 30 million expansion of its antibody-drug conjugate (ADC) and high-potency API (HPAPI) manufacturing capabilities at its Devens, Massachusetts, facility in the United States, aiming to meet rising demand for potent oncology compounds.
- In May 2024, Sterling Pharma Solutions reported that it had invested USD 3 million to expand its ADC and HPAPI manufacturing capabilities at its Wisconsin facility, including a new GMP suite to support clinical and commercial production of cytotoxic payloads.
How is Recent Developments Helping the Market?
Recent developments are significantly accelerating growth in the global cytotoxic drugs contract manufacturing market by enhancing efficiency, safety, and scalability. Advancements in high-containment manufacturing technologies, such as isolators and closed-system equipment, have improved worker safety and regulatory compliance, enabling manufacturers to handle highly potent cytotoxic compounds more effectively. Increased outsourcing by pharmaceutical and biotechnology companies is driving demand for specialized contract manufacturers with expertise in complex oncology drugs. Strategic collaborations, facility expansions, and investments in advanced sterile injectable capabilities are further strengthening production capacity. Additionally, innovations in formulation technologies and process optimization are reducing costs and improving product quality. Regulatory support and faster approval pathways for oncology therapeutics are also encouraging manufacturers to expand their capabilities, ultimately supporting market growth and improving global access to life-saving cancer treatments.
Market Segment
This study forecasts revenue at global, regional, and country levels from 2020 to 2035. Decision Advisors has segmented the global cytotoxic drugs contract manufacturing market based on the below-mentioned segments:
Global Cytotoxic Drugs Contract Manufacturing Market, By Product Type
- Active Pharmaceutical Ingredients (APIs)
- Finished Dosage Forms (FDFs)
Global Cytotoxic Drugs Contract Manufacturing Market, By Route of Administration
- Injectable
- Oral
- Topical
Global Cytotoxic Drugs Contract Manufacturing Market, By End User
- Pharmaceutical Companies
- Biotechnology Companies
- Research Organizations
Global Cytotoxic Drugs Contract Manufacturing Market, By Regional Analysis
- North America
- US
- Canada
- Mexico
- Europe
- Germany
- UK
- France
- Italy
- Spain
- Russia
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- Australia
- Rest of Asia Pacific
- South America
- Brazil
- Argentina
- Rest of South America
- Middle East & Africa
- UAE
- Saudi Arabia
- Qatar
- South Africa
- Rest of the Middle East & Africa
Frequently Asked Questions (FAQ)
Q. Why are high-potency API (HPAPI) capabilities crucial in this market?
A. HPAPI capabilities are essential because cytotoxic drugs require specialized containment, handling, and safety systems due to their toxic nature. Contract manufacturers with HPAPI expertise can ensure compliance with stringent regulations while maintaining product integrity, making them highly preferred by pharmaceutical companies.
Q. How do strategic partnerships influence market competitiveness?
A. Strategic partnerships between pharmaceutical firms and CDMOs enhance innovation, expand manufacturing capacity, and improve global reach. These collaborations enable faster drug development, efficient technology transfer, and better regulatory compliance, strengthening competitive positioning in the market.
Q. What challenges do new entrants face in this industry?
A. New entrants face high barriers such as significant capital investment in containment facilities, strict regulatory approvals, and the need for specialized technical expertise. These challenges limit market entry and favor established players with advanced infrastructure.
Q. How does the rise of personalized medicine impact the market?
A. The growth of personalized medicine, especially targeted cancer therapies, increases demand for flexible and small-batch manufacturing. This drives CDMOs to adopt advanced technologies and scalable processes, creating new growth opportunities in the cytotoxic drugs contract manufacturing market.
Check Licence
Choose the plan that fits you best: Single User, Multi-User, or Enterprise solutions tailored for your needs.
We Have You Covered
- 24/7 Analyst Support
- Clients Across the Globe
- Tailored Insights
- Technology Tracking
- Competitive Intelligence
- Custom Research
- Syndicated Market Studies
- Market Overview
- Market Segmentation
- Growth Drivers
- Market Opportunities
- Regulatory Insights
- Innovation & Sustainability
Report Details
| Scope | Global |
| Pages | 240 |
| Delivery | PDF & Excel via Email |
| Language | English |
| Release | Apr 2026 |
| Access | Download from this page |
