Global Enzyme Contract Manufacturing Market
Global Enzyme Contract Manufacturing Market Size, Share, and COVID-19 Impact Analysis, By Source (Plant, Animal, and Microbe), By End User (Industrial Enzymes [Food and Beverage, Animal Feed, Detergent, Paper, and Others], Specialty Enzymes [Pharmaceutical Manufacturing, Diagnostics, Research and Development]) and Others]), and By Region (North America, Europe, Asia-Pacific, Latin America, Middle East, and Africa), Analysis and Forecast 2025-2035
Report Overview
Table of Contents
Global Enzyme Contract Manufacturing Market Size Insights Forecasts to 2035
- The Global Enzyme Contract Manufacturing Market Size Was Estimated at USD 2.71 Billion in 2024
- The Market Size is Expected to Grow at a CAGR of around 8.46 % from 2025 to 2035
- The Worldwide Enzyme Contract Manufacturing Market Size is Expected to Reach USD 6.62 Billion by 2035
- North America is expected to grow the fastest during the forecast period.
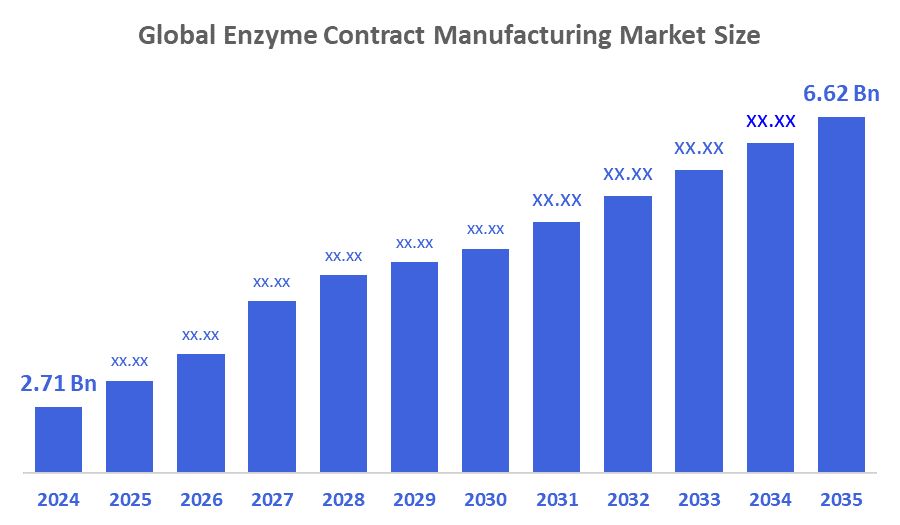
According to a research report published by Decisions Advisors and Consulting, The Global Enzyme Contract Manufacturing Market Size Was Worth Around USD 2.71 Billion In 2024 And Is Predicted To Grow To Around USD 6.62 Billion By 2035 With A Compound Annual Growth Rate (CAGR) Of 8.46 % From 2025 To 2035. The demand for healthy additives in the food and beverage industry, the need for bio-based cleaning solutions, the growing use of therapies, the preference for greener products like biofuels, government support, and other factors are driving the growth of the global enzyme contract manufacturing market.
Market Overview
Enzyme contract manufacturing is the process of synthesising enzymes by professionals who have extensive training and experience in developing, refining, and growing enzyme production systems. Enzyme contract manufacturing includes the manpower, equipment, and infrastructure required to create enzymes in large quantities at a lower cost than internal production. Enzyme contract manufacturing is growing due in large part to the growing medicinal utility of enzymes. They are used either alone or in conjunction with other medications to safely and effectively treat a range of illnesses. Furthermore, therapeutic enzymes may play a bigger role in the treatment and management of some diseases as their prevalence increases, including cancer, HIV, skin conditions, cardiovascular disease, and others. The enzyme contract manufacturing market is expected to increase in the upcoming years due to the recent emergence of enzymes from a variety of sources, including plants, animals, and microorganisms, as effective therapeutic solutions for a variety of diseases. Numerous study articles have highlighted the therapeutic benefits of various enzymes, such as lipases and collagenases, which have caused pharmaceutical and biotech businesses to concentrate more on creating medications and treatments based on enzymes.
Russian authorities approved a program to support producers of enzyme preparations, food additives, and feed additives starting January 2027. Companies that build or modernise such facilities will be eligible for reimbursement of up to 20% of direct costs. The initiative aimed to strengthen domestic production of critical inputs for food and livestock industries.
SEKISUI Diagnostics completed a £15.7 million cGMP expansion at its UK facility, boosting microbial substance manufacturing capacity. The investment strengthens production capabilities to meet growing demand for biopharmaceuticals and supports advanced microbial-based drug development in compliance with global quality standards. Large-scale manufacture of enzymes, proteins, antibody fragments, and plasmids for gene therapy is made possible by the expanded UK site, which enhances SEKISUI Diagnostics' microbial CDMO capabilities. A new 1,000L fermentation and purification suite is prepared to fulfil the growing biopharma demand.
Report Coverage
This research report categorizes the enzyme contract manufacturing market based on various segments and regions, forecasts revenue growth, and analyzes trends in each submarket. The report analyses the key growth drivers, opportunities, and challenges influencing the enzyme contract manufacturing market. Recent market developments and competitive strategies, such as expansion, product launch, development, partnership, merger, and acquisition, have been included to draw the competitive landscape in the market. The report strategically identifies and profiles the key market players and analyzes their core competencies in each sub-segment of the enzyme contract manufacturing market.
Driving Factors
The growing prevalence of non-communicable diseases such as cancer, diabetes, cardiovascular disease, and neurological disorders is driving up demand for enzymes in treatments. Treatments depend on enzymes like streptokinase and asparaginase, which improve blood flow and lessen the negative effects of chemotherapy. Simultaneously, large-scale enzyme utilisation in processes like cellulose breakdown, fermentation, and waste treatment is necessary for the global adoption of biofuels, which is being driven by government initiatives and sustainability goals. Smart decision-making, quick structure detection, process optimisation, cost effectiveness, etc., have all contributed to the rapid breakthroughs in biotechnology, including the integration of AI and ML, all of the factors driving the market growth. Moreover, recombinant DNA technology clones the genes, allowing for the large-scale manufacture of enzymes, creates news market opportunities.
BioMarin Pharmaceutical Inc. acquired Inozyme Pharma in an all-cash deal valued at about $270 million. The acquisition brings INZ-701, a Phase 3 enzyme replacement therapy for ENPP1 Deficiency, into BioMarin’s pipeline. First pivotal data in children is expected in early 2026, with a potential launch targeted for 2027, strengthening BioMarin’s rare disease portfolio.
Restraining Factors
Enzyme production is expensive due to the cost of raw materials, intricate multi-step procedures, and strict regulations. Approximately 80 per cent of expenses are related to purification alone, and infrastructure and quality control requirements are significant cost drivers. Further, the additional burden of adhering to EU, FDA, and FAO/WHO regulations.
Market Segmentation
The enzyme contract manufacturing market share is classified into source and end user.
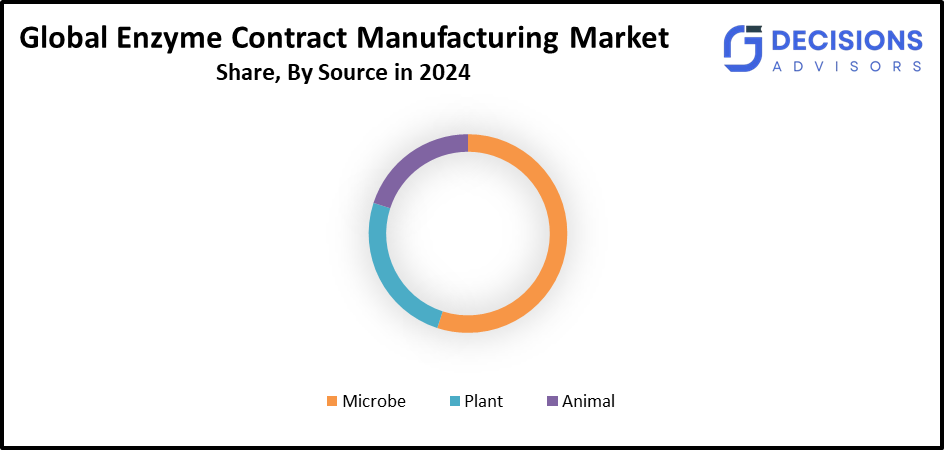
- The microbe segment accounted for the largest share in 2024 and is anticipated to grow at a significant CAGR during the forecast period.
Based on the source, the enzyme contract manufacturing market is segmented into plant, animal, and microbe. Among these, the microbe segment accounted for the largest share in 2024 and is anticipated to grow at a significant CAGR during the forecast period. It is a result of the microorganisms' quick growth, their simpler application in a variety of industries, their adaptable genetics, their low environmental waste, their inexpensive production costs, and technological advancements like fermentation and genetics. The microorganisms have a fast rate of reproduction, which causes their population to grow exponentially.
- The industrial enzymes segment accounted for the highest market revenue in 2024 and is anticipated to grow at a significant CAGR during the forecast period.
Based on the end user, the enzyme contract manufacturing market is divided into industrial enzymes [food and beverage, animal feed, detergent, paper, and others], specialty enzymes [pharmaceutical manufacturing, diagnostics, research and development and others]. Among these, the industrial enzymes segment accounted for the highest market revenue in 2024 and is anticipated to grow at a significant CAGR during the forecast period. This is a result of customers' growing preference for natural products, their health advantages, and their many uses in the food sector, including dairy, baking, fish and meat processing, and the manufacturing of beverages like wine and beer. The use of enzymes in food production preserves the food's natural nutrients, reducing the rising incidence of food-related illnesses.
Regional Segment Analysis of the Enzyme Contract Manufacturing Market
- North America (U.S., Canada, Mexico)
- Europe (Germany, France, U.K., Italy, Spain, Rest of Europe)
- Asia-Pacific (China, Japan, India, Rest of APAC)
- South America (Brazil and the Rest of South America)
- The Middle East and Africa (UAE, South Africa, Rest of MEA)
Asia Pacific is anticipated to hold the largest share of the enzyme contract manufacturing market over the predicted timeframe.
Asia Pacific is anticipated to hold the largest share of the enzyme contract manufacturing market over the predicted timeframe. The market for enzyme contract manufacturing in Asia-Pacific is expanding steadily due to increased demand from the food, pharmaceutical, and healthcare sectors. Expansion is being driven by the growing use of industrial enzymes in processes including food processing, animal feed, detergents, and papermaking. Contract manufacturing opportunities are also being enhanced by the region's robust biotechnology base, expanding R&D investments, and the introduction of novel enzyme products. Moreover, their affordable production and access in countries like China, India, and Japan are becoming major hubs. The market is expected to gain from growing biopharmaceutical production and increased attention to environmentally friendly, sustainable industrial processes, making Asia-Pacific a key participant in the worldwide outsourcing of enzymes.
Morepen Laboratories has signed a Rs 825 crore contract to expand into the Contract Development and Manufacturing Organisation (CDMO) segment. The multi-year deal with a leading global pharma company will begin supplies within 4–5 months, strengthening Morepen’s international manufacturing presence and marking one of its largest mandates to date.
North America is expected to grow at a rapid CAGR in the enzyme contract manufacturing market during the forecast period. The market expansion occurs because end-user industries like pharmaceuticals, food production, breweries and other sectors drive demand. Moreover, key players, technological advancements, environmental rules, quality requirements and other factors establish market growth. The development of new enzymes for medical research and diagnostic tools has occurred because genetic science has achieved technological progress.
Codexis, Inc. signed a lease for a 34,000-square-foot GMP manufacturing facility in Hayward, California. The site will expand Codexis’ capabilities into GMP manufacturing of siRNA and other oligonucleotides using its ECO Synthesis platform. Facility modifications are expected to begin in early 2026, with flexibility to scale enzyme production.
Competitive Analysis:
The report offers the appropriate analysis of the key organizations/companies involved within the enzyme contract manufacturing market, along with a comparative evaluation primarily based on their product offering, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborative analysis focusing on the current news and developments of the companies, which includes product development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
List of Key Companies
- BRAIN Biotech AG
- Creative Enzymes
- EKF Diagnostics Holdings plc
- Enzyme Bioscience
- Enzymology Research Centre, Inc.
- IBEX Technologies Inc.
- Meteoric Biopharmaceuticals Limited.
- Prozomix Limited
- SEKISUI CHEMICAL CO., LTD.
- Tex Biosciences (P) Ltd.
- ZYMVOL
- Others
Key Target Audience
- Market Players
- Investors
- End-users
- Government Authorities
- Consulting and Research Firm
- Venture capitalists
- Value-Added Resellers (VARs)
Recent Development
- In January 2026, Quantumzyme Corp. unveiled a next-generation enzyme engineering platform aimed at boosting efficiency in pharmaceutical manufacturing. The breakthrough introduces high-performance enzymes with improved stability, selectivity, and solvent compatibility, enabling the use of lower-cost options like isopropyl alcohol instead of ethyl acetate. This innovation supports more sustainable and cost-effective drug production.
- In June 2025, NIZO and Enzymit announced a collaboration to scale cell-free enzyme production, combining NIZO’s expertise in food-grade fermentation and process development with Enzymit’s proprietary cell-free enzyme engineering technology. The partnership aims to accelerate sustainable enzyme manufacturing, reduce production costs, and open new applications in the food and pharmaceutical industries.
- In May 2025, BRAIN Biotech AG completed the buy-out of minority shares in its Dutch subsidiary Breatec B.V., making it fully owned. Alongside this, the company announced a new continental European production site near Eindhoven, Netherlands, and the expansion of its Baking Application Centre to strengthen enzyme-based baking solutions and consolidate its biocatalysts division.
- In April 2024, Ginkgo Bioworks and UK-based Prozomix announced a partnership to develop next-generation enzymes for sustainable API manufacturing. The collaboration combines Ginkgo’s AI/ML-driven enzyme engineering expertise with Prozomix’s extensive enzyme sequence libraries and manufacturing capabilities. Together, they aim to create advanced enzyme plates that improve efficiency, reduce costs, and support greener pharmaceutical production.
Market Segment
This study forecasts revenue at global, regional, and country levels from 2020 to 2035. Decisions Advisors has segmented the enzyme contract manufacturing market based on the below-mentioned segments:
Global Enzyme Contract Manufacturing Market, By Source
- Plant
- Animal
- Microbe
Global Enzyme Contract Manufacturing Market, By End User
- Industrial Enzymes
- Food and Beverage
- Animal Feed
- Detergent
- Paper
- Others
- Specialty Enzymes
- Pharmaceutical Manufacturing
- Diagnostics
- Research and Development
- Others
Global Enzyme Contract Manufacturing Market, By Regional Analysis
- North America
- US
- Canada
- Mexico
- Europe
- Germany
- UK
- France
- Italy
- Spain
- Russia
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- Australia
- Rest of Asia Pacific
- South America
- Brazil
- Argentina
- Rest of South America
- Middle East & Africa
- UAE
- Saudi Arabia
- Qatar
- South Africa
- Rest of the Middle East & Africa
Frequently Asked Questions (FAQ)
- What factors are influencing outsourcing decisions in enzyme manufacturing?
Companies increasingly outsource enzyme production to reduce capital expenditure, accelerate time-to-market, access specialised fermentation technologies, and leverage regulatory expertise. Outsourcing also enables flexibility in scaling production volumes without long-term infrastructure investments.
- How does sustainability impact the enzyme contract manufacturing market?
Sustainability plays a major role, as enzymes support green chemistry, reduce energy consumption, and lower carbon emissions in industrial processes. Contract manufacturers adopting eco-friendly production methods and waste reduction technologies gain competitive advantages.
- What technological innovations are improving enzyme yield and efficiency?
Advancements such as precision fermentation, synthetic biology, computational protein design, and high-throughput screening are enhancing enzyme stability, specificity, and scalability. These technologies help CDMOs optimise productivity while lowering operational costs.
- How does regulatory compliance affect global enzyme CDMOs?
Contract manufacturers must comply with international standards such as GMP, ISO certifications, and regional pharmaceutical regulations. Regulatory readiness influences client trust, cross-border exports, and partnerships with multinational pharmaceutical and food companies.
- What challenges do small and mid-sized enzyme CDMOs face?
Smaller CDMOs often face barriers such as high equipment costs, limited access to advanced bioreactors, skilled workforce shortages, and competition from established multinational players with broader production capacities.
- How is demand for speciality enzymes shaping future contract manufacturing strategies?
The rising application of speciality enzymes in biologics, personalised medicine, and molecular diagnostics is encouraging CDMOs to expand high-purity production lines, invest in advanced purification systems, and develop customised enzyme solutions.
- What role do emerging economies play in the future of enzyme contract manufacturing?
Emerging economies are becoming attractive outsourcing destinations due to lower operational costs, expanding biotech infrastructure, government incentives, and skilled scientific talent pools, contributing to global supply chain diversification.
Check Licence
Choose the plan that fits you best: Single User, Multi-User, or Enterprise solutions tailored for your needs.
We Have You Covered
- 24/7 Analyst Support
- Clients Across the Globe
- Tailored Insights
- Technology Tracking
- Competitive Intelligence
- Custom Research
- Syndicated Market Studies
- Market Overview
- Market Segmentation
- Growth Drivers
- Market Opportunities
- Regulatory Insights
- Innovation & Sustainability
Report Details
| Scope | Global |
| Pages | 230 |
| Delivery | PDF & Excel via Email |
| Language | English |
| Release | Mar 2026 |
| Access | Download from this page |
