Global Extracorporeal Co2 Removal Device Market
Global Extracorporeal Co2 Removal Device Market Size, Share, and COVID-19 Impact Analysis, By Technology (Membrane Technology, Absorption Technology, Cryogenic Technology, Adsorption Technology), By Application (Hospital Use, Home Care, Emergency Medical Services, Transport), By Device Type (Portable Devices, Stationary Devices, Single-Use Devices), and By Region (North America, Europe, Asia-Pacific, Latin America, Middle East, and Africa), Analysis and Forecast 2025-2035
Report Overview
Table of Contents
Global Extracorporeal Co2 Removal Device Market Size Insights Forecasts to 2035
- The Global Extracorporeal Co2 Removal Device Market Size Was Estimated at USD 114.53 Million in 2024
- The Market Size is Expected to Grow at a CAGR of around 6.43 % from 2025 to 2035
- The Worldwide Extracorporeal Co2 Removal Device Market Size is Expected to Reach USD 227.43 Million by 2035
- North America is expected to grow the fastest during the forecast period.
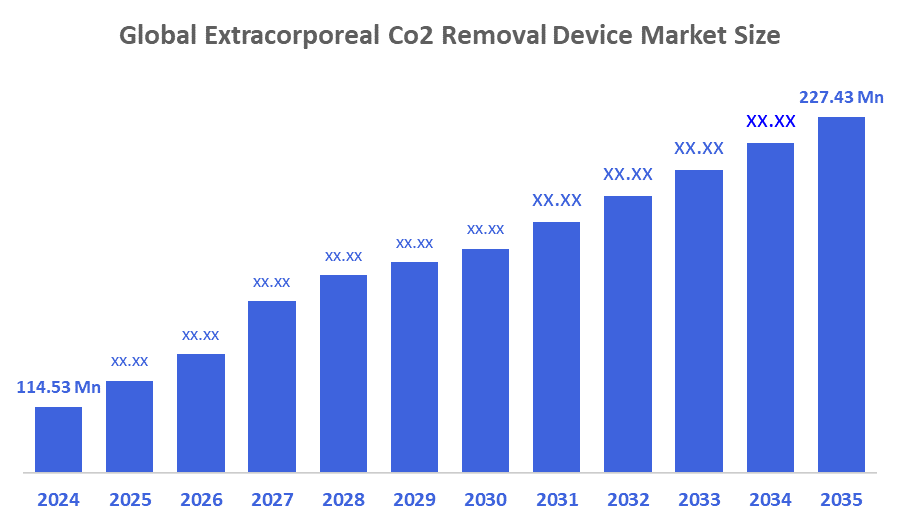
According to a research report published by Decisions Advisors and Consulting, The Global Extracorporeal Co2 Removal Device Market Size Was Worth Around USD 114.53 Million In 2024 And Is Predicted To Grow To Around USD 227.43 Million By 2035 With A Compound Annual Growth Rate (CAGR) Of 6.43 % From 2025 To 2035. The increasing prevalence of acute pulmonary illnesses, the expanding global burden of respiratory failure, and the growing demand for less invasive respiratory support technologies are all driving the extracorporeal CO2 removal devices market's steady advancement. Alternative CO2 removal methods have been adopted as a result of the problems associated with traditional ventilation tactics, such as barotrauma and oxygen poisoning.
Market Overview
The extracorporeal co? removal (ecco?r) device market is defined as the global industry segment focused on medical technologies that enable the removal of carbon dioxide from a patient’s blood outside the body. The devices function as supplementary solutions that replace mechanical ventilation in patients suffering from acute respiratory distress syndrome, chronic obstructive pulmonary disease, and various types of hypercapnic respiratory failure. The market includes devices and consumables, and associated services, which hospitals use in intensive care units, emergency departments and specialised respiratory treatment facilities. The increasing respiratory disease rates and rising icu patient numbers and requirement for advanced life-support systems, which minimise ventilator-related complications, drive market expansion. The system still faces several difficulties because of its expensive nature and need for expert education, and the dangers associated with extracorporeal blood circulation. The ecco?r market serves as an essential point where critical care medicine meets medical technology development, which works to enhance patient health by using non-invasive breathing assistance methods.
Report Coverage
This research report categorizes the extracorporeal CO2 removal device market based on various segments and regions, forecasts revenue growth, and analyzes trends in each submarket. The report analyses the key growth drivers, opportunities, and challenges influencing the extracorporeal Co2 removal device market. Recent market developments and competitive strategies, such as expansion, product launch, development, partnership, merger, and acquisition, have been included to draw the competitive landscape in the market. The report strategically identifies and profiles the key market players and analyzes their core competencies in each sub-segment of the extracorporeal CO2 removal device market.
Driving Factors
The increasing need for cutting-edge respiratory support technologies in the treatment of patients with acute respiratory failure, chronic obstructive pulmonary disease, and acute respiratory distress syndrome (ARDS) is another factor propelling the market's expansion. Moreover, there is a growing need for their use, particularly in intensive care units (ICUs), where they lessen the severity of lung damage brought on by ventilators by permitting lower tidal volumes during ventilation. Technological developments have improved the efficiency, compactness, and ease of integration of extracorporeal CO2 removal (ECCO?R) systems with other extracorporeal life support systems, including extracorporeal membrane oxygenation (ECMO). Furthermore, ECCO?R offers a substitute for non-invasive ventilation (NIV) in patients with persistent hypercapnia, particularly in cases where NIV is poorly tolerated or does not adequately clear CO?. This has caused a rise in the use of ECCO?R devices by pulmonologists and anesthesiologists who treat high-risk patients in specialist pulmonary care units.
Baxter International has announced the global launch of the PrisMax 2 system, a next-generation platform designed to enhance critical care delivery in intensive care units (ICUs). This launch positions Baxter to compete strongly in the critical care technology space, especially as demand for advanced extracorporeal therapies continues to grow.
For instance, in Europe, Medtronic introduced its VitalFlow ECMO system, a cutting-edge extracorporeal support platform for critical cardiac and respiratory treatment. This launch represents ongoing investment in extracorporeal technology, such as sophisticated gas exchange and CO2 management, and comes after Medtronic acquired MC3 Cardiopulmonary.
Restraining Factors
Market expansion is constrained by high device costs, difficult regulatory approvals, and sparse clinical use. Particularly in developing nations, reliance on specialised infrastructure, complexities, and reimbursement issues further impede growth. Widespread integration into critical care practices is further delayed by clinicians' slow awareness.
Market Segmentation
The extracorporeal Co2 removal device market share is classified into technology type, application, and device type.
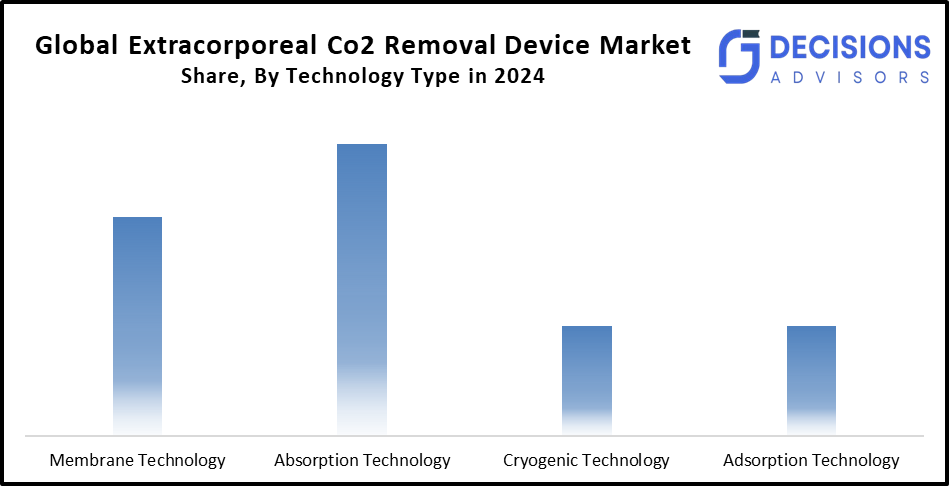
- The absorption technology segment accounted for the largest market share in 2024 and is anticipated to grow at a substantial CAGR over the forecast period.
Based on the technology type, the extracorporeal Co2 removal device market is divided into membrane technology, absorption technology, cryogenic technology, and adsorption technology. Among these, the absorption technology segment accounted for the largest market share in 2024 and is anticipated to grow at a substantial CAGR over the forecast period. The segment growth is fueled by the necessity for patient movement and flexibility. Their rapid deployment capabilities are crucial in acute care settings.
- The hospital use segment accounted for the highest market revenue in 2024 and is anticipated to grow at a significant CAGR during the forecast period.
Based on the application, the extracorporeal Co2 removal device market is divided into hospital use, home care, emergency medical services, and transport. Among these, the hospital use segment accounted for the highest market revenue in 2024 and is anticipated to grow at a significant CAGR during the forecast period. This is distinguished by its dependence on highly qualified healthcare workers and sophisticated medical systems. By incorporating these devices into critical care environments, hospitals improve surgical and intensive care patient outcomes.
- The portable devices segment accounted for the highest market revenue in 2024 and is anticipated to grow at a significant CAGR during the forecast period.
Based on the device type, the extracorporeal Co2 removal device market is divided into portable devices, stationary devices, and single-use devices. Among these, the portable devices segment accounted for the highest market revenue in 2024 and is anticipated to grow at a significant CAGR during the forecast period. This is driven by the requirement for adaptability and patient mobility. Moreover, they can be quickly deployed in an emergency, and they are crucial in acute care circumstances.
Regional Segment Analysis of the Extracorporeal Co2 Removal Device Market
- North America (U.S., Canada, Mexico)
- Europe (Germany, France, U.K., Italy, Spain, Rest of Europe)
- Asia-Pacific (China, Japan, India, Rest of APAC)
- South America (Brazil and the Rest of South America)
- The Middle East and Africa (UAE, South Africa, Rest of MEA)
Asia Pacific is anticipated to hold the largest share of the extracorporeal Co2 removal device market over the predicted timeframe.
Asia Pacific is anticipated to hold the largest share of the extracorporeal Co2 removal device market over the predicted timeframe. The market for extracorporeal CO? removal devices is expanding significantly in the Asia Pacific as a result of the rising prevalence of COPD and acute respiratory failure in nations like China, Japan, South Korea, and India. In addition, the need for sophisticated respiratory support systems in critical care settings has increased because of the development of chronic respiratory disorders brought on by rapid urbanisation and industrial pollution. The country's ageing population and the growing incidence of chronic respiratory diseases like COPD are the main causes of this. For the purpose of treating respiratory disorders in older patients who are frequently unsuitable candidates for extracorporeal CO2 removal devices, Japanese healthcare providers are increasingly using cutting-edge respiratory support technology. The market for extracorporeal CO? removal devices and Japan's well-established healthcare infrastructure in China, along with the country's proactive embrace of cutting-edge medical technologies, have further boosted the use of ECCO?R systems in Chinese healthcare institutions.
North America is expected to grow at a rapid CAGR in the extracorporeal Co2 removal device market during the forecast period. The growing clinical usage in specialised care settings is driving the North American market for extracorporeal CO2 removal devices. The incorporation of ECCO?R technologies in the management of patients with acute respiratory failure and chronic obstructive pulmonary disease (COPD) is supported by the region's sophisticated healthcare infrastructure and high awareness among healthcare professionals. The market for extracorporeal CO? removal devices is expanding significantly in the United States due to the rising incidence of chronic respiratory conditions and the growing need for less intrusive alternatives to mechanical ventilation. Additionally, the market is expanding due to the growing demand for cutting-edge medical equipment and the growing spending in R&D on respiratory illnesses.
For example, the American Lung Association created its Research Institute in 2023 with the goal of increasing scientific knowledge and lung condition therapy. The organisation pledged to increase its yearly investment in lung illness research to USD 25 million.
The U.S. Food and Drug Administration (FDA) has cleared the Novalung® system, developed by Fresenius Medical Care, for the treatment of acute respiratory and cardiopulmonary failure. This is a significant milestone because Novalung is the first extracorporeal membrane oxygenation (ECMO) system cleared for use beyond six hours as extracorporeal life support.
Competitive Analysis:
The report offers the appropriate analysis of the key organizations/companies involved within the extracorporeal Co2 removal device market, along with a comparative evaluation primarily based on their product offering, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborative analysis focusing on the current news and developments of the companies, which includes product development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
List of Key Companies
- Aferetica s.r.l.
- ALung Technologies, Inc.
- Baxter International Inc
- Eurosets SRL
- Fresenius Medical Care AG & Co KGaA
- Getinge AB
- LivaNova Plc ESTOR S.P.A.
- Medica S.p.A.
- Medtronic Plc
- Terumo Corp
- X-COR Therapeutics
- Xenios AG
- Others
Key Target Audience
- Market Players
- Investors
- End-users
- Government Authorities
- Consulting and Research Firm
- Venture capitalists
- Value-Added Resellers (VARs)
Market Segment
This study forecasts revenue at global, regional, and country levels from 2020 to 2035. Decisions Advisors has segmented the extracorporeal Co2 removal device market based on the below-mentioned segments:
Global Extracorporeal Co2 Removal Device Market, By Technology Type
- Membrane Technology
- Absorption Technology
- Cryogenic Technology
- Adsorption Technology
Global Extracorporeal Co2 Removal Device Market, By Device Type
- Portable Devices
- Stationary Devices
- Single-Use Devices
Global Extracorporeal Co2 Removal Device Market, By Application
- Hospital Use
- Home Care
- Emergency Medical Services
- Transport
Global Extracorporeal Co2 Removal Device Market, By Regional Analysis
- North America
- US
- Canada
- Mexico
- Europe
- Germany
- UK
- France
- Italy
- Spain
- Russia
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- Australia
- Rest of Asia Pacific
- South America
- Brazil
- Argentina
- Rest of South America
- Middle East & Africa
- UAE
- Saudi Arabia
- Qatar
- South Africa
- Rest of the Middle East & Africa
Frequently Asked Questions (FAQ)
1. How does Extracorporeal CO? Removal (ECCO?R) differ from traditional ECMO therapy?
ECCO?R primarily focuses on removing carbon dioxide at lower blood flow rates, whereas ECMO (Extracorporeal Membrane Oxygenation) provides both oxygenation and CO? removal at higher flow rates. ECCO?R is generally considered less invasive and is often used to support lung-protective ventilation strategies rather than full cardiopulmonary support.
2. What patient populations are most likely to benefit from ECCO?R therapy?
Patients with acute exacerbations of chronic obstructive pulmonary disease (COPD), acute respiratory distress syndrome (ARDS), severe asthma, and hypercapnic respiratory failure are among the primary beneficiaries. It is particularly useful for patients who fail non-invasive ventilation but may not yet require full ECMO support.
3. What are the key clinical risks associated with ECCO?R devices?
Potential risks include bleeding, thrombosis, hemolysis, infection at cannulation sites, and complications related to anticoagulation therapy. Proper patient selection and skilled clinical management are essential to minimise these risks.
4. How is artificial intelligence (AI) influencing the development of ECCO?R systems?
AI integration is being explored to optimise blood flow control, monitor gas exchange efficiency, predict complications, and personalise treatment parameters. Smart monitoring systems may improve safety and clinical outcomes while reducing ICU workload.
5. What role does reimbursement policy play in market adoption?
Reimbursement frameworks significantly influence hospital purchasing decisions. Favourable insurance coverage and government funding for advanced respiratory therapies can accelerate adoption, while unclear or limited reimbursement structures may restrict market penetration.
6. How are clinical trials shaping the future of the ECCO?R market?
Ongoing and future clinical trials are critical for validating safety, demonstrating improved survival or reduced ventilation duration, and expanding approved indications. Positive trial outcomes may support broader regulatory approvals and stronger physician confidence.
7. What emerging innovations could transform the ECCO?R device landscape by 2035?
Future innovations may include wearable or miniaturised ECCO?R systems, improved biocompatible materials to reduce clotting risks, hybrid systems combining renal and respiratory support, and simplified interfaces designed for faster deployment in emergency and transport settings.
Check Licence
Choose the plan that fits you best: Single User, Multi-User, or Enterprise solutions tailored for your needs.
We Have You Covered
- 24/7 Analyst Support
- Clients Across the Globe
- Tailored Insights
- Technology Tracking
- Competitive Intelligence
- Custom Research
- Syndicated Market Studies
- Market Overview
- Market Segmentation
- Growth Drivers
- Market Opportunities
- Regulatory Insights
- Innovation & Sustainability
Report Details
| Scope | Global |
| Pages | 250 |
| Delivery | PDF & Excel via Email |
| Language | English |
| Release | Mar 2026 |
| Access | Download from this page |
