Global Friedreichs Ataxia Market
Global Friedreichs Ataxia Market Size, Share, By Patient Age Group (Pediatric, Adolescent, Adult, Elderly) By Diagnosis Method (Genetic Testing, Clinical Examination, MRI, EMG, Neurological Assessment) By Treatment Type (Gene Therapy, Pharmacological Treatments, Physical Therapy, Nutritional Supplements, Supportive Care) By End User (Hospitals, Clinics, Home Care, Research Institutes) and By Region (North America, Europe, Asia-Pacific, Latin America, Middle East & Africa), Analysis and Forecast 2025 - 2035
Report Overview
Table of Contents
Market Snapshot
- Market Size (2025): USD 2.97 Billion
- Projected Market Size (2035): USD 8.63 Billion
- Compound Annual Growth Rate (CAGR): 11.26%
- Largest Regional Market: North America
- Fastest Growing Region: Asia-Pacific
- 3rd Largest Region: Europe
- Base Year: 2025
- Historical Period: 2021–2024
- Forecast Period: 2025–2035
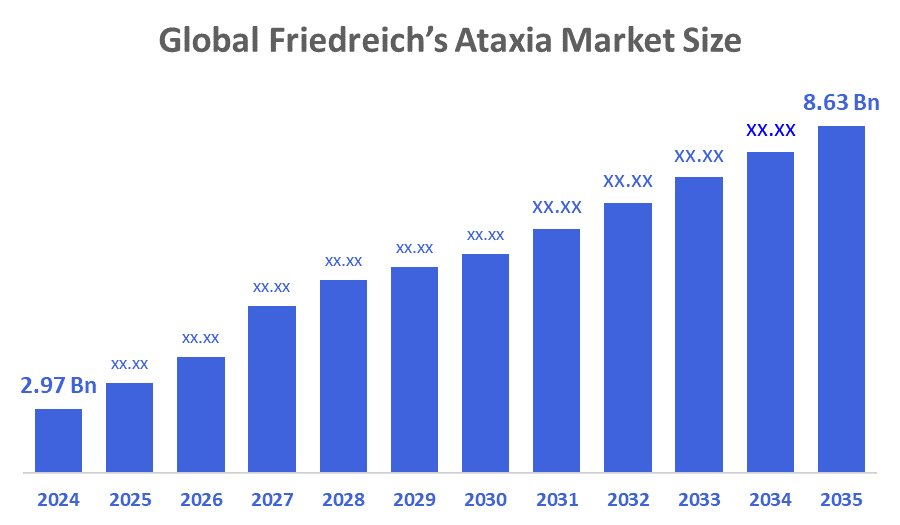
According to Decision Advisors, The Global Friedreich’s Ataxia Market Size is expected to Grow from USD 2.97 Billion in 2025 to USD 8.63 Billion by 2035, at a CAGR of 11.26% during the forecast period 2025-2035. The global friedreich’s ataxia market growth is driven by increasing prevalence of rare neurological disorders, rising patient awareness, expansion of specialized care centers, advancements in supportive and multidisciplinary therapies, growing healthcare expenditure, and enhanced public–private initiatives promoting early diagnosis and disease management.
Market Overview/ Introduction
The global friedreich’s ataxia (fa) market comprises therapies, diagnostics, and care services for patients affected by this rare, inherited neurodegenerative disorder caused by FXN gene mutations. Friedreich’s Ataxia is a genetic disorder characterized by progressive damage to the nervous system, leading to impaired muscle coordination (ataxia), cardiomyopathy, diabetes, and other neurological complications, typically manifesting in childhood or adolescence. The market includes pharmacological treatments, gene therapies, supportive care, and advanced diagnostics, aiming to improve patient outcomes and quality of life. The market scope spans patient age groups from pediatric to elderly, with genetic testing, clinical assessments, and advanced imaging forming the primary diagnostic approaches. Key treatment options include pharmacological therapies, gene therapy, physical therapy, and nutritional supplementation, delivered through hospitals, clinics, and specialized centers. Geographically, North America and Europe dominate due to advanced healthcare infrastructure, while Asia-Pacific is emerging rapidly with rising awareness, improved diagnostics, and expanding healthcare access. Future opportunities lie in disease-modifying therapies, gene therapy innovations, AI-enabled diagnostics, expanded pediatric care, and public-private collaborations. Growth is further supported by increased funding, early intervention programs, and rising patient awareness globally.
- FDA and EMA Orphan Drug Designation provides tax credits, fee waivers, and extended market exclusivity for friedreich’s ataxia therapies such as Larimar Therapeutics Nomlabofusp and Lexeo LX2006, encouraging development and accelerating global market growth.
- Larimar Therapeutics CTN 001 received FDA Breakthrough Therapy designation in 2024, enabling faster clinical development and regulatory review, supporting quicker market access for innovative treatments and driving expansion of the global friedreich’s ataxia market.
- Organizations such as FARA and EFACTS collaborate with governments to fund research, maintain patient registries, and support clinical trials, strengthening pipeline development and evidence generation, thereby promoting growth in the global friedreich’s ataxia market.
- Governments in Japan and China are implementing rare disease initiatives that facilitate drug approvals, expand healthcare access, and promote therapy adoption, accelerating regional growth and strengthening the Asia-Pacific contribution to the global market.
Notable Insights: -
- Asia-Pacific holds the largest regional market share, accounting for approximately 20.74% of the global friedreich’s ataxia market.
- North America is the fastest-growing regional market share, accounting for approximately 48.6% of the global friedreich’s ataxia market.
- Europe is the second-largest regional market, holding approximately 32% of the global friedreich’s ataxia market.
- By age group, the pediatric segment held a dominant position, accounting for approximately 72% of the global friedreich’s ataxia market share in 2025.
- By diagnosis method, the genetic testing segment is the leading contributor, representing around 96% of the global market share in 2025.
- By treatment type, the pharmacological treatments segment dominated the market with an estimated 88% share in 2025.
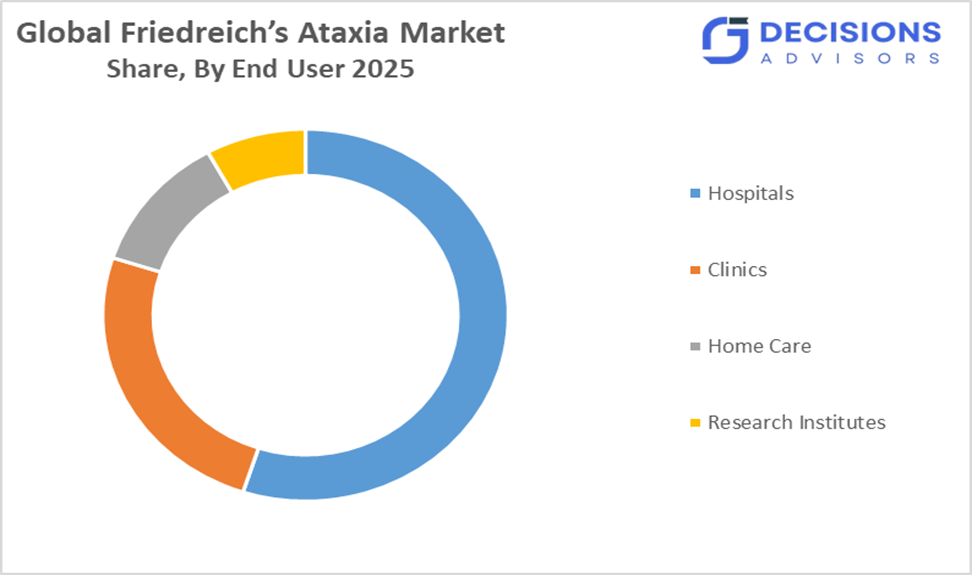
- By end user, the hospitals segment held the largest share, accounting for approximately 55% of the global friedreich’s ataxia market in 2025.
- The compound annual growth rate of the global friedreich’s ataxia market is 11.26%.
- The market is likely to achieve a valuation of USD 8.63 billion by 2035.
What is role of technology in grooming the market?
The role of technology in grooming the global friedreich’s ataxia (FA) market is significant, as it enables early diagnosis, accelerates therapy development, and improves patient management. Advanced genetic testing and high-throughput sequencing allow precise identification of FXN gene mutations, expanding the treatable patient pool. AI and machine learning are improving imaging analysis, clinical trial design, and real-world evidence generation, increasing efficiency and reducing costs. Gene-editing tools like CRISPR and novel gene therapy delivery platforms facilitate development of disease-modifying therapies. Digital platforms and telemedicine enhance patient monitoring and access, supporting adoption and growth of FA treatments worldwide.
How is Recent Developments Helping the Market?
Recent developments are driving growth in the global Friedreich’s Ataxia market by expanding treatment options, increasing patient access, and attracting investment. Larimar Therapeutics’ Nomlabofusp BLA submission and gene therapies like Lexeo’s LX2006 and Solid Biosciences’ SGT?212 provide disease-modifying solutions, addressing the root genetic cause and unmet medical needs. Biogen’s BRAVE pediatric trial broadens access to younger patients, enlarging the treatable population. Regulatory progress, including FDA designations and evaluation of PTC Therapeutics’ Vatiquinone, reduces market entry barriers and enhances confidence among payers and investors. Collectively, these initiatives accelerate adoption, stimulate R&D, and strengthen the overall FA therapeutic market.
Market Drivers
The global friedreich’s ataxia market is driven by the shift from symptom management to disease-modifying therapies, including antioxidant/Nrf2 activators and mitochondrial stabilizers, with omaveloxolone capturing a significant market share. Gene therapy innovations from companies like Lexeo Therapeutics and Solid Biosciences, along with CRISPR-based approaches, are attracting investment and shaping future growth. Increasing R&D and strategic collaborations between pharmaceutical companies and academic institutions are accelerating the development of novel treatments. Improved genetic testing and clinician awareness are enabling earlier and more accurate diagnoses, expanding the treatable patient pool. Regulatory support, including orphan drug designations, is facilitating faster development and approval. The Asia-Pacific region is emerging as a fast-growing market, supported by rising awareness, improving healthcare infrastructure, and increased access to specialized neurological care.
Restrain
The global friedreich’s ataxia market faces significant challenges, including extremely high treatment costs, with therapies often exceeding $300,000 per year, creating payer resistance. The small, dispersed patient population of around 25,000 globally limits clinical trial recruitment and overall market size. Stringent regulatory requirements and lengthy trial timelines delay approvals, while diagnostic delays and disease complexity, including cardiomyopathy, ataxia, and diabetes, hinder broad treatment access and universal therapy development.
Competitive Analysis:
The report offers the appropriate analysis of the key organizations/companies involved within the friedreich’s ataxia market, along with a comparative evaluation primarily based on their product of offering, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborative analysis focusing on the current news and developments of the companies, which includes product development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
Top Companies in Global Friedreich’s Ataxia Market
- Biogen Inc.
- Larimar Therapeutics
- Design Therapeutics
- PTC Therapeutics
- Minoryx Therapeutics
- Lexeo Therapeutics
- Retrotope Inc.
- Voyager Therapeutics
Government Initiatives
|
Country |
Key Government Initiatives |
|
US |
The FDA approved Skyclarys (Omaveloxolone) in February 2023 for adults and adolescents aged 16 and older, providing the first approved therapy for FA and expanding treatment access. The FDA has also granted Orphan Drug, Fast Track, Priority Review, and Rare Pediatric Disease designations to multiple FA candidates, accelerating development. CMS approved a specific ICD?10 diagnostic code (G11.11) for FA to improve insurance coverage and track disease progression. |
|
EU |
The European Commission approved Skyclarys in February 2024 for patients aged 16 and older, enabling treatment access in Europe. Historical EU-funded initiatives like EFACTS (European Friedreich’s Ataxia Consortium for Translational Studies) contributed to patient registries and clinical data infrastructure, supporting research and assessment tools for FA. |
Market Segmentation
The friedreich’s ataxia market share is classified into age group, diagnosis method, treatment type, and end user.
- The pediatric segment dominated the market in 2025, and is anticipated to grow at a significant CAGR of approximately 72 % during the forecast period.
Based on patient age group, the friedreich’s ataxia market is divided into pediatric, adolescent, adult, and elderly. Among these, the pediatric segment dominated the market in 2025, accounting for approximately 72?% of the total market share. This is because the disease primarily manifests during childhood, early diagnosis allows timely pharmacological and supportive interventions, and specialized pediatric care centers improve treatment adherence and outcomes, driving higher adoption and consistent demand in both developed and emerging regions globally.
- The genetic testing segment dominated the market in 2025, and is anticipated to grow at a significant CAGR of approximately 96 % during the forecast period.
Based on the diagnosis method, the friedreich’s ataxia market is divided into genetic testing, clinical examination, magnetic resonance imaging, electromyography, and neurological assessment. Among these, the genetic testing segment dominated the market in 2025, accounting for approximately 96?% of the total market share. This is because genetic testing enables early and precise detection of FA, allowing timely clinical interventions, personalized treatment planning, and accurate disease monitoring, supported by advanced molecular diagnostic infrastructure and growing awareness across North America and other high-income regions.
- The pharmacological treatments segment dominated the market in 2025, and is anticipated to grow at a significant CAGR of approximately 88 % during the forecast period.
Based on treatment type, the friedreich’s ataxia market is divided into pharmacological treatments, gene therapy, physical therapy, nutritional supplements, and supportive care. Among these, the pharmacological treatments segment dominated the market in 2025, accounting for approximately 88?% of the total market share. This is because pharmacological therapies provide immediate symptom relief, improve quality of life, support long-term disease management, and are widely accessible in hospitals and specialized clinics, with high adoption driven by physicians’ preference for established, evidence-based treatments.
- The hospitals segment dominated the market in 2025, and is anticipated to grow at a significant CAGR of approximately 55 % during the forecast period.
Based on end user, the friedreich’s ataxia market is divided into hospitals, clinics, home care, and research institutes. Among these, the hospitals segment dominated the market in 2025, accounting for approximately 55?% of the total market share. This is because hospitals provide comprehensive care including advanced diagnostics, multidisciplinary teams, timely interventions, and ongoing patient monitoring, which are essential for managing complex FA cases and ensuring higher treatment adherence and improved clinical outcomes.
What is the Reason of the Region Dominance?
The dominance of certain regions in the global friedreich’s ataxia market is primarily driven by rising healthcare access, growing awareness of rare neurological diseases, expanding hospital and clinic networks, and early adoption of advanced diagnostic technologies and treatments. Regions such as North America lead due to well-established hospitals, widespread availability of genetic testing and pharmacological treatments, and strong insurance coverage. Asia-Pacific is rapidly growing with investments in specialized care centers, government initiatives, and public-private partnerships. Europe maintains strong market share through advanced healthcare systems, high patient awareness, and extensive adoption of gene therapy, physical therapy, and supportive care programs.
Strategies to Implement for Growth of the Market in Non-Leading Regions
Growth in non-leading regions for the global friedreich’s ataxia market can be supported through strategic investments, increased awareness programs, and expansion of healthcare infrastructure. Companies should introduce affordable and locally adapted diagnostic and treatment solutions that cater to regional patient needs. Partnerships with hospitals, clinics, and rare disease organizations can enhance access to care and education. Expanding distribution channels, including telemedicine and e-health platforms, can reach underserved areas. Additionally, government collaborations, patient advocacy programs, and community engagement initiatives can raise awareness, promote early diagnosis, and improve adoption of pharmacological treatments, gene therapy, and supportive care, collectively driving market growth in these regions.
Regional Segment Analysis of the Friedreich’s Ataxia Market
- North America (U.S., Canada, Mexico)
- Europe (Germany, France, U.K., Italy, Spain, Rest of Europe)
- Asia-Pacific (China, Japan, India, Rest of APAC)
- South America (Brazil and the Rest of South America)
- The Middle East and Africa (UAE, South Africa, Rest of MEA)
Asia Pacific is anticipated to hold the largest share of the friedreich’s ataxia market over the predicted timeframe. Asia-Pacific is projected to be the fastest-growing region in the friedreich’s ataxia market. With around 20.74?% of the total market share, the dominance of this market is driven by rising healthcare access, increasing awareness of rare neurological diseases, expanding hospital and clinic networks, early adoption of advanced diagnostic technologies and treatments, growing public and private investment in patient care, and government initiatives promoting rare disease management. Countries such as China, India, and Japan are investing heavily in specialized care centers, genetic testing programs, and awareness campaigns, which collectively accelerate regional growth and strengthen global market contribution.
North America is expected to grow at a rapid CAGR in the friedreich’s ataxia market during the forecast period. North America is anticipated to hold the largest share of the friedreich’s ataxia market over the predicted timeframe. This region accounts for approximately 48.6?% of the total market share. The dominance of this market is driven by advanced healthcare infrastructure, widespread availability of genetic testing and pharmacological treatments, strong insurance coverage, and high awareness of rare neurological disorders among patients and caregivers. Countries such as the U.S. and Canada have well-established hospitals and clinics offering specialized FA care, supporting early diagnosis, timely intervention, and effective management. Robust government and private research funding further strengthen regional market leadership.
Europe is the 3rd largest region to grow in the friedreich’s ataxia market during the region. Europe is expected to be the second-largest region in the friedreich’s ataxia market. Accounting for approximately 32?% of the total market share, the dominance of this market is driven by well-developed healthcare systems, extensive adoption of gene therapy, physical therapy, and supportive care programs, high patient awareness of rare diseases, and government initiatives for rare neurological disorders. Countries such as Germany, France, and the U.K. provide comprehensive FA care through specialized centers and hospital networks, enabling early diagnosis, advanced treatment options, and ongoing patient management, which collectively support strong market growth.
Future Market Trends in Global Spirit Market: -
- Emergence of Disease-Modifying Therapies
The market is increasingly shifting from symptomatic management to therapies that target the root cause of friedreich’s ataxia. Pipeline drugs like vatiquinone, MIB-626, and leriglitazone are driving innovation, contributing to expanded treatment options and improved patient outcomes.
- Expansion of Gene Therapy and Pediatric Access
Gene therapy is gaining prominence, offering potential long-term solutions by addressing FXN gene mutations. Clinical trials such as the BRAVE study are broadening treatment access to children and adolescents, significantly enlarging the addressable patient population.
- Advancements in Diagnostics and AI Integration
Enhanced diagnostic tools, including 3T MRI, AI-based imaging, and faster EMG testing, are enabling earlier detection and intervention. This trend is supporting timely treatment initiation and better disease management, particularly in research-focused and emerging regions.
Recent Development
- In February 2026, Larimar Therapeutics reiterated a June 2026 submission timeline for its Biologics License Application (BLA) for Nomlabofusp (CTI?1601), a frataxin replacement therapy, reflecting the advancing focus on disease-modifying approaches in the global Friedreich’s Ataxia market.
- In October 2025, Lexeo Therapeutics advanced LX2006 gene therapy for FA-associated cardiomyopathy, reporting positive interim data and FDA Breakthrough Therapy designation, highlighting the potential of gene therapy to address underlying genetic defects.
- In August 2025, PTC Therapeutics received a Complete Response Letter (CRL) from the FDA for Vatiquinone (PTC?743), requesting additional data, indicating regulatory rigor in evaluating emerging antioxidant therapies targeting mitochondrial dysfunction.
- In June 2025, Biogen initiated the BRAVE Phase III pediatric study of Skyclarys (omaveloxolone) for patients aged 2–<16 years, expanding treatment access and underscoring the shift toward pediatric and adolescent FA care.
- In January 2025, Solid Biosciences’ SGT?212 gene therapy program received FDA Fast Track, Orphan Drug, and Rare Pediatric Disease designations, with Phase 1b dosing initiated, signaling progress in innovative frataxin-targeted interventions within the evolving FA market.
Market Segment
This study forecasts revenue at global, regional, and country levels from 2020 to 2035. Decision Advisor has segmented the friedreich’s ataxia market based on the below-mentioned segments:
Global Friedreich’s Ataxia Market, By Patient Age Group
- Pediatric
- Adolescent
- Adult
- Elderly
Global Friedreich’s Ataxia Market, By Diagnosis Method
- Genetic Testing
- Clinical Examination
- Magnetic Resonance Imaging
- Electromyography
- Neurological Assessment
Global Friedreich’s Ataxia Market, By Treatment Type
- Gene Therapy
- Pharmacological Treatments
- Physical Therapy
- Nutritional Supplements
- Supportive Care
Global Friedreich’s Ataxia Market, By End User
- Hospitals
- Clinics
- Home Care
- Research Institutes
Global Friedreich’s Ataxia Market, By Regional Analysis
- North America
- US
- Canada
- Mexico
- Europe
- Germany
- UK
- France
- Italy
- Spain
- Russia
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- Australia
- Rest of Asia Pacific
- South America
- Brazil
- Argentina
- Rest of South America
- Middle East & Africa
- UAE
- Saudi Arabia
- Qatar
- South Africa
- Rest of the Middle East & Africa
Frequently Asked Questions (FAQ)
Q. How is patient lifestyle influencing the FA treatment market?
A. Increased patient engagement in wellness programs, physiotherapy routines, and adherence to supportive care regimens is driving demand for integrated treatment plans, creating opportunities for bundled care services beyond pharmacological therapies.
Q. Are there opportunities for personalized medicine in FA management?
A. Personalized approaches leveraging patient-specific symptom profiles and genetic variability can optimize therapy selection, improve outcomes, and encourage investment in niche therapeutics and precision-supportive care solutions.
Q. How can emerging markets contribute to FA market growth?
A. Rising awareness, better access to diagnostics, and growing healthcare infrastructure in countries like India and Brazil can expand the treatable population, providing untapped demand for innovative therapies and supportive care services.
Q. What role do patient advocacy groups play in market expansion?
A. Advocacy groups raise disease awareness, facilitate early diagnosis, and promote clinical trial participation, indirectly boosting therapy adoption rates and creating demand for both pharmacological and multidisciplinary care solutions globally.
Check Licence
Choose the plan that fits you best: Single User, Multi-User, or Enterprise solutions tailored for your needs.
We Have You Covered
- 24/7 Analyst Support
- Clients Across the Globe
- Tailored Insights
- Technology Tracking
- Competitive Intelligence
- Custom Research
- Syndicated Market Studies
- Market Overview
- Market Segmentation
- Growth Drivers
- Market Opportunities
- Regulatory Insights
- Innovation & Sustainability
Report Details
| Scope | Global |
| Pages | 262 |
| Delivery | PDF & Excel via Email |
| Language | English |
| Release | Mar 2026 |
| Access | Download from this page |
