Global Hereditary Angioedema Therapeutics Market
Global Hereditary Angioedema Therapeutics Market Size, Share, By Treatment Type (C-1 Esterase Inhibitor, Kallikrein Inhibitor), By Route of Administration (Oral, Parenteral), By Age Group (Pediatric, Adult), By Distribution Channel (Hospital Pharmacies, Online Pharmacies), and By Region (North America, Europe, Asia-Pacific, Latin America, Middle East & Africa), Analysis and Forecast 2026-2035
Report Overview
Table of Contents
Market Snapshot
- Market Size (2025): USD 3.65 Billion
- Projected Market Size (2035): USD 9.89 Billion
- Compound Annual Growth Rate (CAGR): 10.48%
- Largest Regional Market: North America
- Fastest Growing Region: Asia-Pacific
- 3rd Largest Region: Europe
- Base Year: 2025
- Historical Period: 2021–2024
- Forecast Period: 2026–2035
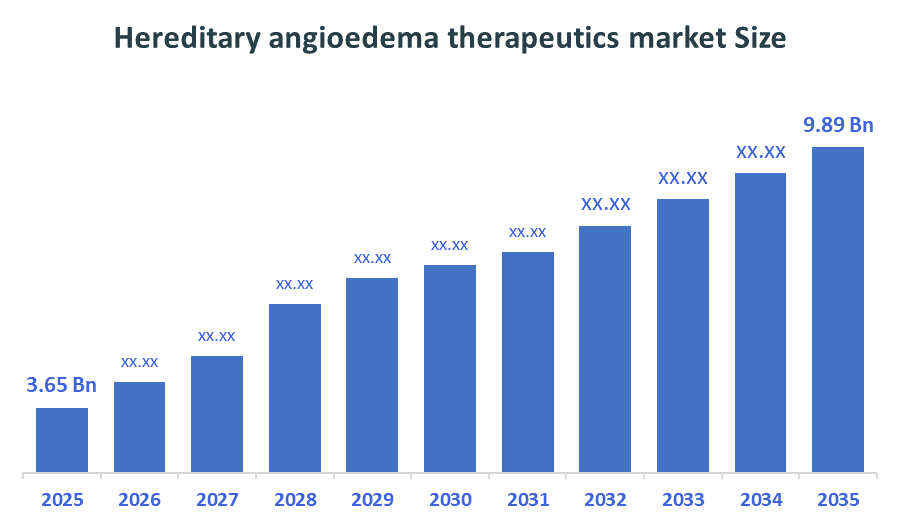
According to Decision Advisors, the Global Hereditary angioedema therapeutics market Size is expected to grow from USD 3.65 billion in 2025 to USD 9.89 billion by 2035, at a CAGR of 10.48% during the forecast period 2026-2035. The global hereditary angioedema therapeutics market is projected to grow significantly over the next decade, fueled by an increased awareness, advancing genetic testing, a strong pipeline of oral and long-acting therapies, advanced diagnostics, novel therapeutic advancements, and rising demand for prophylaxis.
Market Overview/ Introduction
The global hereditary angioedema therapeutics market refers to the trade of pharmaceuticals, which includes C1-esterase inhibitors, monoclonal antibodies, and oral kallikrein inhibitors that treat systemic swelling conditions. The system primarily develops new treatment methods to prevent and treat this uncommon genetic disorder. The hereditary angioedema therapeutics market is expanding because of three factors, which include increased disease awareness, better genetic diagnostic methods, and greater patient detection rates. The development of targeted therapies together with long-acting and oral prophylactic treatments has improved the management of diseases. Government policies that support orphan drug programs and HAEday initiatives create better treatment access while decreasing misdiagnosis rates and maintaining the ongoing need for effective therapies.
- The scheme covered 63 rare diseases, with hereditary angioedema (HAE) recognized under its framework. It provided financial support of up to ?50 lakh (approximately USD 60,000) per patient for treatment at designated Centres of Excellence. By August 2024, over 1,100 patients had benefited.
- The Orphan Drug Act (1983) provided tax credits, grants, and market exclusivity for companies developing rare disease drugs, including HAE therapies. NIH and FDA programs supported clinical trials, accelerated approvals, and expanded patient registries, which encouraged innovation in prophylactic and oral HAE therapies.
- The European Union supported the field through research and innovation framework programmes, making over €5 billion available over 25 years. From 2007 to 2020, it allocated €3.2 billion to more than 550 multinational rare disease research projects under key programmes.
Notable Insights: -
- North America is anticipated to hold the largest share of approximately 39.6% the Hereditary Angioedema Therapeutics Market over the forecast period.
- Asia Pacific is expected to grow at a rapid CAGR of approximately 12.1% in the Hereditary Angioedema Therapeutics Market during the forecast period.
- The C-1 esterase inhibitor segment dominated the market in 2025 and is anticipated to grow at a significant CAGR of approximately 7.95% during the forecast period.
- The parenteral segment dominated the market in 2025 and is anticipated to grow at a significant CAGR of approximately 9.9% during the forecast period.
- The adult segment dominated the market in 2025 and is anticipated to grow at a significant CAGR of approximately 8.3% during the forecast period.
- The hospital pharmacies segment dominated the market in 2025 and is anticipated to grow at a significant CAGR of approximately 8.6% during the forecast period.
- The compound annual growth rate of the Global Hereditary Angioedema Therapeutics Market is 10.48%.
- The market is likely to achieve a valuation of USD 9.89 billion by 2035.
What is role of technology in grooming the market?
Technology plays a crucial role in shaping the hereditary angioedema therapeutics market by enabling precise diagnosis, innovative drug development, and improved patient management. Advances in genetic testing and biomarker identification allow early and accurate detection of hereditary angioedema, leading to timely intervention. Biotechnology innovations have driven the development of targeted therapies such as C1 esterase inhibitors, monoclonal antibodies, and gene-based treatments, significantly improving efficacy and safety. Digital health tools, including telemedicine and mobile monitoring apps, enhance patient adherence and real-time symptom tracking. Additionally, advanced manufacturing technologies ensure better drug quality and scalability. Continuous R&D supported by AI and data analytics accelerates clinical trials and personalized treatment approaches, ultimately boosting market growth.
Market Drivers
The global hereditary angioedema therapeutics market is driven by increasing awareness and improved genetic screening for mutations such as SERPING1 and F12, resulting in higher diagnosis rates and a larger treated patient population. Advancements in targeted therapies, including long-acting subcutaneous and oral prophylactic options like kallikrein inhibitors, are significantly improving disease management. There is a notable shift from treating acute attacks to preventive care, which drives consistent, long-term demand for therapies. Additionally, initiatives like HAEday are enhancing physician education, reducing misdiagnosis rates, and promoting early diagnosis and timely adoption of effective treatments globally.
Restrain
The global hereditary angioedema therapeutics market faces restraints due to the high cost of advanced biologic treatments, limiting accessibility in low- and middle-income regions. Limited disease awareness and underdiagnosis further hinder demand. Additionally, stringent regulatory requirements and complex manufacturing processes delay product approvals and market entry, restricting overall growth.
Competitive Analysis:
The report offers the appropriate analysis of the key organizations/companies involved within the global hereditary angioedema therapeutics market, along with a comparative evaluation primarily based on their product of offering, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborative analysis focusing on the current news and developments of the companies, which includes product development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
Top Companies in Global Hereditary Angioedema Therapeutics Market
- Takeda Pharmaceutical Company Limited
- CSL Behring
- Pharming Group N.V.
- BioCryst Pharmaceuticals, Inc.
- KalVista Pharmaceuticals, Inc.
- Ionis Pharmaceuticals, Inc.
- Sanofi S.A.
- Shire plc
- Attune Pharmaceuticals, Inc.
- Adverum Biotechnologies, Inc.
- Arrowhead Pharmaceuticals, Inc.
- Octapharma AG
Government Initiatives
|
Country |
Key Government Initiatives |
|
US |
The FDA had approved CSL Behring’s Andembry (garadacimab-gxii) as a prophylactic therapy for hereditary angioedema (HAE) in patients aged 12 and older. This once-monthly subcutaneous injection targeting factor XIIa has demonstrated strong efficacy and safety in the pivotal VANGUARD trial, reducing attack frequency by nearly 90% compared to placebo and marking a milestone advancement in HAE therapeutics. |
|
China |
The National Medical Products Administration officially integrated Breakthrough Therapy Designation into its 2026 workflow, aiming to accelerate approvals. This enabled faster entry of oral hereditary angioedema treatments like sebetralstat into the Chinese market, improving patient access and innovation. |
|
India |
The government has enhanced the official digital portal for rare diseases, which now more effectively links HAE patients with corporate social responsibility (CSR) funds to cover the lifelong cost of prophylaxis. |
Study on the Supply, Demand, Distribution, and Market Environment of Hereditary Angioedema Therapeutics Market
The global hereditary angioedema therapeutics market is shaped by a complex interplay of supply, demand, distribution, and market environment factors. Demand is driven by rising diagnosis rates, improved awareness of rare diseases, and the need for long-term prophylactic treatments. Supply depends on advanced biopharmaceutical manufacturing, particularly for plasma-derived and recombinant C1 esterase inhibitors. Key raw materials include human plasma, recombinant proteins, enzymes, and specialized excipients used in biologics production. Distribution channels involve hospital pharmacies, specialty clinics, and online pharmacies, ensuring access to high-cost therapies. The market environment is influenced by regulatory frameworks, orphan drug incentives, and reimbursement policies. Additionally, increasing R&D investments and technological advancements continue to enhance production efficiency and global market accessibility.
Price Analysis and Consumer Behaviour Analysis
The price analysis of the global hereditary angioedema (HAE) therapeutics market reveals extremely high treatment costs, primarily due to the use of advanced biologics and orphan drugs. Individual drug prices can range from around approximately 5,000 to over 12,000 per attack, while some therapies cost approximately 400,000 annually per patient, with drugs accounting for nearly 97% of total treatment expenses. Additionally, newer therapies may cost tens of thousands per dose, further increasing the financial burden. From a consumer behaviour perspective, patients and healthcare providers prioritize efficacy, rapid symptom relief, and long-term prevention over cost due to the life-threatening nature of the disease. There is a strong shift toward prophylactic therapies that reduce attack frequency and improve quality of life, despite higher upfront costs. Insurance coverage, reimbursement policies, and patient assistance programs heavily influence purchasing decisions, while adherence is driven by convenience, such as self-administration and reduced dosing frequency.
Market Segmentation
The hereditary angioedema therapeutics market share is classified into treatment type, route of administration, age group, and distribution channel
- The C-1 esterase inhibitor segment dominated the market in 2025 and is anticipated to grow at a significant CAGR of approximately 7.95% during the forecast period.
Based on the treatment type, the hereditary angioedema therapeutics market is divided into C-1 esterase inhibitor and kallikrein inhibitor. Among these, the C-1 esterase inhibitor segment dominated the market in 2025 and is anticipated to grow at a significant CAGR of approximately 7.95% during the forecast period. Due to its proven efficacy in both acute and prophylactic treatment of hereditary angioedema. Strong clinical adoption, established safety profile, and availability of advanced formulations have supported its continued demand and steady growth during the forecast period.
- The parenteral segment dominated the market in 2025 and is anticipated to grow at a significant CAGR of approximately 9.9% during the forecast period.
Based on the route of administration, the hereditary angioedema therapeutics market is divided into oral and parenteral. Among these, the parenteral segment dominated the market in 2025 and is anticipated to grow at a significant CAGR of approximately 9.9% during the forecast period. Due to its rapid onset of action and high effectiveness in managing acute hereditary angioedema attacks. Strong clinical preference, availability of injectable biologics, and reliability in emergency treatment settings supported its continued growth during the forecast period.
- The adult segment dominated the market in 2025 and is anticipated to grow at a significant CAGR of approximately 8.3% during the forecast period.
Based on the age group, the hereditary angioedema therapeutics market is divided into pediatric and adult. Among these, the adult segment dominated the market in 2025 and is anticipated to grow at a significant CAGR of approximately 8.3% during the forecast period. The growth of the oral segment is growing due to higher diagnosis rates, greater disease awareness, and increased treatment uptake among adults. Availability of advanced prophylactic therapies and higher healthcare access further contributed to sustained demand and significant growth during the forecast period.
- The hospital pharmacies segment dominated the market in 2025 and is anticipated to grow at a significant CAGR of approximately 8.6% during the forecast period.
Based on the distribution channel, the hereditary angioedema therapeutics market is divided into hospital pharmacies and online pharmacies. Among these, the hospital pharmacies segment dominated the market in 2025 and is anticipated to grow at a significant CAGR of approximately 8.6% during the forecast period. The growth of the hospitals & clinics segment is growing due to the availability of specialized treatments, better storage and handling of biologics, and immediate access during emergencies. Strong physician preference and integrated care settings further supported higher dispensing rates and sustained growth.
Strategies to Implement for Growth of the Market in Non-Leading Regions
To drive growth of the global hereditary angioedema (HAE) therapeutics market in non-leading regions, companies should focus on increasing disease awareness and improving early diagnosis through education campaigns and screening programs. Expanding healthcare infrastructure, including specialty clinics and rare disease centers, is essential to improve treatment access. Enhancing affordability through biosimilars, patient assistance programs, and better reimbursement policies can boost adoption. Strengthening distribution networks with local partnerships ensures wider availability of therapies. Additionally, engaging with regulatory bodies for faster approvals and leveraging orphan drug incentives can support market entry. The introduction of convenient treatment options, such as oral and self-administered therapies, further enhances patient compliance and acceptance, driving overall market expansion.
Regional Segment Analysis of the Hereditary Angioedema Therapeutics Market
- North America (U.S., Canada, Mexico)
- Europe (Germany, France, U.K., Italy, Spain, Rest of Europe)
- Asia-Pacific (China, Japan, India, Rest of APAC)
- South America (Brazil and the Rest of South America)
- The Middle East and Africa (UAE, South Africa, Rest of MEA)
North America is anticipated to hold the largest share of approximately 39.6% the hereditary angioedema therapeutics market over the forecast period.
North America is anticipated to hold the largest share of approximately 39.6% the hereditary angioedema therapeutics market over the forecast period. The dominance of this region is primarily driven by advanced healthcare infrastructure, high diagnosis rates, strong reimbursement systems, the presence of leading biopharma companies, and the rapid adoption of innovative therapies.
Asia Pacific is expected to grow at a rapid CAGR of approximately 12.1% in the hereditary angioedema therapeutics market during the forecast period.
Asia Pacific is expected to grow at a rapid CAGR of approximately 12.1% in the hereditary angioedema therapeutics market during the forecast period. This growth is driven by improving healthcare infrastructure, rising awareness and diagnosis of rare diseases, increasing healthcare expenditure, and expanding access to advanced therapies through global collaborations.
Europe is the 3rd largest region to grow in the hereditary angioedema therapeutics market during the period.
Europe was the third largest region to grow in the hereditary angioedema (HAE) therapeutics market during the period due to strong regulatory frameworks, increasing clinical research investments, rising awareness of rare genetic disorders, and expanding access to advanced therapies supported by favorable reimbursement policies and specialized treatment centers.
Future Market Trends in Global Hereditary Angioedema Therapeutics Market: -
1. Shift toward Prophylactic Therapies
The shift toward prophylactic therapies was anticipated as a future trend in the global hereditary angioedema (HAE) therapeutics market because preventive treatments reduce attack frequency, improve patient quality of life, ensure consistent disease management, and create stable revenue streams through long-term therapy adoption and compliance.
2. Expansion of Subcutaneous and Oral Delivery Routes
The expansion of subcutaneous and oral delivery routes was anticipated as a future trend in the global hereditary angioedema (HAE) therapeutics market because these methods improve patient convenience, reduce hospital dependency, enhance compliance, and support home-based care, reflecting a broader shift toward patient-centric treatment approaches globally.
3. Growing Awareness and Diagnostics for Rare Diseases
Growing awareness and improved diagnostics for rare diseases were anticipated as a future trend in the global hereditary angioedema (HAE) therapeutics market because enhanced genetic testing, government initiatives, patient registries, and advocacy programs increased diagnosis rates, expanded treatment access, and supported early intervention for better patient outcomes globally.
Recent Development
In December 2025, as new hereditary angioedema (HAE) drugs had flooded the market, questions arose about whether uptake had matched innovation. The influx of advanced therapies expanded treatment options, improved patient outcomes, and strengthened competition, positively shaping the global HAE therapeutics landscape with broader accessibility and innovation-driven growth.
In October 2025, BioCryst Pharmaceuticals announced the acquisition of Astria Therapeutics in a deal worth about $700 million, which strengthened its rare disease and hereditary angioedema (HAE) pipeline. This strategic move expanded BioCryst’s portfolio with innovative long-acting therapies, enhancing its competitive position in the global HAE therapeutics market.
In June 2025, Companies had increasingly formed regional commercialization partnerships, with KalVista collaborating with partners to expand its market presence in Asia. These alliances strengthened distribution networks, improved patient access to hereditary angioedema (HAE) therapies, and supported growth in emerging markets, positively impacting the global HAE therapeutics landscape.
In April 2025, Strategic collaborations between biotech firms and research organizations had accelerated clinical trials and pipeline development for novel kallikrein inhibitors and monoclonal antibodies. These partnerships strengthened innovation, advanced rare disease research, and expanded therapeutic options, positively impacting the global hereditary angioedema (HAE) therapeutics market.
How is Recent Developments Helping the Market?
Recent developments have significantly strengthened the global hereditary angioedema (HAE) therapeutics market by enhancing treatment options and accessibility. The introduction of novel prophylactic therapies, such as kallikrein inhibitors, has improved long-term disease management and reduced attack frequency. Advances in subcutaneous and oral delivery routes have increased patient convenience, compliance, and reduced reliance on hospital-based care. Growing awareness campaigns, rare disease registries, and improved genetic diagnostics have expanded patient identification, particularly in emerging markets. Additionally, strategic collaborations between biopharma companies and healthcare systems have accelerated innovation, regulatory approvals, and global distribution. Collectively, these developments have improved patient outcomes, broadened therapeutic adoption, and positioned the market for sustained growth across regions.
Market Segment
This study forecasts revenue at global, regional, and country levels from 2020 to 2035. Decision Advisors has segmented the hereditary angioedema therapeutics market based on the below-mentioned segments:
Global Hereditary Angioedema Therapeutics Market, By Treatment Type
- Hormonal Therapy
- Chemotherapy
- Immunotherapy
- Targeted Therapy
- Antimicrobial Therapy
- Others
Global Hereditary angioedema therapeutics market, By Route of Administration
- Oral
- Parenteral
Global Hereditary angioedema therapeutics market, By Distribution Channel
- Hospital Pharmacies
- Online Pharmacies
Global Hereditary angioedema therapeutics market, By Regional Analysis
- North America
- US
- Canada
- Mexico
- Europe
- Germany
- UK
- France
- Italy
- Spain
- Russia
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- Australia
- Rest of Asia Pacific
- South America
- Brazil
- Argentina
- Rest of South America
- Middle East & Africa
- UAE
- Saudi Arabia
- Qatar
- South Africa
- Rest of the Middle East & Africa
Frequently Asked Questions (FAQ)
Q How are emerging biologics transforming treatment outcomes in hereditary angioedema?
A. Emerging biologics are significantly improving HAE treatment outcomes by targeting specific pathways such as kallikrein inhibition and bradykinin production. These therapies offer faster symptom control, reduced attack frequency, and improved safety profiles. Long-acting biologics also minimize dosing frequency, enhancing patient adherence and overall quality of life.
Q. What role do patient assistance programs play in expanding market access?
A. Patient assistance programs play a critical role in improving access to expensive HAE therapies, especially in low- and middle-income regions. These programs help reduce financial burdens through subsidies, free drug access, or insurance support, thereby increasing treatment adoption and improving long-term disease management.
Q. How is the competitive landscape evolving in the HAE therapeutics market?
A. The competitive landscape is evolving with increased participation from biotech firms focusing on rare diseases. Companies are investing in innovative pipelines, including gene therapies and oral prophylactics. Strategic collaborations, acquisitions, and regional partnerships are intensifying competition and accelerating global market expansion.
Q. What impact does delayed diagnosis have on the HAE therapeutics market?
A. Delayed diagnosis negatively impacts the market by limiting the number of treated patients and increasing the risk of severe complications. It also leads to higher emergency care costs and poor patient outcomes. Improving diagnostic awareness and access to genetic testing is essential to unlocking unmet market potential.
- Introduction
- Objectives of the Study
- Market Definition
- Research Scope
- Research Methodology and Assumptions
- Executive Summary
- Premium Insights
- Porter’s Five Forces Analysis
- Value Chain Analysis
- Top Investment Pockets
- Market Attractiveness Analysis By Treatment Type
- Market Attractiveness Analysis By Route of Administration
- Market Attractiveness Analysis By Age Group
- Market Attractiveness Analysis By Distribution Channel
- Market Attractiveness Analysis By Region
- Industry Trends
- Market Dynamics
- Market Evaluation
- Drivers
- Increasing awareness and improved genetic screening for mutations
- Restraints
- High cost of advanced biologic treatments
- Opportunities
- Increased investment in R&D activities by manufacturers
- Challenges
- Limiting accessibility in low and middle income regions
- Global Hereditary Angioedema Therapeutics Market Analysis and Projection, By Treatment Type
- Segment Overview
- C-1 Esterase Inhibitor
- Kallikrein Inhibitor
- Global Hereditary Angioedema Therapeutics Market Analysis and Projection, By Route of Administration
- Segment Overview
- Oral
- Parenteral
- Global Hereditary Angioedema Therapeutics Market Analysis and Projection, By Age Group
- Segment Overview
- Pediatric
- Adult
- Global Hereditary Angioedema Therapeutics Market Analysis and Projection, By Distribution Channel
- Segment Overview
- Hospital Pharmacies
- Online Pharmacies
- MarketGlobal Hereditary Angioedema Therapeutics Market Analysis and Projection, By Regional Analysis
- Segment Overview
- North America
- U.S.
- Canada
- Mexico
- Europe
- Germany
- France
- U.K.
- Italy
- Spain
- Asia-Pacific
- Japan
- China
- India
- South America
- Brazil
- Middle East and Africa
- UAE
- South Africa
- Global Hereditary Angioedema Therapeutics Market-Competitive Landscape
- Overview
- Market Share of Key Players in the Hereditary Angioedema Therapeutics Market
- Global Company Market Share
- North America Company Market Share
- Europe Company Market Share
- APAC Company Market Share
- Competitive Situations and Trends
- Coverage Launches and Developments
- Partnerships, Collaborations, and Agreements
- Mergers & Acquisitions
- Expansions
- Company Profiles
- Takeda Pharmaceutical Company Limited
- Business Overview
- Company Snapshot
- Company Market Share Analysis
- Company Coverage Portfolio
- Recent Developments
- SWOT Analysis
- CSL Behring
- Business Overview
- Company Snapshot
- Company Market Share Analysis
- Company Coverage Portfolio
- Recent Developments
- SWOT Analysis
- Pharming Group N.V.
- Business Overview
- Company Snapshot
- Company Market Share Analysis
- Company Coverage Portfolio
- Recent Developments
- SWOT Analysis
- BioCryst Pharmaceuticals, Inc.
- Business Overview
- Company Snapshot
- Company Market Share Analysis
- Company Coverage Portfolio
- Recent Developments
- SWOT Analysis
- KalVista Pharmaceuticals, Inc.
- Business Overview
- Company Snapshot
- Company Market Share Analysis
- Company Coverage Portfolio
- Recent Developments
- SWOT Analysis
- Ionis Pharmaceuticals, Inc.
- Business Overview
- Company Snapshot
- Company Market Share Analysis
- Company Coverage Portfolio
- Recent Developments
- SWOT Analysis
- Sanofi S.A.
- Business Overview
- Company Snapshot
- Company Market Share Analysis
- Company Coverage Portfolio
- Recent Developments
- SWOT Analysis
- Shire plc
- Business Overview
- Company Snapshot
- Company Market Share Analysis
- Company Coverage Portfolio
- Recent Developments
- SWOT Analysis
- Attune Pharmaceuticals, Inc.
- Business Overview
- Company Snapshot
- Company Market Share Analysis
- Company Coverage Portfolio
- Recent Developments
- SWOT Analysis
- Adverum Biotechnologies, Inc.
- Business Overview
- Company Snapshot
- Company Market Share Analysis
- Company Coverage Portfolio
- Recent Developments
- SWOT Analysis
- Arrowhead Pharmaceuticals, Inc.
- Business Overview
- Company Snapshot
- Company Market Share Analysis
- Company Coverage Portfolio
- Recent Developments
- SWOT Analysis
- Octapharma AG
- Business Overview
- Company Snapshot
- Company Market Share Analysis
- Company Coverage Portfolio
- Recent Developments
- SWOT Analysis
- Takeda Pharmaceutical Company Limited
List of Table
- Global Hereditary Angioedema Therapeutics Market, By Treatment Type, 2024-2035(USD Billion)
- Global C-1 Esterase Inhibitor, Hereditary Angioedema Therapeutics Market, By Region, 2024-2035(USD Billion)
- Global Kallikrein Inhibitor, Hereditary Angioedema Therapeutics Market, By Region, 2024-2035(USD Billion)
- Global Hereditary Angioedema Therapeutics Market, By Route of Administration, 2024-2035(USD Billion)
- Global Oral, Hereditary Angioedema Therapeutics Market, By Region, 2024-2035(USD Billion)
- Global Parenteral, Hereditary Angioedema Therapeutics Market, By Region, 2024-2035(USD Billion)
- Global Hereditary Angioedema Therapeutics Market, By Age Group, 2024-2035(USD Billion)
- Global Pediatric, Hereditary Angioedema Therapeutics Market, By Region, 2024-2035(USD Billion)
- Global Adult, Hereditary Angioedema Therapeutics Market, By Region, 2024-2035(USD Billion)
- Global Hereditary Angioedema Therapeutics Market, By Distribution Channel, 2024-2035(USD Billion)
- Global Hospital Pharmacies, Hereditary Angioedema Therapeutics Market, By Region, 2024-2035(USD Billion)
- Global Online Pharmacies, Hereditary Angioedema Therapeutics Market, By Region, 2024-2035(USD Billion)
- North America Hereditary Angioedema Therapeutics Market, By Treatment Type, 2024-2035(USD Billion)
- North America Hereditary Angioedema Therapeutics Market, By Route of Administration, 2024-2035(USD Billion)
- North America Hereditary Angioedema Therapeutics Market, By Age Group, 2024-2035(USD Billion)
- North America Hereditary Angioedema Therapeutics Market, By Distribution Channel, 2024-2035(USD Billion)
- U.S. Hereditary Angioedema Therapeutics Market, By Treatment Type, 2024-2035(USD Billion)
- U.S. Hereditary Angioedema Therapeutics Market, By Route of Administration, 2024-2035(USD Billion)
- U.S. Hereditary Angioedema Therapeutics Market, By Age Group, 2024-2035(USD Billion)
- U.S. Hereditary Angioedema Therapeutics Market, By Distribution Channel, 2024-2035(USD Billion)
- Canada Hereditary Angioedema Therapeutics Market, By Treatment Type, 2024-2035(USD Billion)
- Canada Hereditary Angioedema Therapeutics Market, By Route of Administration, 2024-2035(USD Billion)
- Canada Hereditary Angioedema Therapeutics Market, By Age Group, 2024-2035(USD Billion)
- Canada Hereditary Angioedema Therapeutics Market, By Distribution Channel, 2024-2035(USD Billion)
- Mexico Hereditary Angioedema Therapeutics Market, By Treatment Type, 2024-2035(USD Billion)
- Mexico Hereditary Angioedema Therapeutics Market, By Route of Administration, 2024-2035(USD Billion)
- Mexico Hereditary Angioedema Therapeutics Market, By Age Group, 2024-2035(USD Billion)
- Mexico Hereditary Angioedema Therapeutics Market, By Distribution Channel, 2024-2035(USD Billion)
- Europe Hereditary Angioedema Therapeutics Market, By Treatment Type, 2024-2035(USD Billion)
- Europe Hereditary Angioedema Therapeutics Market, By Route of Administration, 2024-2035(USD Billion)
- Europe Hereditary Angioedema Therapeutics Market, By Age Group, 2024-2035(USD Billion)
- Europe Hereditary Angioedema Therapeutics Market, By Distribution Channel, 2024-2035(USD Billion)
- Germany Hereditary Angioedema Therapeutics Market, By Treatment Type, 2024-2035(USD Billion)
- Germany Hereditary Angioedema Therapeutics Market, By Route of Administration, 2024-2035(USD Billion)
- Germany Hereditary Angioedema Therapeutics Market, By Age Group, 2024-2035(USD Billion)
- Germany Hereditary Angioedema Therapeutics Market, By Distribution Channel, 2024-2035(USD Billion)
- France Hereditary Angioedema Therapeutics Market, By Treatment Type, 2024-2035(USD Billion)
- France Hereditary Angioedema Therapeutics Market, By Route of Administration, 2024-2035(USD Billion)
- France Hereditary Angioedema Therapeutics Market, By Age Group, 2024-2035(USD Billion)
- France Hereditary Angioedema Therapeutics Market, By Distribution Channel, 2024-2035(USD Billion)
- U.K. Hereditary Angioedema Therapeutics Market, By Treatment Type, 2024-2035(USD Billion)
- U.K. Hereditary Angioedema Therapeutics Market, By Route of Administration, 2024-2035(USD Billion)
- U.K. Hereditary Angioedema Therapeutics Market, By Age Group, 2024-2035(USD Billion)
- U.K. Hereditary Angioedema Therapeutics Market, By Distribution Channel, 2024-2035(USD Billion)
- Italy Hereditary Angioedema Therapeutics Market, By Treatment Type, 2024-2035(USD Billion)
- Italy Hereditary Angioedema Therapeutics Market, By Route of Administration, 2024-2035(USD Billion)
- Italy Hereditary Angioedema Therapeutics Market, By Age Group, 2024-2035(USD Billion)
- Italy Hereditary Angioedema Therapeutics Market, By Distribution Channel, 2024-2035(USD Billion)
- Spain Hereditary Angioedema Therapeutics Market, By Treatment Type, 2024-2035(USD Billion)
- Spain Hereditary Angioedema Therapeutics Market, By Route of Administration, 2024-2035(USD Billion)
- Spain Hereditary Angioedema Therapeutics Market, By Age Group, 2024-2035(USD Billion)
- Spain Hereditary Angioedema Therapeutics Market, By Distribution Channel, 2024-2035(USD Billion)
- Asia Pacific Hereditary Angioedema Therapeutics Market, By Treatment Type, 2024-2035(USD Billion)
- Asia Pacific Hereditary Angioedema Therapeutics Market, By Route of Administration, 2024-2035(USD Billion)
- Asia Pacific Hereditary Angioedema Therapeutics Market, By Age Group, 2024-2035(USD Billion)
- Asia Pacific Hereditary Angioedema Therapeutics Market, By Distribution Channel, 2024-2035(USD Billion)
- Japan Hereditary Angioedema Therapeutics Market, By Treatment Type, 2024-2035(USD Billion)
- Japan Hereditary Angioedema Therapeutics Market, By Route of Administration, 2024-2035(USD Billion)
- Japan Hereditary Angioedema Therapeutics Market, By Age Group, 2024-2035(USD Billion)
- Japan Hereditary Angioedema Therapeutics Market, By Distribution Channel, 2024-2035(USD Billion)
- China Hereditary Angioedema Therapeutics Market, By Treatment Type, 2024-2035(USD Billion)
- China Hereditary Angioedema Therapeutics Market, By Route of Administration, 2024-2035(USD Billion)
- China Hereditary Angioedema Therapeutics Market, By Age Group, 2024-2035(USD Billion)
- China Hereditary Angioedema Therapeutics Market, By Distribution Channel, 2024-2035(USD Billion)
- India Hereditary Angioedema Therapeutics Market, By Treatment Type, 2024-2035(USD Billion)
- India Hereditary Angioedema Therapeutics Market, By Route of Administration, 2024-2035(USD Billion)
- India Hereditary Angioedema Therapeutics Market, By Age Group, 2024-2035(USD Billion)
- India Hereditary Angioedema Therapeutics Market, By Distribution Channel, 2024-2035(USD Billion)
- South America Hereditary Angioedema Therapeutics Market, By Treatment Type, 2024-2035(USD Billion)
- South America Hereditary Angioedema Therapeutics Market, By Route of Administration, 2024-2035(USD Billion)
- South America Hereditary Angioedema Therapeutics Market, By Age Group, 2024-2035(USD Billion)
- South America Hereditary Angioedema Therapeutics Market, By Distribution Channel, 2024-2035(USD Billion)
- Brazil Hereditary Angioedema Therapeutics Market, By Treatment Type, 2024-2035(USD Billion)
- Brazil Hereditary Angioedema Therapeutics Market, By Route of Administration, 2024-2035(USD Billion)
- Brazil Hereditary Angioedema Therapeutics Market, By Age Group, 2024-2035(USD Billion)
- Brazil Hereditary Angioedema Therapeutics Market, By Distribution Channel, 2024-2035(USD Billion)
- The Middle East and Africa Hereditary Angioedema Therapeutics Market, By Treatment Type, 2024-2035(USD Billion)
- The Middle East and Africa Hereditary Angioedema Therapeutics Market, By Route of Administration, 2024-2035(USD Billion)
- The Middle East and Africa Hereditary Angioedema Therapeutics Market, By Age Group, 2024-2035(USD Billion)
- The Middle East and Africa Hereditary Angioedema Therapeutics Market, By Distribution Channel, 2024-2035(USD Billion)
- UAE Hereditary Angioedema Therapeutics Market, By Treatment Type, 2024-2035(USD Billion)
- UAE Hereditary Angioedema Therapeutics Market, By Route of Administration, 2024-2035(USD Billion)
- UAE Hereditary Angioedema Therapeutics Market, By Age Group, 2024-2035(USD Billion)
- UAE Hereditary Angioedema Therapeutics Market, By Distribution Channel, 2024-2035(USD Billion)
- South Africa Hereditary Angioedema Therapeutics Market, By Treatment Type, 2024-2035(USD Billion)
- South Africa Hereditary Angioedema Therapeutics Market, By Route of Administration, 2024-2035(USD Billion)
- South Africa Hereditary Angioedema Therapeutics Market, By Age Group, 2024-2035(USD Billion)
- South Africa Hereditary Angioedema Therapeutics Market, By Distribution Channel, 2024-2035(USD Billion)
List of Figures
- Global Hereditary Angioedema Therapeutics Market Segmentation
- Hereditary Angioedema Therapeutics Market: Research Methodology
- Market Size Estimation Methodology: Bottom-Up Approach
- Market Size Estimation Methodology: Top-down Approach
- Data Triangulation
- Porter’s Five Forces Analysis
- Value Chain Analysis
- Top investment pocket in the Hereditary Angioedema Therapeutics Market
- Top Winning Strategies, 2024-2035
- Top Winning Strategies, By Development, 2024-2035(%)
- Top Winning Strategies, By Company, 2024-2035
- Moderate Bargaining power of Buyers
- Moderate Bargaining power of Suppliers
- Moderate Bargaining power of New Entrants
- Low threat of Substitution
- High Competitive Rivalry
- Top Player Positioning, 2024
- Market Share Analysis, 2024
- Restraint and Drivers: Hereditary Angioedema Therapeutics Market
- Hereditary Angioedema Therapeutics Market Segmentation, By Treatment Type
- Hereditary Angioedema Therapeutics Market For C-1 Esterase Inhibitor, By Region, 2024-2035 ($ Billion)
- Hereditary Angioedema Therapeutics Market For Kallikrein Inhibitor, By Region, 2024-2035 ($ Billion)
- Hereditary Angioedema Therapeutics Market For <<Sub1_se3>>, By Region, 2024-2035 ($ Billion)
- Hereditary Angioedema Therapeutics Market For <<Sub1_se4>>, By Region, 2024-2035 ($ Billion)
- Hereditary Angioedema Therapeutics Market For <<Sub1_se5>>, By Region, 2024-2035 ($ Billion)
- Hereditary Angioedema Therapeutics Market Segmentation, By Route of Administration
- Hereditary Angioedema Therapeutics Market For Oral, By Region, 2024-2035 ($ Billion)
- Hereditary Angioedema Therapeutics Market For Parenteral, By Region, 2024-2035 ($ Billion)
- Hereditary Angioedema Therapeutics Market For <<Sub2_se3>>, By Region, 2024-2035 ($ Billion)
- Hereditary Angioedema Therapeutics Market For <<Sub2_se4>>, By Region, 2024-2035 ($ Billion)
- Hereditary Angioedema Therapeutics Market For <<Sub2_se5>>, By Region, 2024-2035 ($ Billion)
- Hereditary Angioedema Therapeutics Market Segmentation, By Age Group
- Hereditary Angioedema Therapeutics Market For Pediatric, By Region, 2024-2035 ($ Billion)
- Hereditary Angioedema Therapeutics Market For Adult, By Region, 2024-2035 ($ Billion)
- Hereditary Angioedema Therapeutics Market For <<Sub3_se3>>, By Region, 2024-2035 ($ Billion)
- Hereditary Angioedema Therapeutics Market For <<Sub3_se4>>, By Region, 2024-2035 ($ Billion)
- Hereditary Angioedema Therapeutics Market For <<Sub3_se5>>, By Region, 2024-2035 ($ Billion)
- Hereditary Angioedema Therapeutics Market Segmentation, By Distribution Channel
- Hereditary Angioedema Therapeutics Market For Hospital Pharmacies, By Region, 2024-2035 ($ Billion)
- Hereditary Angioedema Therapeutics Market For Online Pharmacies, By Region, 2024-2035 ($ Billion)
- Hereditary Angioedema Therapeutics Market For <<Sub4_se3>>, By Region, 2024-2035 ($ Billion)
- Hereditary Angioedema Therapeutics Market For <<Sub4_se4>>, By Region, 2024-2035 ($ Billion)
- Hereditary Angioedema Therapeutics Market For <<Sub4_se5>>, By Region, 2024-2035 ($ Billion)
- Hereditary Angioedema Therapeutics Market Segmentation, By <<Seg5>>
- Hereditary Angioedema Therapeutics Market For <<Sub5_se1>>, By Region, 2024-2035 ($ Billion)
- Hereditary Angioedema Therapeutics Market For <<Sub5_se2>>, By Region, 2024-2035 ($ Billion)
- Hereditary Angioedema Therapeutics Market For <<Sub5_se3>>, By Region, 2024-2035 ($ Billion)
- Hereditary Angioedema Therapeutics Market For <<Sub5_se4>>, By Region, 2024-2035 ($ Billion)
- Hereditary Angioedema Therapeutics Market For <<Sub5_se5>>, By Region, 2024-2035 ($ Billion)
- Takeda Pharmaceutical Company Limited: Net Sales, 2024-2035 ($ Billion)
- Takeda Pharmaceutical Company Limited: Revenue Share, By Segment, 2024 (%)
- Takeda Pharmaceutical Company Limited: Revenue Share, By Region, 2024 (%)
- CSL Behring: Net Sales, 2024-2035 ($ Billion)
- CSL Behring: Revenue Share, By Segment, 2024 (%)
- CSL Behring: Revenue Share, By Region, 2024 (%)
- Pharming Group N.V.: Net Sales, 2024-2035 ($ Billion)
- Pharming Group N.V.: Revenue Share, By Segment, 2024 (%)
- Pharming Group N.V.: Revenue Share, By Region, 2024 (%)
- BioCryst Pharmaceuticals, Inc.: Net Sales, 2024-2035 ($ Billion)
- BioCryst Pharmaceuticals, Inc.: Revenue Share, By Segment, 2024 (%)
- BioCryst Pharmaceuticals, Inc.: Revenue Share, By Region, 2024 (%)
- KalVista Pharmaceuticals, Inc.: Net Sales, 2024-2035 ($ Billion)
- KalVista Pharmaceuticals, Inc.: Revenue Share, By Segment, 2024 (%)
- KalVista Pharmaceuticals, Inc.: Revenue Share, By Region, 2024 (%)
- Ionis Pharmaceuticals, Inc.: Net Sales, 2024-2035 ($ Billion)
- Ionis Pharmaceuticals, Inc.: Revenue Share, By Segment, 2024 (%)
- Ionis Pharmaceuticals, Inc.: Revenue Share, By Region, 2024 (%)
- Sanofi S.A.: Net Sales, 2024-2035 ($ Billion)
- Sanofi S.A.: Revenue Share, By Segment, 2024 (%)
- Sanofi S.A.: Revenue Share, By Region, 2024 (%)
- Shire plc: Net Sales, 2024-2035 ($ Billion)
- Shire plc: Revenue Share, By Segment, 2024 (%)
- Shire plc: Revenue Share, By Region, 2024 (%)
- Attune Pharmaceuticals, Inc.: Net Sales, 2024-2035 ($ Billion)
- Attune Pharmaceuticals, Inc.: Revenue Share, By Segment, 2024 (%)
- Attune Pharmaceuticals, Inc.: Revenue Share, By Region, 2024 (%)
- Adverum Biotechnologies, Inc.: Net Sales, 2024-2035 ($ Billion)
- Adverum Biotechnologies, Inc.: Revenue Share, By Segment, 2024 (%)
- Adverum Biotechnologies, Inc.: Revenue Share, By Region, 2024 (%)
- Arrowhead Pharmaceuticals, Inc.: Net Sales, 2024-2035 ($ Billion)
- Arrowhead Pharmaceuticals, Inc.: Revenue Share, By Segment, 2024 (%)
- Arrowhead Pharmaceuticals, Inc.: Revenue Share, By Region, 2024 (%)
- Octapharma AG: Net Sales, 2024-2035 ($ Billion)
- Octapharma AG: Revenue Share, By Segment, 2024 (%)
- Octapharma AG: Revenue Share, By Region, 2024 (%)
Check Licence
Choose the plan that fits you best: Single User, Multi-User, or Enterprise solutions tailored for your needs.
We Have You Covered
- 24/7 Analyst Support
- Clients Across the Globe
- Tailored Insights
- Technology Tracking
- Competitive Intelligence
- Custom Research
- Syndicated Market Studies
- Market Overview
- Market Segmentation
- Growth Drivers
- Market Opportunities
- Regulatory Insights
- Innovation & Sustainability
Report Details
| Scope | Global |
| Pages | 245 |
| Delivery | PDF & Excel via Email |
| Language | English |
| Release | Apr 2026 |
| Access | Download from this page |
