Global Humanized Antibody Market
Global Humanized Antibody Market Size, Share, By Type (Monoclonal Antibodies, Bispecific Antibodies, Antibody-Drug Conjugates), By Application (Oncology, Autoimmune Diseases, Infectious Diseases, Others), By End User (Hospitals, Research Institutes, Specialty Clinics), and By Region (North America, Europe, Asia-Pacific, Latin America, Middle East & Africa), Analysis and Forecast 2025?2035.
Report Overview
Table of Contents
Market Snapshot
- Market Size (2025): USD 39.3 Billion
- Projected Market Size (2035): USD 108.1 Billion
- Compound Annual Growth Rate (CAGR): 10.65%
- Largest Regional Market: North America
- Fastest Growing Region: Asia Pacific
- 3rd Largest Region: Europe
- Base Year: 2025
- Historical Period: 2021–2025
- Forecast Period: 2025–2035
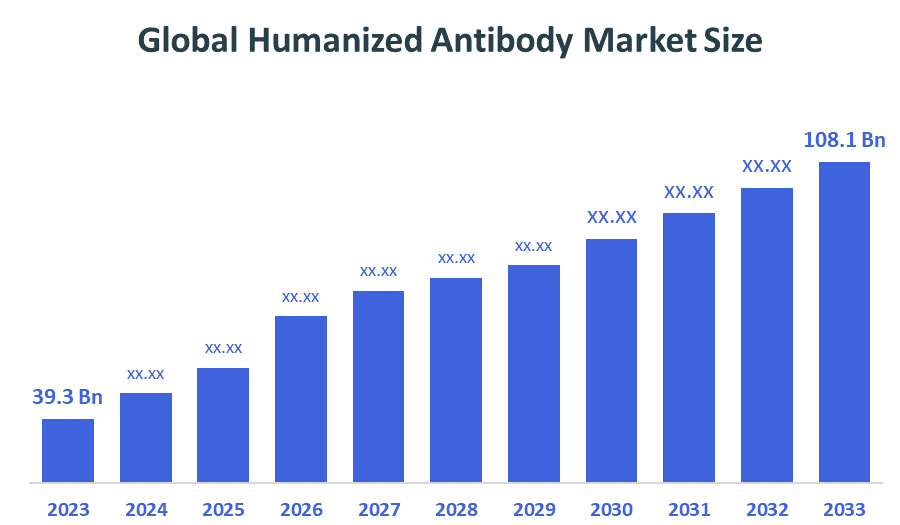
According to Decision Advisors, the Global Humanized Antibody Market Size is expected to grow from USD 39.3 billion in 2024 to USD 108.1 billion by 2035, at a CAGR of 10.65% during the forecast period 2025-2035. The global humanized antibody market is projected to grow significantly over the next decade. The market is expanding due to increasing adoption of targeted biologics, rising prevalence of chronic diseases, advancements in antibody engineering technologies, growing demand for personalized medicine, and increasing approvals of monoclonal antibody therapies.
Market Overview/ Introduction
The humanized antibody market refers to the global industry involved in the research, development, manufacturing, and commercialization of humanized antibodies, which are engineered antibodies derived from non-human sources and modified to closely resemble human antibodies. This modification reduces immunogenicity while maintaining high specificity and therapeutic effectiveness. The global humanized antibody market is currently in a high-growth phase. The expansion of this field is really helped by government plans like the US Project NextGen and India’s Biopharma SHAKTI. These plans give a lot of money for research and development and for trials. This helps us get ready for pandemics and makes sure people can afford biologics. The main good thing about this market is that it uses a process called humanization. In this process they replace mouse protein sequences. This is a deal because it reduces allergic reactions and helps the drug stay in the bloodstream for a longer time. It is better than the versions that used a mix of mouse and human parts. The humanization process is a part of making biologics, like the US Project NextGen and India’s Biopharma SHAKTI are supporting Future opportunities are emerging in the development of Antibody-Drug Conjugates (ADCs) and bispecific antibodies, which act like guided missiles to deliver chemotherapy directly to cancer cells or bridge immune cells to tumours. Capitalizing on this, company initiatives from giants like Roche, Merck, and AstraZeneca are shifting toward massive investments in AI-driven antibody discovery and local manufacturing hubs in the Asia-Pacific region to bypass supply chain hurdles.
- Lupin and Zentiva Sign License and Supply Agreement for Commercializing Certolizumab.
Notable Insights: -
- North America is anticipated to hold the largest share of the humanized antibody market over the predicted timeframe.
- Asia Pacific is expected to grow at a rapid CAGR in the humanized antibody market during the forecast period.
- By type, the monoclonal antibodies segment dominated the market in 2025 and is projected to grow at a substantial CAGR during the forecast period.
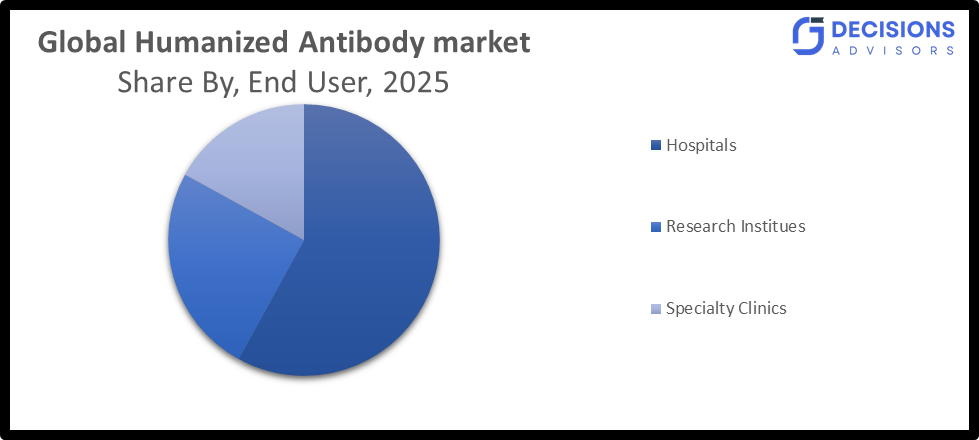
- By end user, the hospitals segment dominated the market in 2025 with a share 58%, and is projected to grow at a substantial CAGR during the forecast period.
- The compound annual growth rate of the global spirit market is 10.65%.
- The market is likely to achieve a valuation of USD 108.1 billion by 2035.
What is role of technology in grooming the market?
Technology plays a crucial role in advancing the humanized antibody market by enabling the development of highly specific and effective therapies. Techniques such as recombinant DNA technology, hybridoma technology, and phage display allow for precise antibody engineering. Artificial intelligence (AI) and machine learning are increasingly used in antibody discovery, optimization, and clinical trial design, significantly reducing development timelines. Additionally, advancements in bioinformatics and high-throughput screening enable identification of novel targets and faster drug development. Bioprocessing innovations and improved manufacturing technologies have enhanced production efficiency and scalability, reduced costs and increasing accessibility of antibody-based therapies.
Market Drivers
The humanized antibody market is driven by the increasing prevalence of cancer and autoimmune diseases, which require targeted and effective treatment approaches. The growing adoption of monoclonal antibody therapies due to their high specificity and reduced side effects compared to traditional treatments is significantly boosting demand. Advancements in antibody engineering technologies have enabled the development of highly effective therapies with improved safety profiles. Additionally, increasing investments in research and development, rising number of clinical trials, and continuous product approvals are accelerating market growth. The expanding use of antibody-drug conjugates (ADCs) and bispecific antibodies is also creating new opportunities, particularly in oncology treatment.
Restrain
The humanized antibody market faces challenges due to the high cost of antibody therapies, which limits accessibility in low- and middle-income regions. Complex manufacturing processes, stringent regulatory requirements, and risk of adverse immune responses also hinder market growth. Additionally, biosimilar competition may impact pricing and profit margins for original biologics.
Competitive Analysis:
The report offers the appropriate analysis of the key organizations/companies involved within the humanized antibody market, along with a comparative evaluation primarily based on their product of offering, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborative analysis focusing on the current news and developments of the companies, which includes product development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
Top Companies in the Global Humanized Antibody Market
- Roche Holding AG
- Amgen Inc.
- AbbVie Inc
- Merck & Co
- Pfizer Inc.
- Novartis AG
- Johnson & Johnson
- Lupin
- Bristol-Myers Squibb
- AstraZeneca
Government Initiatives
|
Country |
Key Government Initiatives |
|
US |
Government Procurement: The Department of Defense (DOD) has previously awarded multi-billion dollar contracts (e.g., $2.9B to Regeneron, $329.8M to Eli Lilly) to purchase large-scale doses of monoclonal antibody treatments. |
|
Europe |
Regional Development Funds: Projects like the PROPHYMAB project in France received EUR 15M from the French government to research antibodies for immunocompromised patients |
|
UK |
Government launches UK-wide antibody surveillance programme Home antibody tests available for up to 8,000 people a day. |
Market Segmentation
The humanized antibody market share is classified into type, application, and end user
- The monoclonal antibodies segment dominated the market in 2025 and is projected to grow at a substantial CAGR during the forecast period.
Based on the type, the humanized antibody market is divided into monoclonal antibodies, bispecific antibodies and antibody-drug conjugates. Among these, the monoclonal antibodies segment dominated the market in 2025 and is projected to grow at a substantial CAGR during the forecast period. This dominance is primarily due to their extensive clinical use across multiple therapeutic areas, especially oncology and autoimmune diseases. Monoclonal antibodies offer high specificity, improved safety profiles, and targeted mechanisms of action, making them more effective compared to conventional therapies. The increasing number of regulatory approvals, strong clinical pipeline, and rising adoption of biologics are further driving this segment. Additionally, advancements in antibody engineering and the growing availability of biosimilars are contributing to market expansion. Bispecific antibodies and ADCs are emerging as high-growth segments due to their ability to enhance therapeutic efficacy and deliver targeted treatment, particularly in cancer care.
- The oncology segment accounted for the largest share in 2025, and is anticipated to grow at a significant CAGR during the forecast period.
Based on the application, the humanized antibody market is divided into oncology, autoimmune diseases, infectious diseases and others. Among these, the oncology segment accounted for the largest share in 2025, and is anticipated to grow at a significant CAGR during the forecast period. This dominance is driven by the rising global burden of cancer and the increasing demand for targeted therapies that improve survival rates while minimizing side effects. Humanized antibodies are widely used in cancer immunotherapy due to their ability to specifically target tumor antigens. The autoimmune diseases segment is also witnessing substantial growth due to increasing prevalence of conditions such as rheumatoid arthritis and multiple sclerosis, along with rising adoption of biologic therapies. Meanwhile, infectious diseases are gaining attention with the development of antibody-based treatments for viral infections.
- The hospitals segment dominated the market in 2025 with a share 58%, and is projected to grow at a substantial CAGR during the forecast period.
Based on the end user, the humanized antibody market is divided into hospitals, research institutes, specialty clinics. Among these, the hospitals segment dominated the market in 2025 with a share 58%, and is projected to grow at a substantial CAGR during the forecast period, owing to the availability of advanced healthcare infrastructure, skilled medical professionals, and the capacity to administer complex biologic therapies such as monoclonal antibodies and antibody-drug conjugates. Hospitals also handle a large patient volume, particularly for chronic and life-threatening conditions like cancer and autoimmune diseases, which significantly contributes to their leading position.
Strategies to Implement for Growth of the Market in Non-Leading Regions
Growth can be accelerated through expansion of healthcare infrastructure, increased access to biologics, and development of cost-effective biosimilars. Strengthening local manufacturing capabilities and forming strategic partnerships with regional healthcare providers can improve market penetration. Additionally, increasing awareness about advanced biologic therapies and improving reimbursement policies will further enhance adoption in emerging markets.
Regional Segment Analysis of the Humanized Antibody Market
- North America (U.S., Canada, Mexico)
- Europe (Germany, France, U.K., Italy, Spain, Rest of Europe)
- Asia-Pacific (China, Japan, India, Rest of APAC)
- South America (Brazil and the Rest of South America)
- The Middle East and Africa (UAE, South Africa, Rest of MEA)
North America is anticipated to hold the largest share of the humanized antibody market over the predicted timeframe.
North America is anticipated to hold the largest share of the humanized antibody market over the predicted timeframe. The dominance of this region is primarily driven by the strong presence of leading biopharmaceutical companies, high adoption of advanced biologic therapies, and well-established healthcare infrastructure. The region benefits from significant investments in research and development, along with a high number of clinical trials focused on monoclonal antibodies and next-generation antibody therapies. Additionally, favorable reimbursement policies and rapid regulatory approvals have accelerated the commercialization of innovative treatments. The increasing prevalence of chronic diseases such as cancer and autoimmune disorders is further supporting the demand for humanized antibody therapies. Moreover, the growing adoption of precision medicine and biomarker-based treatments is enhancing treatment outcomes and driving overall market growth in the region.
Asia Pacific is expected to grow at a rapid CAGR in the humanized antibody market during the forecast period.
Asia Pacific is expected to grow at a rapid CAGR in the humanized antibody market during the forecast period. This growth is driven by the rising prevalence of chronic diseases, increasing healthcare expenditure, and expanding access to advanced biologic therapies across emerging economies such as China and India. The region benefits from a large patient population, improving healthcare infrastructure, and growing awareness regarding targeted treatments. Additionally, increasing government initiatives to support biotechnology research and local biopharmaceutical manufacturing are contributing to market expansion. The growing presence of global pharmaceutical companies and increasing number of clinical trials in the region are further accelerating growth. Furthermore, the rising adoption of biosimilars and cost-effective biologics is improving accessibility, thereby boosting demand for humanized antibody therapies.
Europe is the 3rd largest region to grow in the humanized antibody market during the period.
Europe is the third-largest region in the humanized antibody market, supported by advanced healthcare systems and strong research capabilities in biologics and antibody-based therapies. Key countries such as Germany, France, and the United Kingdom play a significant role in driving market growth due to their well-established pharmaceutical industries and increasing focus on innovation. The region has witnessed a steady rise in the adoption of monoclonal antibodies and biosimilars, supported by favorable regulatory frameworks and approvals from the European Medicines Agency (EMA). Additionally, the increasing prevalence of chronic diseases and rising demand for targeted therapies are contributing to market expansion. Strong government support for research and development, along with collaborations between academic institutions and industry players, is further strengthening the market in Europe.
Future Market Trends in the Global Humanized Antibody Market: -
- Rapid Growth of Bispecific Antibodies and Next-Generation Formats
The market is witnessing a strong shift toward advanced antibody formats such as bispecific antibodies and multispecific antibodies. These therapies can bind to multiple targets simultaneously, improving treatment efficacy compared to traditional monoclonal antibodies. Their increasing use in oncology and immunotherapy is driving innovation, with pharmaceutical companies heavily investing in next-generation antibody platforms. This trend is expected to significantly enhance treatment outcomes and expand therapeutic applications in the coming years.
- Expansion of Antibody-Drug Conjugates (ADCs) in Targeted Therapy
Antibody-drug conjugates (ADCs) are emerging as a key growth driver in the humanized antibody market, particularly in cancer treatment. ADCs combine the targeting ability of antibodies with potent cytotoxic drugs, allowing precise delivery of treatment to diseased cells while minimizing damage to healthy tissues. The ADC market is projected to grow steadily over the forecast period, supported by increasing approvals and strong clinical pipeline activity.
- Integration of Artificial Intelligence (AI) in Antibody Discovery
Artificial intelligence and machine learning are transforming antibody discovery and development processes. AI-driven platforms are enabling faster identification of target antigens, optimization of antibody structures, and prediction of binding affinity. These technologies significantly reduce development timelines and costs while improving success rates in clinical trials. The integration of AI is expected to accelerate innovation and drive the next wave of humanized antibody therapeutics.
Recent Development
In March 2026, Positive results from three phase 3 studies of amlitelimab, a fully human non-T cell depleting monoclonal antibody that selectively targets OX40-ligand (OX40L), in moderate-to-severe atopic dermatitis (AD) as a monotherapy and in combination with topical therapies, showed improvements in skin clearance and disease severity with amlitelimab treatment compared to placebo in patients aged 12 years and older.
In September 2025, Beckman Coulter Life Sciences launches industry-first infrared dye antibody conjugates to expand human phenotyping experiments.
In July 2025, Global Pharma major Lupin Limited (Lupin) and Zentiva Group (Zentiva), a pan-European pharmaceutical company, have entered into a license and supply agreement for commercialization of Lupin’s biosimilar Certolizumab Pegol, across multiple markets globally.
How is Recent Developments Helping the Market?
These trends and recent developments are significantly accelerating the growth of the humanized antibody market by enhancing therapeutic effectiveness, reducing development timelines, and expanding treatment applications. The rise of next-generation antibody formats, combined with AI-driven innovation and increasing approvals, is strengthening the clinical pipeline and improving patient outcomes. Additionally, continuous investments in R&D and growing demand for targeted therapies are ensuring sustained market expansion over the forecast period.
Market Segment
This study forecasts revenue at global, regional, and country levels from 2020 to 2035. Decision Advisors has segmented the humanized antibody market based on the below-mentioned segments:
Global Humanized Antibody Market, By Type
- Monoclonal Antibodies
- Bispecific Antibodies
- Antibody-Drug Conjugates
Global Humanized Antibody Market, By Application
- Oncology
- Autoimmune Diseases
- Infectious Diseases
- Others
Global Humanized Antibody Market, By End User
- Hospitals
- Research Institutes
- Specialty Clinics
Global Humanized Antibody Market, By Regional Analysis
- North America
- US
- Canada
- Mexico
- Europe
- Germany
- UK
- France
- Italy
- Spain
- Russia
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- Australia
- Rest of Asia Pacific
- South America
- Brazil
- Argentina
- Rest of South America
- Middle East & Africa
- UAE
- Saudi Arabia
- Qatar
- South Africa
- Rest of the Middle East & Africa
Frequently Asked Questions (FAQ)
Q. How does antibody humanization improve drug safety and effectiveness?
A. Antibody humanization reduces the immunogenicity of non-human antibodies by modifying them to resemble human antibodies, which minimizes immune reactions and improves compatibility with the human body. This enhances both safety and therapeutic effectiveness, especially for long-term treatments.
Q. What is the role of biosimilars in the humanized antibody market?
A. Biosimilars play a crucial role by offering cost-effective alternatives to branded biologics. They help increase treatment accessibility, especially in developing regions, while also driving competition and reducing overall healthcare costs.
Q. How are manufacturing challenges impacting the growth of this market?
A. The production of humanized antibodies involves complex bioprocessing techniques, strict quality control, and high investment costs. These challenges can limit scalability and increase pricing, thereby affecting widespread adoption in cost-sensitive markets.
Q. What opportunities exist for humanized antibodies beyond oncology?
A. Beyond oncology, humanized antibodies are increasingly being used in autoimmune diseases, infectious diseases, and neurological disorders. Growing research in these areas is expected to expand their therapeutic applications and create new growth opportunities in the future.
Check Licence
Choose the plan that fits you best: Single User, Multi-User, or Enterprise solutions tailored for your needs.
We Have You Covered
- 24/7 Analyst Support
- Clients Across the Globe
- Tailored Insights
- Technology Tracking
- Competitive Intelligence
- Custom Research
- Syndicated Market Studies
- Market Overview
- Market Segmentation
- Growth Drivers
- Market Opportunities
- Regulatory Insights
- Innovation & Sustainability
Report Details
| Scope | Global |
| Pages | 245 |
| Delivery | PDF & Excel via Email |
| Language | English |
| Release | Apr 2026 |
| Access | Download from this page |
