Global Idiopathic Thrombocytopenic Purpura Therapeutics Market
Global Idiopathic Thrombocytopenic Purpura Therapeutics Market Size, Share, By Product (Corticosteroids, Intravenous Immunoglobulins (IVIG), Thrombopoietin Receptor Agonists (TPO-RA) and Others), By Route of Administration (Oral, and Injectable), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies and Online Pharmacies), and By Region (North America, Europe, Asia-Pacific, Latin America, Middle East & Africa), Analysis and Forecast 2026-2035
Report Overview
Table of Contents
Market Snapshot
- Market Size (2025): USD 6.64 Billion
- Projected Market Size (2035): USD 11.86 Billion
- Compound Annual Growth Rate (CAGR): 5.97%
- Largest Regional Market: North America
- Fastest Growing Region: Asia-Pacific
- 3rd Largest Region: Europe
- Base Year: 2025
- Historical Period: 2021–2024
- Forecast Period: 2026–2035
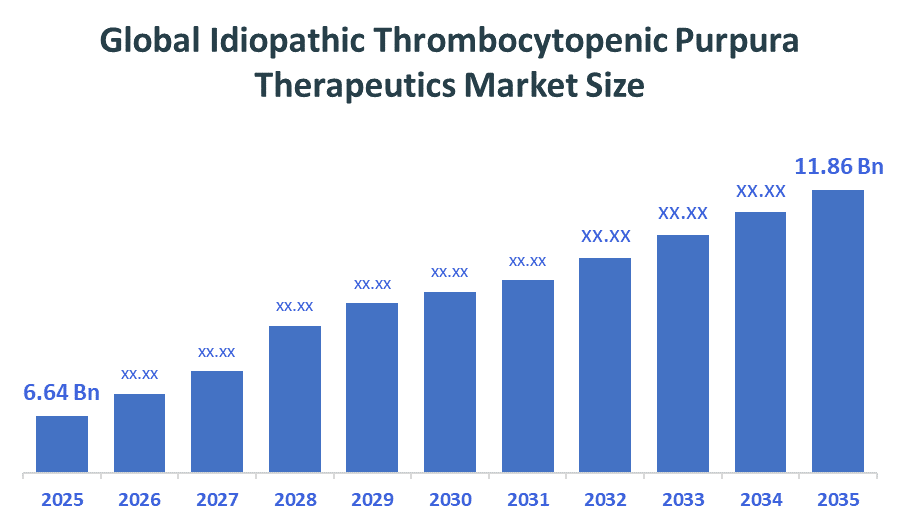
According to Decision Advisors, the Global Idiopathic Thrombocytopenic Purpura Therapeutics Market Size is expected to grow from USD 6.64billion in 2025 to USD 11.86billion by 2035, at a CAGR of 5.97% during the forecast period 2026-2035. The global Idiopathic Thrombocytopenic Purpura Therapeutics Market is projected to grow significantly over the next decade, fuelled by a rising elderly population, increased R&D for novel therapies, improving diagnostic, growing prevalence of chronic diseases, and better diagnostic technologies.
Market Overview/ Introduction
The Global Idiopathic Thrombocytopenic Purpura (ITP) Therapeutics Market comprises the development and distribution of treatment options such as corticosteroids, immunoglobulins, and thrombopoietin receptor agonists. Market growth is driven by the increasing prevalence of autoimmune and platelet disorders, along with advancements in diagnostic capabilities and rising demand for effective treatment solutions. Enhanced awareness among patients and healthcare professionals has led to earlier diagnosis and timely treatment intervention. Additionally, the introduction of thrombopoietin receptor agonists and targeted biologics has significantly improved treatment outcomes. Expanding healthcare infrastructure in emerging economies, supported by government initiatives and reimbursement programs, is further improving patient access to therapies. Moreover, continuous research and development efforts by pharmaceutical companies are playing a crucial role in driving innovation and overall market expansion.
- The Union Budget 2026 had laid the foundation for biopharma growth and affordable care, strengthening support for rare disease therapeutics such as Idiopathic Thrombocytopenic Purpura. The initiatives had promoted innovation, expanded collaborations, and improved patient access to advanced hematology treatments.
- Ayushman Bharat – Pradhan Mantri Jan Arogya Yojana (AB?PMJAY) had provided health coverage of ?5 lakh per family per year for secondary and tertiary care hospitalization to 12 crore families. In March 2024, 37 lakh families of Accredited Social Health Activists, Anganwadi Workers, and Anganwadi Helpers had been included, and the scheme was further expanded to cover 6 crore senior citizens aged 70 years and above, belonging to 4.5 crore families, irrespective of socio?economic status
Notable Insights: -
- North America is anticipated to hold the largest share of approximately 42.1% the idiopathic thrombocytopenic purpura therapeutics market over the forecast period.
- Asia Pacific is expected to grow at a rapid CAGR of approximately 7.10% in the idiopathic thrombocytopenic purpura therapeutics market during the forecast period.
- The thrombopoietin receptor agonists (TPO-RA) segment dominated the market in 2025 and is anticipated to grow at a significant CAGR of approximately 6.1% during the forecast period.
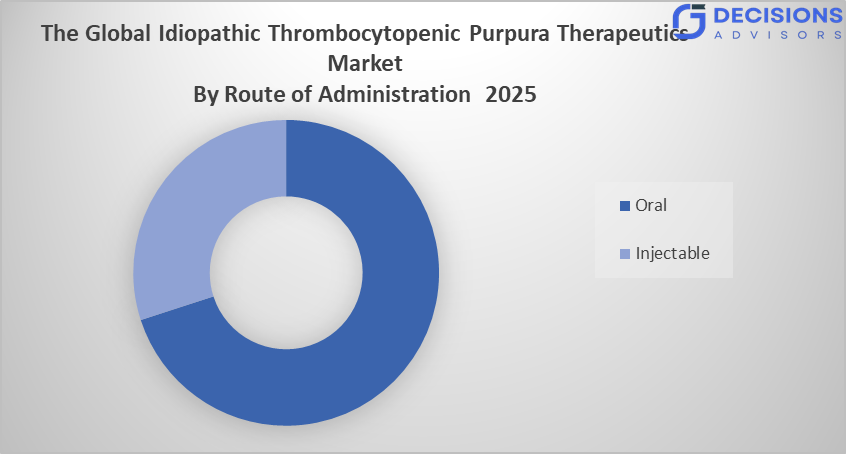
- The oral segment dominated the market in 2025 and is anticipated to grow at a significant CAGR of approximately 6.1% during the forecast period.
- The hospital pharmacies segment dominated the market in 2025 and is anticipated to grow at a significant CAGR of approximately 5.9% during the forecast period.
- The compound annual growth rate of the Global Idiopathic Thrombocytopenic Purpura Therapeutics Market is 5.97%.
- The market is likely to achieve a valuation of USD 11.86billion by 2035.
What is role of technology in grooming the market?
Technology plays a crucial role in advancing the global idiopathic thrombocytopenic purpura (ITP) therapeutics market by enhancing diagnosis, treatment, and patient management. Innovations in biotechnology have enabled companies like Novartis and Roche to develop targeted biologics and monoclonal antibodies with improved efficacy and safety. Digital health tools, including telemedicine and remote monitoring, support continuous patient tracking and better adherence to treatment. Artificial intelligence and data analytics accelerate drug discovery, optimize clinical trials, and enable personalized treatment strategies. Additionally, advanced manufacturing technologies improve the scalability and quality of biologics. These technological advancements collectively enhance treatment outcomes, expand access, and drive sustainable growth in the ITP therapeutics market.
Market Drivers
The Global Idiopathic Thrombocytopenic Purpura (ITP) Therapeutics Market is experiencing steady growth due to several key driving factors. The rising aging population is more vulnerable to immune system disorders and platelet dysfunction, significantly increasing the demand for effective treatment options. In addition, the development of innovative therapies, especially thrombopoietin receptor agonists (TPO-RAs), is transforming the treatment landscape by offering improved efficacy and safety profiles. The growing incidence of ITP linked to infections and underlying autoimmune conditions is further accelerating the need for disease management solutions. Moreover, advancements in diagnostic technologies and early detection methods are enabling the timely identification of patients, thereby boosting treatment rates and supporting overall market expansion globally.
Restrain
The Global Idiopathic Thrombocytopenic Purpura (ITP) Therapeutics Market is restrained by high treatment costs, especially for biologics from companies like Amgen, limiting access in developing regions. Adverse side effects, relapse risks, limited awareness, and stringent regulatory approvals further hinder adoption and delay market growth worldwide.
Competitive Analysis:
The report offers the appropriate analysis of the key organizations/companies involved within the global idiopathic thrombocytopenic purpura therapeutics market, along with a comparative evaluation primarily based on their product offerings, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborative analysis focusing on the current news and developments of the companies, which includes product development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
Top Companies in Global Idiopathic Thrombocytopenic Purpura Therapeutics Market
- Amgen Inc.
- F. Hoffmann-La Roche Ltd
- Novartis AG
- GSK plc
- Grifols S.A.
- Rigel Pharmaceuticals Inc.
- Sanofi S.A.
- Takeda Pharmaceutical Company Limited
- CSL Limited
- Octapharma AG
- Pfizer Inc.
- Bristol Myers Squibb
Government Initiatives
|
Country |
Key Government Initiatives |
|
USA |
The NIH is currently (March 2026) seeking public input on its next five-year framework, which emphasizes investigator-initiated science over rigid, specific funding opportunities. This allows researchers focusing on rare haematological disorders more flexibility to secure grants for novel ITP mechanisms |
|
France |
France offers an even more aggressive 14-day to 49-day authorization window for mononational trials involving "first-in-class" therapies or those for rare diseases like ITP |
|
Canada |
Health Canada has moved toward a "risk-based" regulatory process in 2026, aligning with "trusted foreign regulators" to accelerate the availability of innovative health products and reduce the regulatory burden for lower-risk manufacturing |
Study on the Supply, Demand, Distribution, and Market Environment of Idiopathic Thrombocytopenic Purpura Therapeutics Market
The Global Idiopathic Thrombocytopenic Purpura (ITP) Treatment Market displays a complicated landscape involving supply, demand, distribution, and market conditions. Pharmaceutical companies propel production on the supply side through ongoing R&D, reliant on raw materials like active pharmaceutical ingredients, biological components, antibodies, and plasma-derived substances utilized in corticosteroids and immunoglobulins. Nonetheless, fluctuations in raw material prices and interruptions in the supply chain can affect production expenses. Demand is increasing because of the higher prevalence of ITP and better diagnosis rates, elevating the need for advanced treatments. Distribution is mainly handled by hospital pharmacies, retail pharmacies, and online platforms, guaranteeing access to both injectable and oral therapies. In general, the market landscape is influenced by technological progress, regulatory policies, and increasing funding for hematology research, fostering continued global expansion.
Price Analysis and Consumer Behaviour Analysis
Price analysis and consumer behavior in the global Idiopathic Thrombocytopenic Purpura (ITP) therapeutics market reflect high treatment costs and value-driven decision-making. Advanced therapies such as thrombopoietin receptor agonists can exceed approximately $100,000 annually per patient, while 6-month treatment costs for drugs like eltrombopag range from approximately $36,000–$50,000. Intravenous immunoglobulin costs approximately $3,000–$4,000 per infusion, and rituximab therapy may exceed approximately $19,000 per treatment cycle. Consumers prioritize efficacy, safety, and long-term outcomes but are highly influenced by affordability and insurance coverage. In emerging markets, price sensitivity is higher, leading to a preference for corticosteroids or generics, while developed regions show higher adoption of premium biologics, shaping overall demand patterns.
Market Segmentation
The idiopathic thrombocytopenic purpura therapeutics market share is classified into product, route of administration, and distribution channel
- The thrombopoietin receptor agonists (TPO-RA) segment dominated the market in 2025 and is anticipated to grow at a significant CAGR of approximately 6.1% during the forecast period.
Based on the product, the idiopathic thrombocytopenic purpura therapeutics market is divided into corticosteroids, intravenous immunoglobulins (IVIG), thrombopoietin receptor agonists (TPO-RA), and others. Among these, the thrombopoietin receptor agonists (TPO-RA) segment dominated the market in 2025 and is anticipated to grow at a significant CAGR of approximately 6.1% during the forecast period. Due to their superior efficacy in increasing platelet counts, reduced bleeding risks, and favorable safety profile. Their ability to provide long-term disease control has driven widespread adoption among clinicians and patients.
- The oral segment dominated the market in 2025 and is anticipated to grow at a significant CAGR of approximately 6.5% during the forecast period.
Based on the route of administration, the idiopathic thrombocytopenic purpura therapeutics market is divided into oral and injectable. Among these, the oral segment dominated the market in 2025 and is anticipated to grow at a significant CAGR of approximately 6.5% during the forecast period. The growth of the oral segment is growing due to its convenience, improved patient compliance, and ease of long-term administration compared to injectable therapies. Increasing availability of effective oral drugs and preference for non-invasive treatment options are expected to drive its steady growth.
- The hospital pharmacies segment dominated the market in 2025 and is anticipated to grow at a significant CAGR of approximately 5.9% during the forecast period.
Based on the distribution channel, the idiopathic thrombocytopenic purpura therapeutics market is divided into hospital pharmacies, retail pharmacies, and online pharmacies. Among these, the hospital pharmacies segment dominated the market in 2025 and is anticipated to grow at a significant CAGR of approximately 5.9% during the forecast period. The growth of the hospital pharmacies segment is growing due to the availability of specialized treatments, access to advanced therapies, and presence of skilled healthcare professionals. Increasing hospitalization rates and preference for monitored treatment environments are expected to support its continued growth.
Strategies to Implement for Growth of the Market in Non-Leading Regions
Growth in non-leading regions can be driven through targeted strategies focused on access, affordability, and awareness. Companies like Novartis and Roche can expand distribution networks and collaborate with local healthcare providers to improve diagnosis and treatment reach. Implementing tiered pricing and patient assistance programs will enhance affordability in low- and middle-income countries. Increasing investment in local clinical trials helps address region-specific needs and regulatory pathways. Additionally, partnerships with governments and organizations like the World Health Organization can strengthen healthcare infrastructure and awareness campaigns. Leveraging digital health solutions, including telemedicine and remote monitoring, further supports early diagnosis, treatment adherence, and overall disease management in underserved markets.
Regional Segment Analysis of the Idiopathic Thrombocytopenic Purpura Therapeutics Market
- North America (U.S., Canada, Mexico)
- Europe (Germany, France, U.K., Italy, Spain, Rest of Europe)
- Asia-Pacific (China, Japan, India, Rest of APAC)
- South America (Brazil and the Rest of South America)
- The Middle East and Africa (UAE, South Africa, Rest of MEA)
North America is anticipated to hold the largest share of approximately 42.1% the idiopathic thrombocytopenic purpura therapeutics market over the forecast period.
North America is anticipated to hold the largest share of approximately 42.1% the idiopathic thrombocytopenic purpura therapeutics market over the forecast period. The dominance of this region is primarily driven by advanced healthcare infrastructure, high awareness levels, strong presence of key market players, and increased adoption of innovative treatment options.
Asia Pacific is expected to grow at a rapid CAGR of approximately 7.10% in the idiopathic thrombocytopenic purpura therapeutics market during the forecast period.
Asia Pacific is expected to grow at a rapid CAGR of approximately 7.10% in the idiopathic thrombocytopenic purpura therapeutics market during the forecast period. This growth is driven by rising disease awareness, improving healthcare infrastructure, increasing patient population, and expanding access to advanced treatment options across emerging economies.
Europe is the 3rd largest region to grow in the idiopathic thrombocytopenic purpura therapeutics market during the period.
Europe was the third largest region to grow in the idiopathic thrombocytopenic purpura therapeutics market during the period, supported by well-established healthcare systems, increasing awareness of autoimmune disorders, growing access to advanced therapies, and favorable government initiatives promoting early diagnosis and treatment.
Future Market Trends in Global Idiopathic Thrombocytopenic Purpura Therapeutics Market: -
1. Shift Toward Targeted & Personalized Therapies
The shift toward targeted and personalized therapies is driven by improved understanding of immune mechanisms in idiopathic thrombocytopenic purpura. Advances in biologics by companies like Novartis enable precise targeting of disease pathways, reducing side effects and improving outcomes. Growing demand for effective, long-term, patient-specific treatments further accelerates this trend.
2. Rising Adoption of Combination Therapies
The rising adoption of combination therapies is driven by their ability to deliver improved treatment outcomes in idiopathic thrombocytopenic purpura. Combining corticosteroids, immunoglobulins, and thrombopoietin receptor agonists enhances platelet response and reduces relapse rates. Companies like Amgen are advancing such approaches, offering better efficacy, lower toxicity, and more durable disease control compared to monotherapy.
3. Expansion of Clinical Trials & Digital Healthcare Integration
The expansion of clinical trials and digital healthcare integration is driven by the need for faster drug development and improved patient management in idiopathic thrombocytopenic purpura. Companies like Roche are leveraging global trials and digital tools such as remote monitoring and telemedicine, enabling real-time data collection, broader patient access, and more efficient evaluation of emerging therapies.
Recent Development
- In March 2026, Gamgertamig (OM336) was a clinical?stage BCMAxCD3 T?cell engager that enabled rapid and deep plasma and B?cell depletion through a short?duration, subcutaneously administered treatment course, representing a novel immune?modulating approach in hematologic and autoimmune disorders.
- In March 2026, the immune thrombocytopenia clinical trial pipeline had expanded as more than 30 companies drove innovation in therapeutics, strengthening the Idiopathic Thrombocytopenic Purpura market with diverse biologics, small molecules, and immune?modulating approaches that enhanced treatment prospects and competitive momentum.
- In March 2026, 3SBio had announced its 2025 annual results, surpassing the RMB 10 billion revenue milestone. The achievement was propelled by dual engines of innovative R&D and global collaboration, reinforcing the company’s financial strength and commitment to advancing immune?mediated and hematologic therapeutics.
- In December 2025, Novartis ianalumab significantly extends disease control in patients with immune thrombocytopenia with only four once-monthly doses.
How is Recent Developments Helping the Market?
Recent developments are significantly accelerating growth in the global Idiopathic Thrombocytopenic Purpura therapeutics market. Innovations by companies such as Novartis and Amgen in monoclonal antibodies and thrombopoietin receptor agonists are improving treatment efficacy and safety. Advancements in targeted immunotherapies and steroid-sparing approaches are enhancing long-term disease management and patient outcomes. Strategic collaborations and licensing agreements between major pharma and emerging biotech firms are expediting clinical trials and regulatory approvals. Additionally, increasing investments in rare disease research and growing adoption of personalized medicine are enabling more precise treatment strategies. These developments collectively expand therapeutic options, improve accessibility, and drive sustained market growth worldwide.
Market Segment
This study forecasts revenue at global, regional, and country levels from 2020 to 2035. Decision Advisors has segmented the idiopathic thrombocytopenic purpura therapeutics market based on the below-mentioned segments:
Global Idiopathic Thrombocytopenic Purpura Therapeutics Market, By Product
- Intravenous Immunoglobulins (IVIG)
- Thrombopoietin Receptor Agonists (TPO-RA)
- Others
Global Idiopathic Thrombocytopenic Purpura Therapeutics Market, By Route of Administration
- Oral
- Injectable
Global Idiopathic Thrombocytopenic Purpura Therapeutics Market, By Distribution Channel
- Hospital Pharmacies
- Retail Pharmacies
- Online Pharmacies
Global Idiopathic Thrombocytopenic Purpura Therapeutics Market, By Regional Analysis
- North America
- US
- Canada
- Mexico
- Europe
- Germany
- UK
- France
- Italy
- Spain
- Russia
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- Australia
- Rest of Asia Pacific
- South America
- Brazil
- Argentina
- Rest of South America
- Middle East & Africa
- UAE
- Saudi Arabia
- Qatar
- South Africa
- Rest of the Middle East & Africa
Frequently Asked Questions (FAQ)
Q What are the major challenges faced by the ITP therapeutics market?
A. The market faces challenges such as high treatment costs, limited accessibility in low-income regions, and variability in patient response to therapies. Additionally, long-term safety concerns and frequent relapses create complexities in disease management, affecting overall treatment adoption and market growth.
Q. How do biologics influence the growth of the ITP therapeutics market?
A. Biologics significantly contribute to market growth by offering targeted treatment approaches with improved efficacy and reduced side effects. They help in better immune modulation, leading to sustained platelet counts and improved patient outcomes, thereby increasing their adoption globally.
Q. What role do emerging markets play in the expansion of the ITP therapeutics market?
A. Emerging markets play a crucial role by providing untapped growth opportunities due to increasing healthcare investments, improving diagnostic capabilities, and rising awareness. Expanding healthcare access and government initiatives further support the adoption of advanced therapies in these regions.
Q. How does patient adherence impact the ITP therapeutics market?
A. Patient adherence directly affects treatment outcomes and market growth. Therapies that are easy to administer, have fewer side effects, and require less frequent dosing tend to improve adherence, leading to better clinical results and increased demand for such treatments.
Check Licence
Choose the plan that fits you best: Single User, Multi-User, or Enterprise solutions tailored for your needs.
We Have You Covered
- 24/7 Analyst Support
- Clients Across the Globe
- Tailored Insights
- Technology Tracking
- Competitive Intelligence
- Custom Research
- Syndicated Market Studies
- Market Overview
- Market Segmentation
- Growth Drivers
- Market Opportunities
- Regulatory Insights
- Innovation & Sustainability
Report Details
| Scope | Global |
| Pages | 240 |
| Delivery | PDF & Excel via Email |
| Language | English |
| Release | Apr 2026 |
| Access | Download from this page |
