Global Immune Thrombocytopenic Purpura Market
Global Immune Thrombocytopenic Purpura Market Size, Share, By Drug Type (Steroid, Immunoglobulins, Thrombopoietin, Receptor Agonist, and Others), By Disease Type (Acute ITP, Chronic ITP, and Persistent ITP), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, and Online Pharmacies), By and By Region (North America, Europe, Asia-Pacific, Latin America, Middle East, and Africa), Analysis and Forecast 2025 ? 2035.
Report Overview
Table of Contents
Market Snapshot
- Market Size (2025): USD 3.9 Billion
- Projected Market Size (2035): USD 4.83 Billion
- Compound Annual Growth Rate (CAGR): 2.16%
- Largest Regional Market: North America
- Fastest Growing Region: Asia Pacific
- 2nd Largest Region: Europe
- Base Year: 2025
- Historical Period: 2021–2024
- Forecast Period: 2025–2035
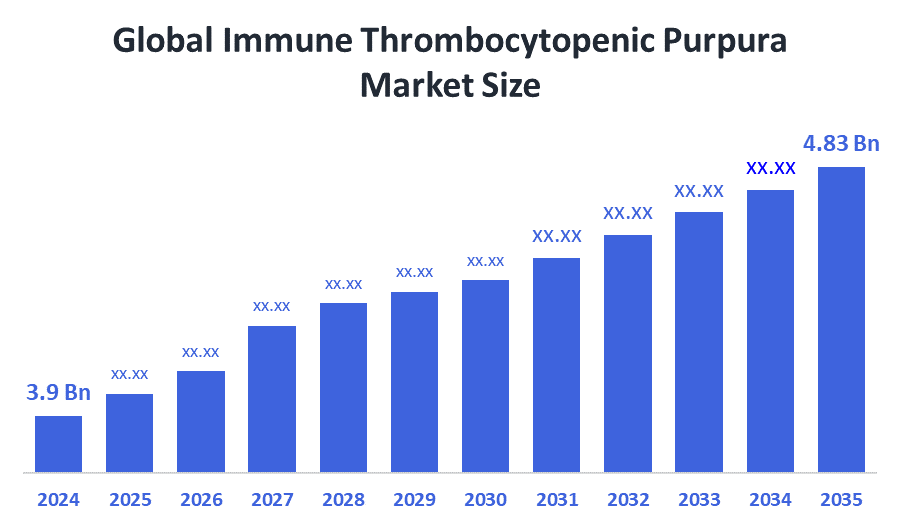
According to Decision Advisors, the Global Immune Thrombocytopenic Purpura Market Size is expected to grow from USD 3.9 Billion in 2025 to USD 4.83 billion by 2035, at a CAGR of 2.16% during the forecast period 2025-2035. The global immune thrombocytopenic purpura market is projected to grow significantly over the next decade as the diagnosed patient population is increasing, while advanced therapies are being adopted, research and development efforts are proceeding with their pipeline creation and government programs are being implemented.
Market Overview/ Introduction
The market for immune thrombocytopenic purpura (ITP) exists to create, manufacture and sell treatment solutions which target ITP, a rare autoimmune disorder that causes patients to have diminished platelet counts resulting in higher chances of experiencing bleeding and bruising. The market provides various treatment solutions, which include corticosteroids, immunoglobulins, thrombopoietin receptor agonists, immunosuppressants and new targeted treatment methods that contain BTK inhibitors. The market operates because of three main factors, including the growing number of people who experience autoimmune disorders, the rising public knowledge, and the better treatment solutions, research and development initiatives that are now happening.
The FDA granted orphan drug status to emerging therapies like gamgertamig, supporting rare disease drug development. Governments are increasingly promoting clinical trials and fast-track designations to bring innovative therapies faster. The FDA continues post-marketing safety monitoring of blood disorder drugs to ensure patient safety. Next-generation therapies show development through BTK inhibitors, monoclonal antibodies, and FcRn inhibitors, which create new approaches for disease treatment. The innovation process is moving forward at high speed because there are more than 30 companies that have developed more than 25 pipeline drugs.
- The U.S. Food and Drug Administration (FDA) approved the new treatment rilzabrutinib in September 2025. The treatment provides additional options for patients with chronic ITP who have not responded to standard therapies.
Notable Insights: -
- North America holds the largest regional market share, approximately 39.5% in the global immune thrombocytopenic purpura market.
- Asia Pacific is the fastest-growing region in the global immune thrombocytopenic purpura market.
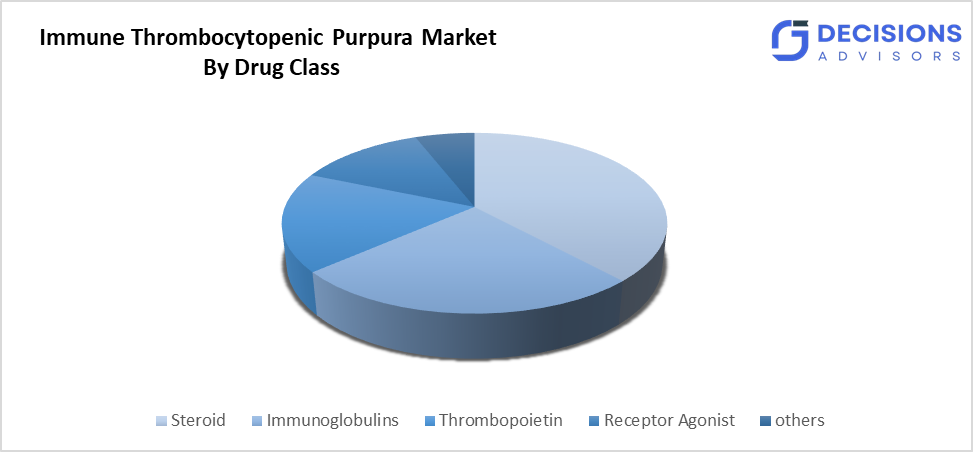
- By drug type, the steroid segment held a dominant position of over 45% in terms of market share in 2025.
- By distribution channel, the retail pharmacies segment is the dominating one, accounting for over 37% of the global market share in 2025.
- The compound annual growth rate of the global immune thrombocytopenic purpura market is 2.16%.
- The market is likely to achieve a valuation of USD 4.83 billion by 2035.
What is role of technology in grooming the market?
Technology plays a critical role in shaping this growth by enhancing diagnosis, treatment, and patient management. Advanced diagnostic technologies, which include automated blood analyzers and AI-based tools, deliver faster and more accurate detection of platelet disorders resulting in higher early diagnosis rates. Digital health platforms, with telemedicine services, enable better access to hematology specialists who serve remote locations. Biotech innovations have created targeted treatments, which include thrombopoietin receptor agonists and BTK inhibitors that enhance platelet production while decreasing immune system-based platelet destruction. Clinicians use electronic health records together with data analytics to track disease development while creating customized treatment strategies. Mobile health applications assist patients in following their treatment plans by monitoring their symptoms and medication intake. Technology accelerates both drug discovery and clinical trials through its ability to model data and create simulated environments. The advancements in healthcare technology produce better patient results while making medical services more accessible, which leads to ongoing international market expansion for ITP treatment products.
Market Drivers
The global immune thrombocytopenic purpura market is propelled by The number of ITP cases continues to grow because current diagnostic methods and public understanding of the disease have improved. Innovative treatments such as thrombopoietin receptor agonists (TPO-RAs) and BTK inhibitors are improving patient outcomes and expanding the market. The pharmaceutical industry is investing more resources into clinical trials, which will speed up the process of developing new treatment methods. Medical professionals now have access to advanced diagnostic tools which provide accurate test results that can identify patients at an early stage of treatment. Increased healthcare spending with improved insurance benefits has made ITP treatments more accessible to patients, particularly in developed countries. Awareness campaigns and education among healthcare professionals are leading to earlier diagnosis and timely treatment.
In August 2025, the U.S. FDA approved Wayrilz (rilzabrutinib), the first BTK inhibitor for ITP, marking a breakthrough in treatment.
Restrain
The main restraints on the global immune thrombocytopenic purpura market include the high costs of innovative medical treatments, which include thrombopoietin receptor agonists and biologics, creating obstacles for patients, in particular reside in low- and middle-income areas. The long-term administration of corticosteroids with immunosuppressive drugs results in dangerous adverse effects, which include infections and osteoporosis, as well as weight gain that decreases patient adherence. The ITP condition experiences diagnostic errors and delayed detection because developing regions lack access to essential research facilities, which leads to both treatment delays and market expansion limitations.
Competitive Analysis:
The report offers the appropriate analysis of the key organizations/companies involved within the immune thrombocytopenic purpura market, along with a comparative evaluation primarily based on their product of offering, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborative analysis focusing on the current news and developments of the companies, which includes product development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
Top Companies in Global Immune Thrombocytopenic Purpura Market
- Amgen Inc.
- Novartis AG
- CSL Limited
- Grifols S.A.
- Rigel Pharmaceuticals, Inc.
- Pfizer Inc.
- Bristol-Myers Squibb
- F. Hoffmann-La Roche Ltd
- GlaxoSmithKline plc
- Takeda Pharmaceutical Company
Government Initiatives
|
Country |
Key Government Initiatives |
|
Europe |
The European Commission has granted approval for Wayrilz, which contains rilzabrutinib as a new treatment option for immune thrombocytopenia in adult patients who have not responded to other therapies. This follows the positive opinion by the European Medicines Agency's Committee for Medicinal Products for Human Use. |
|
US |
The US Food and Drug Administration (FDA) has approved Wayrilz (rilzabrutinib) for adults with persistent or chronic immune thrombocytopenia (ITP) who have had an insufficient response to a previous treatment. |
|
Japan |
Argenx SE, which develops immunological therapies, develops treatments that enhance the quality of life for individuals who face severe autoimmune diseases, announced that VYVGART (efgartigimod alfa) received approval from Japan's Ministry of Health Labour and Welfare (MHLW) for intravenous (IV) administration to treat adults suffering from primary immune thrombocytopenia (ITP). |
Market Segmentation
The immune thrombocytopenic purpura market share is classified into drug type, disease type, and distribution channel.
- The steroid segment dominated the market in 2025, and is projected to grow at a substantial CAGR of approximately 45% during the forecast period.
Based on the drug type, the immune thrombocytopenic purpura market is divided into steroid, immunoglobulins, thrombopoietin, receptor agonist, and others. Among these, the steroid segment dominated the market in 2025 and is projected to grow at a substantial CAGR of approximately 45% during the forecast period. The growth of the segment is driven by the medical community uses these treatments because their platelet count results improve quickly after treatment. The treatments cost less than other options, and they can be found in many locations, which makes them suitable for use according to the first treatment guidelines. The treatments maintain their strong performance and cost efficiency, which leads to their ongoing application in emergencies and long-term medical care throughout the world.
- The non-surgical treatments segment accounted for the largest share in 2025, and is anticipated to grow at a significant CAGR of approximately 67% during the forecast period.
Based on the disease type, the immune thrombocytopenic purpura market is divided into acute ITP, chronic ITP, and persistent ITP. Among these, the non-surgical treatments segment accounted for the largest share in 2025, and is anticipated to grow at a significant CAGR of approximately 67% during the forecast period. Chronic ITP dominates because it persists for more than 12 months and requires long-term monitoring and treatment. The condition is more common in adults than both acute and persistent forms of the disease. The condition's continuous need for treatment together with its high rate of relapses and its increased demand for medical services, all contribute to its growth, which makes up a larger portion of the complete ITP market.
- The retail pharmacies segment dominated the market in 2025, and is projected to grow at a substantial CAGR of approximately 37% during the forecast period.
Based on the distribution channel, the immune thrombocytopenic purpura market is divided into hospital pharmacies, retail pharmacies, and online pharmacies. Among these, the hospital pharmacies segment dominated the market in 2025, and is projected to grow at a substantial CAGR of approximately 37% during the forecast period. The combination of widespread drug distribution and simple purchasing options together with high patient demand for local drugstores drives their need to acquire medications from nearby pharmacies. The organization manages extensive prescription deliveries together with ongoing medical care for patients with chronic diseases because their system provides uninterrupted access to pharmaceuticals and pharmacist assistance, which results in increased operational capacity and market penetration.
Regional Segment Analysis of the Immune Thrombocytopenic Purpura Market
- North America (U.S., Canada, Mexico)
- Europe (Germany, France, U.K., Italy, Spain, Rest of Europe)
- Asia-Pacific (China, Japan, India, Rest of APAC)
- South America (Brazil and the Rest of South America)
- The Middle East and Africa (UAE, South Africa, Rest of MEA)
North America is anticipated to hold the largest share of the immune thrombocytopenic purpura market over the predicted timeframe.
North America is anticipated to hold the largest share of the Immune Thrombocytopenic Purpura market over the predicted timeframe. This market is accounting about approximately 39.5% of the total market share. North America dominates the global ITP market due to its advanced healthcare system and widespread disease knowledge and major pharmaceutical companies maintain their operations throughout the region. In 2024, North America accounted for approximately 39.5% of the global ITP market share which made it the primary regional market driver. The United States alone represents the largest share within this region, contributing nearly 66% of the total market across major global markets (7MM). Hospitals provide advanced medical treatments to patients who require them because current medical facilities receive lump-sum payments from insurance companies which cover all hospital costs. The market expansion is fueled by two factors: high healthcare expenditures and the early implementation of cutting-edge medical solutions. North America maintains its dominance in the global ITP market because it possesses extensive clinical research and regulatory backing which fast-tracks drug approval processes.
Asia Pacific is expected to grow at a rapid CAGR in the immune thrombocytopenic purpura market during the forecast period. Asia Pacific is poised to be the fastest-growing region in the global Immune Thrombocytopenic Purpura market with a remarkable CAGR of 7% during the forecast period of 2025 to 2035. China and India serve as major economic drivers because their large populations create economic development in their respective countries. The Asia-Pacific region will experience its highest growth because of two factors which include the developing healthcare infrastructure and the rising patient awareness along with the increasing diagnosis rates of autoimmune diseases such as ITP. The countries of China India and Japan achieve economic growth through better access to biologic treatments which receive government funding through their insurance programs. The global ITP market was valued at ~USD 3.57 billion in 2025 and is projected to reach ~USD 4.53 billion by 2035 at ~2.4% CAGR which Asia-Pacific will experience as its most rapid regional growth. The region experiences better treatment access because of two developments which include increased use of advanced therapies and the establishment of local drug manufacturing facilities.
Europe is the 2nd largest region to grow in the Immune Thrombocytopenic Purpura market during the period. This growth is driven by Robust healthcare systems, early diagnosis, and access to advanced treatments such as thrombopoietin receptor agonists were the primary factors that would assist in building up the case in favor of regional-market superiority, with the UK contributing significantly to the total European market (30% to the EU4+UK).
Strategies to Implement for Growth of the Market in Non-Leading Regions
Strategies to grow the immune thrombocytopenic purpura (ITP) market in non-leading regions should focus on improving access, awareness, and affordability. Disease awareness increases through educational programs which train both patients and healthcare professionals. Healthcare systems require both infrastructure improvements and better access to hematology specialists. Governments and private players should work together to deliver subsidized medical treatments while expanding insurance coverage options. Local manufacturing development will lead to lower drug prices and better product accessibility. Telemedicine implementation will enable healthcare providers to serve remote area patients. The development of clinical trials and regional partnerships will help companies understand local market requirements which will enhance their product adoption and business growth.
Future Market Trends in Global Immune Thrombocytopenic Purpura Market:
- Rise of targeted and novel therapies
Targeted therapies are transforming the ITP market, with novel agents like BTK inhibitors improving outcomes. In 2025, the FDA approved rilzabrutinib, the first BTK inhibitor for ITP, showing ~23–29% durable platelet response in Phase III trials. This innovation highlights a shift toward disease-modifying, precision-based treatments.
- Growing adoption of thrombopoietin receptor agonists
Thrombopoietin receptor agonists (TPO-RAs) are increasingly adopted, holding ~42% market share in 2025 and growing at 6.7% CAGR due to their ability to boost platelet production and manage chronic ITP effectively. Recent developments, including rising sales of romiplostim and ongoing clinical trials, highlight strong demand and expanding therapeutic use.
- Advancements in diagnostics and early detection
Advancements in AI-based diagnostics are transforming early detection, with models achieving 90–98% sensitivity and outperforming dermatologists in 63% of studies. Recent 2025 research shows AI improved diagnostic accuracy from 64.7% to 70.3% while reducing diagnosis time by ~40%, enabling faster and more accurate CLE detection.
Recent Development
- In December 2025, Novartis announced positive results from VAYHIT2, a Phase III trial evaluating ianalumab plus eltrombopag in patients with primary immune thrombocytopenia (ITP) previously treated with corticosteroids1-3. Ianalumab (9 mg/kg) plus eltrombopag extended ITP disease control by 45%, based on the primary endpoint of time to treatment failure (TTF), which assesses how long patients maintain safe platelet levels during and after the treatment period.
- In October 2025, Grifols, a global healthcare company and leading producer of plasma-derived medicines and innovative diagnostic solutions, announced that Yimmugo, an innovative intravenous immunoglobulin (IVIg) therapeutic, produced by Biotest – a Grifols Group company – will officially launch in the United States for the treatment of primary immunodeficiencies (PID).
- In July 2025, Sobi North America, the North American affiliate of Swedish Orphan Biovitrum AB, announced that the U.S. Food and Drug Administration (FDA) approved Doptelet (avatrombopag) for the treatment of thrombocytopenia in pediatric patients one year and older with persistent or chronic immune thrombocytopenia (ITP) who have had an insufficient response to a prior therapy.
How is Recent Developments Helping the Market?
Recent developments are significantly supporting the growth of the immune thrombocytopenic purpura (ITP) market by the process of developing effective treatments, accurate diagnostic methods and better patient access to healthcare services. The introduction of advanced therapies such as Bruton’s Tyrosine Kinase (BTK) inhibitors, which include rilzabrutinib, has created new treatment possibilities for patients who do not respond to standard medical treatment. The growing use of thrombopoietin receptor agonists (TPO-RAs) is helping to increase platelet production while simultaneously decreasing the frequency of bleeding episodes. The recent advances in diagnostic technology enable medical professionals to identify diseases at earlier stages with more precise methods, which results in expanded access to testing for additional patients. The regulatory framework provides support through its mechanisms of expedited approval processes and orphan drug designations, which enable pharmaceutical companies to introduce new treatments to the marketplace at an accelerated pace.
Market Segment
This study forecasts revenue at global, regional, and country levels from 2020 to 2035. Decision Advisors has segmented the immune thrombocytopenic purpura market based on the below-mentioned segments:
Global Immune Thrombocytopenic Purpura Market, By Drug Type
- Steroid
- Immunoglobulins
- Thrombopoietin
- Receptor Agonist
- Others
Global Immune Thrombocytopenic Purpura Market, By Disease Type
- Acute ITP
- Chronic ITP
- Persistent ITP
Global Immune Thrombocytopenic Purpura Market, By Distribution Channel
- Hospital Pharmacies
- Retail Pharmacies
- Online Pharmacies
Global Immune Thrombocytopenic Purpura Market, By Regional Analysis
- North America
- US
- Canada
- Mexico
- Europe
- Germany
- UK
- France
- Italy
- Spain
- Russia
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- Australia
- Rest of Asia Pacific
- South America
- Brazil
- Argentina
- Rest of South America
- Middle East & Africa
- UAE
- Saudi Arabia
- Qatar
- South Africa
- Rest of the Middle East & Africa
Frequently Asked Questions (FAQ)
Q: Which route of administration is most common in ITP treatment?
A: Oral therapies dominate with a 55-65% market share, as they are convenient, easy to administer, and preferred for long-term use. Targeted therapies and TPO-RAs are mostly oral, improving patient adherence and reducing hospital visits compared to injectable immunoglobulins or steroids.
Q: Which region leads the ITP market?
A: North America leads the global ITP market with a 39.5% share, due to advanced healthcare infrastructure, high patient awareness, widespread adoption of innovative therapies, strong research and development, and early diagnosis capabilities in the region.
Q: What role do biologics play in the ITP market?
A: Biologics, including monoclonal antibodies, are increasingly adopted for patients unresponsive to corticosteroids. They target specific immune pathways, reduce platelet destruction, and minimize side effects, representing a growing segment. Biologics are projected to achieve a 15-18% CAGR through 2032, improving long-term patient outcomes and expanding the ITP treatment landscape.
Q: How does technology impact the ITP market?
A: Technology improves ITP management through AI-assisted diagnostics, telemedicine, digital health monitoring, and automated blood tests. These tools enable earlier detection, better treatment adherence, and personalized care. Technology also accelerates clinical trials and drug development, making therapies safer, more effective, and expanding market reach globally.
Check Licence
Choose the plan that fits you best: Single User, Multi-User, or Enterprise solutions tailored for your needs.
We Have You Covered
- 24/7 Analyst Support
- Clients Across the Globe
- Tailored Insights
- Technology Tracking
- Competitive Intelligence
- Custom Research
- Syndicated Market Studies
- Market Overview
- Market Segmentation
- Growth Drivers
- Market Opportunities
- Regulatory Insights
- Innovation & Sustainability
Report Details
| Scope | Global |
| Pages | 210 |
| Delivery | PDF & Excel via Email |
| Language | English |
| Release | Mar 2026 |
| Access | Download from this page |
