Japan Excipients Market
Japan Excipients Market Size, Share, By Product Type (Polymers, Alcohols, Sugars, Minerals, Gelatin) By Formulation (Tablets, Capsules, Parenteral, Topical, Liquid dosage form) and By Application (Oral dosage forms, Injectable formulations, Topical applications, Inhalation products, Ophthalmic preparations, Nutraceuticals & R&D), Analysis and Forecast 2025-2035
Report Overview
Table of Contents
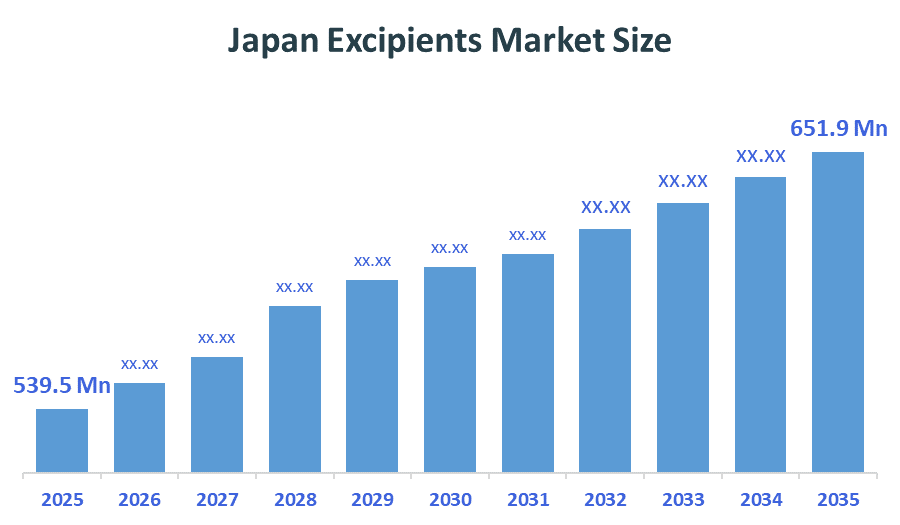
The Japan Excipients Market size is forecast to grow from approximately USD 539.5 Million in 2025 to around USD 651.9Million by 2035. According to Decision Advisors, a detailed research report on the buprenorphine market indicates that multifunctional excipients, dominate the market, accounting for approximately 43-46% of the total share in Japan. BASF SE leads the market with an estimated 12-15% Japan market share. The company's annual revenue of approximately USD 75-78 Billion is making it one of the most influential forces shaping industry trends and overall market growth.
Market Snapshot
- Japan Excipients Market Size (2025): USD 539.5Million
- Projected Japan Excipients Market Size (2035): USD 651.9Million
- Japan Excipients Market Compound Annual Growth Rate (CAGR): 1.91%
- Base Year: 2025
- Historical Period: 2021–2025
- Forecast Period: 2026–2035
Market Overview/Introduction
The Japan Excipients Market consists of three main activities, which include manufacturing, distributing, and selling inactive substances that pharmaceutical companies, nutraceutical manufacturers, and biopharmaceutical developers use to create stable drug formulations. The performance of active pharmaceutical ingredients improves through the use of excipients, which include binders and fillers, and lubricants and stabilizers that do not provide any medicinal benefits. Right now, Japan is moving past basic filler ingredients and stepping into an era of high-tech, functional materials. The process now requires more than simply packing a capsule because it needs to create medicines that provide better results for patients who use them. The Japanese government Life Sciences Fund provides its major support to this transformation, which aims to establish the country as a self-sufficient leader in pharmaceutical manufacturing. At the same time, British giants like Croda are ditching old-school methods for high-tech, plant-based materials that are much kinder to the planet. The real game-changer, though, is personalized medicine where smart ingredients are custom-tailored to a patient’s own biology, ensuring the treatment hits exactly when and where it's needed. As Japan continues to lead in genomics and 3D-printed drugs, seeing a massive opportunity for smart ingredients. These are custom-tailored to a patient’s own biology, ensuring that complex therapies are not only more effective but also released exactly when and where the body needs them most.
- Japan’s Ministry of Health, Labour and Welfare recently simplified approval requirements for excipients listed in major foreign pharmacopoeias like USP and EP. This reduces documentation burdens, accelerates imports, and supports global harmonization, making it easier for international excipient suppliers to enter the Japanese pharmaceutical market.
- The government regularly updates the Japanese Pharmacopoeia (JP), with full revisions every five years and interim supplements. These updates refine excipient standards, safety, and quality requirements, ensuring compliance with evolving scientific and regulatory expectations while maintaining high-quality benchmarks for pharmaceutical manufacturing.
Notable Insights: -
- The polymers segment dominated the market in 2025 and holds the largest market share, accounting for approximately 32% during the forecast period.
- The tablets segment dominated the market in 2025 and holds the largest market share, accounting for approximately 38% during the forecast period.
- The oral dosage segment dominated the market in 2025 and holds the largest market share, accounting for approximately 45% during the forecast period.
- The compound annual growth rate of the Japan Excipients Market is 1.91%.
- The market is likely to achieve a valuation of USD 651.9million by 2035.
What is the role of technology in grooming the market?
Technology plays a transformative role in shaping the Japan excipients market by enabling the development of advanced, multifunctional excipients that improve drug stability, solubility, and targeted delivery. Innovations such as nanotechnology, 3D printing, and continuous manufacturing enhance formulation efficiency and reduce production costs. Digital tools and AI-driven analytics support faster drug development and quality control. Additionally, technology facilitates compliance with evolving regulatory standards and supports personalized medicine trends. These advancements are driving demand for high-performance excipients, positioning Japan as a key hub for innovation and growth in the global pharmaceutical excipients market.
Market Drivers
The Japan Excipients Market is driven by rising demand for advanced formulation solutions that improve drug stability, bioavailability, and patient compliance. Increasing focus on biologics, biosimilars, and precision medicines has accelerated the need for high-quality and functional excipients. The growth of contract manufacturing organizations and the expansion of pharmaceutical production facilities further support market expansion. Additionally, continuous innovation in drug delivery technologies, including sustained and targeted release systems, is boosting excipient utilization. Strong regulatory emphasis on quality standards and traceability, along with increasing investments in research collaborations between academia and industry, are further propelling the adoption of specialized excipients across the Japanese pharmaceutical sector.
Restrain
The Japan Excipients Market faces restraints due to stringent regulatory requirements, high development and compliance costs, and supply chain disruptions post. Brexit. Limited availability of novel excipients and lengthy approval timelines also hinder innovation, while price pressures from generic drug manufacturers restrict overall market profitability and growth potential.
Study on the Supply, Demand, Distribution, and Market Environment of the Japan Excipients Market
The Japan excipients market operates within a highly regulated and quality-focused environment, ensuring consistent supply and safe pharmaceutical production. Supply is driven by domestic chemical and specialty material manufacturers, supported by advanced R&D capabilities and strict compliance with regulatory standards. Demand is fueled by Japan’s strong pharmaceutical industry, aging population, and increasing need for chronic disease treatments, particularly in oral and controlled-release formulations. Distribution follows a structured network involving pharmaceutical companies, contract manufacturers, and authorized distributors, ensuring efficient and compliant delivery. The market environment is characterized by innovation in functional excipients, growing adoption of high-performance polymers, and rising demand for biologics-compatible materials. Additionally, globalization and partnerships with international suppliers enhance product availability, while stringent regulations maintain high quality, shaping a stable yet innovation-driven market landscape in Japan.
Price Analysis and Consumer Behaviour Analysis
Price analysis in the Japan excipients market reflects a tiered pricing structure, ranging from low-cost commodity excipients to high-value customized and specialty formulations. Commodity excipients such as sugars and basic polymers are priced competitively and influenced by global raw material costs, while advanced excipients command premium pricing due to strict quality, regulatory compliance, and technical support requirements. Long-term supply agreements and high switching costs further stabilize pricing dynamics. From a consumer behavior perspective, demand is primarily institutional (pharmaceutical companies and manufacturers) rather than individual-driven. Buyers prioritize quality, regulatory compliance, and supply reliability over price. Once an excipient is approved in a formulation, companies show strong supplier loyalty due to high revalidation costs. Increasing demand for advanced drug delivery systems and biologics is also shifting preferences toward high-performance and multifunctional excipients.
Market Segmentation
The Japan Excipients Market share is classified into product type, distribution channel, and alcohol volume
- The polymers segment dominated the market in 2025 and holds the largest market share, accounting for approximately 32% during the forecast period.
Based on the product type, the excipients market is divided into polymers, alcohols, sugars, minerals, and gelatin. Among these, the polymers segment dominated the market in 2025 and holds the largest market share, accounting for approximately 32% during the forecast period. The polymers segment is driven by rising demand for advanced drug delivery systems in Japan, where polymers enhance stability, solubility, and controlled release. Growth in biologics, generics, and personalized medicines, along with strong pharmaceutical R&D and regulatory support, further accelerates the adoption of functional and multifunctional polymer excipients.
- The tablets segment dominated the market in 2025 and holds the largest market share, accounting for approximately 38% during the forecast period.
Based on the distribution channel, the excipients market is divided into tablets, capsules, parenteral, topical, and liquid dosage forms. Among these, the tablets segment dominated the market in 2025 and holds the largest market share, accounting for approximately 38% during the forecast period. The tablets segment in Japan is driven by high demand for oral solid dosage forms due to their convenience, cost-efficiency, and patient compliance. Strong presence of generic drug manufacturers, increasing chronic disease prevalence, and advanced manufacturing technologies further boost large-scale tablet production, supporting significant market growth.
- The oral dosage segment dominated the market in 2025 and holds the largest market share, accounting for approximately 45% during the forecast period.
Based on the alcohol volume, the excipients market is divided into oral dosage forms, injectable formulations, topical applications, inhalation products, ophthalmic preparations, and nutraceuticals & R&D. Among these, the oral dosage segment dominated the market in 2025 and holds the largest market share, accounting for approximately 45% during the forecast period. The oral dosage forms segment in Japan is driven by strong demand for convenient and non-invasive drug delivery systems, particularly for chronic disease management. High consumption of generics and OTC drugs, well-established pharmaceutical manufacturing infrastructure, and continuous innovation in oral formulations further support sustained market growth.
Recent Development
- In April 2026, Japan’s pharmaceutical excipients sector witnessed increased emphasis on advanced drug delivery technologies, with manufacturers prioritizing excipients that enhance bioavailability, stability, and sustained release. This trend reflected rising demand for patient-centric formulations and complex therapies, as companies invested in multifunctional and controlled-release excipient systems to improve therapeutic outcomes.
- In March 2025, Japan’s Pharmaceuticals and Medical Devices Agency issued updated guidance for oral solid dosage forms, reinforcing the role of excipient functionality and encouraging innovation in formulation design. The policy supported development of specialty excipients for tablets and capsules, reflecting continued focus on advanced ingredients improving drug performance and bioavailability.
Competitive Analysis
The report offers the appropriate analysis of the key organisations/companies involved within the Japan excipients market, along with a comparative evaluation primarily based on their product offering, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborative analysis focusing on the current news and developments of the companies, which includes product development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
Top Key Companies in Japan Excipients Market
- Nippon Fine Chemical Co., Ltd.
- Mitsubishi Chemical Corporation
- Sumitomo Chemical Co., Ltd.
- Asahi Kasei Corporation
- Shin-Etsu Chemical Co., Ltd.
- Others
Key Target Audience
- Market Players
- Investors
- End-users
- Government Authorities
- Consulting and Research Firm
- Venture capitalists
- Value-Added Resellers (VARs)
Market Segment
This study forecasts revenue at the Japan, regional, and country levels from 2020 to 2035. Decision Advisor has segmented the Japan excipients market basedon the below-mentioned segment
Japan Excipients market, By Product Type
- Polymers
- Alcohols
- Sugars
- Minerals
- Gelatin
Japan Excipients Market, By Formulation
- Tablets
- Capsules
- Parenteral
- Topical
- Liquid dosage form
Japan Excipients Market, By Application
- Oral dosage forms
- Injectable formulations
- Topical applications
- Inhalation products
- Ophthalmic preparations
- Nutraceuticals & R&D
Frequently Asked Questions (FAQ)
Q. How is sustainability influencing excipient development in Japan?
A. Sustainability is increasingly shaping excipient innovation in Japan, with companies adopting plant-based, biodegradable, and low-toxicity materials. Regulatory encouragement and environmental goals are pushing manufacturers to reduce carbon footprints and replace synthetic additives, creating demand for green excipients in both pharmaceuticals and nutraceuticals.
Q. What role do contract manufacturing organizations (CMOs) play in this market?
A. CMOs play a crucial role by outsourcing drug production for pharmaceutical firms, increasing demand for reliable and compliant excipients. Their focus on efficiency, scalability, and regulatory adherence drives the need for standardized, high-quality excipients, supporting consistent growth in supply partnerships across Japan.
Q. How does Japan’s aging population impact excipient demand?
A. Japan’s aging population significantly boosts demand for patient-friendly formulations such as easy-to-swallow tablets, controlled-release drugs, and geriatric-focused medicines. This demographic trend drives innovation in excipients that enhance drug stability, taste masking, and extended-release properties for chronic disease management.
Q. What opportunities exist for international excipient suppliers in Japan?
A. International suppliers benefit from Japan’s increasing regulatory harmonization and demand for advanced excipients. Opportunities lie in supplying high-performance, multifunctional ingredients and forming partnerships with local pharmaceutical firms, especially in areas like biologics, personalized medicine, and novel drug delivery systems.
Check Licence
Choose the plan that fits you best: Single User, Multi-User, or Enterprise solutions tailored for your needs.
We Have You Covered
- 24/7 Analyst Support
- Clients Across the Globe
- Tailored Insights
- Technology Tracking
- Competitive Intelligence
- Custom Research
- Syndicated Market Studies
- Market Overview
- Market Segmentation
- Growth Drivers
- Market Opportunities
- Regulatory Insights
- Innovation & Sustainability
Report Details
| Scope | country |
| Pages | 240 |
| Delivery | PDF & Excel via Email |
| Language | English |
| Release | Apr 2026 |
| Access | Download from this page |
