Global Metastatic HR+/HER2? Breast Cancer Market
Global Metastatic HR+/HER2? Breast Cancer Market Size, Share, By Therapy Type (Endocrine Therapy, Targeted Therapy, Chemotherapy, Emerging Therapy, and Others), By Drug Class (CDK4/6 inhibitors, PI3K Inhibitors, mTOR Inhibitors, SERDs, PARP Inhibitors, and ADCs), By Distribution Channel (Hospitals, Oncology Clinics, Specialty Cancer Centers, and Retail & Specialty Pharmacies), and By Region (North America, Europe, Asia-Pacific, Latin America, Middle East, and Africa), Analysis and Forecast 2025 ? 2035.
Report Overview
Table of Contents
Market Snapshot
- Market Size (2025): USD 11.05 Billion
- Projected Market Size (2035): USD 30.58 Billion
- Compound Annual Growth Rate (CAGR): 9.7%
- Largest Regional Market: North America
- Fastest Growing Region: Asia Pacific
- 2nd Largest Region: Europe
- Base Year: 2025
- Historical Period: 2021–2024
- Forecast Period: 2025–2035
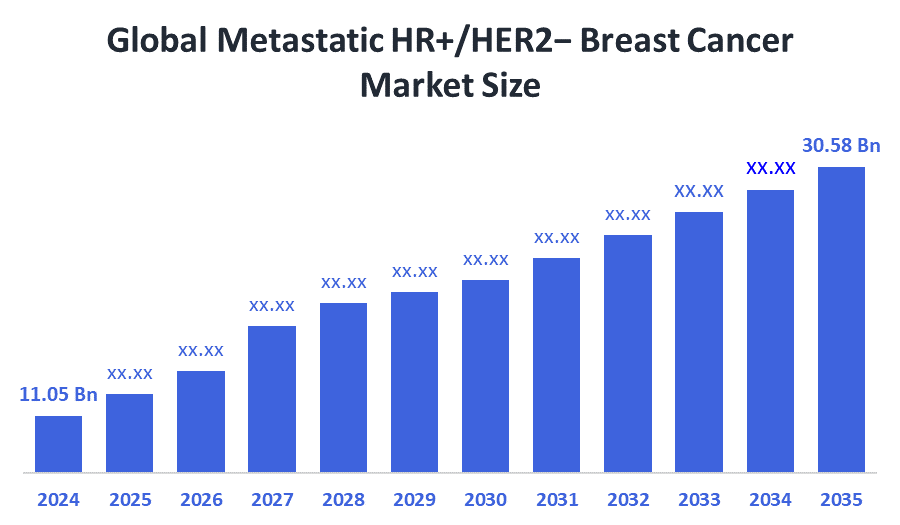
According to Decision Advisors, the Global Metastatic HR+/HER2− Breast Cancer Market Size is expected to grow from USD 11.05billion in 2025 to USD 30.58 billion by 2035, at a CAGR of 9.7% during the forecast period 2025-2035. The global metastatic HR+/HER2− breast cancer market is projected to grow significantly over the next decade due to hormone replacement therapy (HRT), which is forecast to boost the breast cancer treatment market in patients with metastatic HR+/HER2- in the years to come.
Market Overview/ Introduction
The metastatic HR+/HER2− breast cancer market refers to the worldwide commercial market which includes therapeutic products and diagnostic tools and medical services that healthcare professionals use to treat patients with hormone receptor-positive breast cancer who have tumors that express estrogen receptors and progesterone receptors and who also have Human epidermal growth factor receptor 2-negative status and metastatic Stage IV cancer which has extended beyond their breast tissue to other body parts.
The Indian government announced an exemption from basic customs duty on 36 life-saving drugs, which include multiple cancer treatments, to improve access to these medications. India plans to open daycare cancer centers at all district hospitals, with 200 centers scheduled for construction during the 2025-26 fiscal year.
Next-generation oral Selective Estrogen Receptor Degraders (SERDs) like elacestrant (Orserdu) and upcoming candidates are set to become the standard treatment method because they provide improved drug absorption rates and greater treatment ease for patients. The development of biomarker-guided therapies for PIK3CA, AKT1 and ESR1 mutation treatment enables doctors to create customized treatment plans that deliver better second- and third-line treatment outcomes. Strong R&D efforts concentrate on developing treatment methods that combine CDK4/6 inhibitors with Inavolisib (PI3K inhibitor) and other new drugs for advanced endocrine resistance treatment.
- Novartis declared that the US Food and Drug Administration (FDA) approved Kisqali (ribociclib) to be used with an aromatase inhibitor as an adjuvant therapy for patients with hormone receptor-positive and human epidermal growth factor receptor 2-negative (HR+/HER2-) stage II and III early breast cancer (EBC) who face high recurrence risks and include those with node-negative (N0) disease in 2024.
- The US approved DATROWAY (datopotamab deruxtecan-dlnk) in January 2025 to treat adult patients who have unresectable or metastatic breast cancer that shows positive hormone receptors and negative HER2 expression according to IHC 0 IHC 1+ and IHC 2+/ISH- testing and who have already received both endocrine therapy and chemotherapy for their unresectable or metastatic condition.
Notable Insights: -
- North America holds the largest regional market share, approximately 41% in the global Metastatic HR+/HER2− Breast Cancer market.
- Asia Pacific is the fastest-growing region in the global Metastatic HR+/HER2− Breast Cancer market.
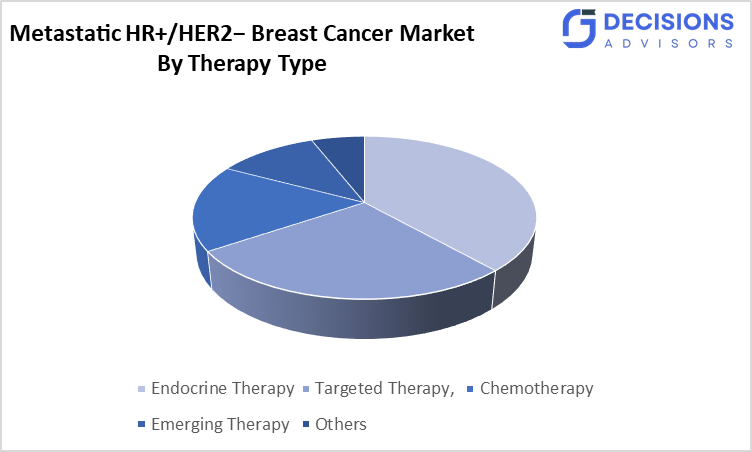
- By therapy type, the endocrine therapy segment held a dominant position of over 42% in terms of market share in 2025.
- By distribution channel, the hospital segment is the dominating one, accounting for over 35% of the global market share in 2025.
- The compound annual growth rate of the global Metastatic HR+/HER2− Breast Cancer market is 9.7%.
- The market is likely to achieve a valuation of USD 30.58 billion by 2035.
What is role of technology in grooming the market?
Technology is playing a critical role in shaping the global metastatic HR+/HER2− breast cancer market by transitioning it from a one-size-fits-all approach to a high-value precision medicine model. AI algorithms now classify breast cancer subtypes and grade tumors with accuracy that rivals human experts, specifically identifying the HER2-low and ultralow segments. Machine learning models analyze multi-omics data to predict which patients will benefit most from specific combinations, such as CDK4/6 inhibitors or oral SERDs, potentially increasing response rates by 20–30%. AI-powered histopathological tools can identify morphological changes linked to ESR1 or PIK3CA mutations directly from standard slides, reducing the need for expensive genetic testing. Telemedicine and remote monitoring platforms are expanding market reach into underserved or rural regions, ensuring patients stay on complex, long-term endocrine therapy regimens.
How is Recent Developments Helping the Market?
Recent developments, such as the expansion of targetable patient populations and the introduction of next-generation targeted classes. The 2025 approval of Enhertu for HER2-low and HER2-ultralow patients has unlocked a massive new market segment previously classified only as HER2-negative. The launch of oral SERDs like Orserdu and Inluriyo, approved in September 2025, provides the first specific options for the 20–40% of patients who develop ESR1 mutations following aromatase inhibitor therapy. The January 2025 approval of Datroway (datopotamab deruxtecan) for HR+/HER2− patients previously treated with chemotherapy provides a high-premium, targeted alternative to standard cytotoxic drugs. The integration of circulating tumor DNA (ctDNA) testing allows for the early detection of resistance mutations (like ESR1) before they appear on scans, enabling a preemptive switch to more effective therapies like Camizestrant.
Market Drivers
The global metastatic HR+/HER2− breast cancer market is propelled by HR+/HER2− is the most common breast cancer subtype, accounting for approximately 70% of all cases. The launch of oral SERDs like Orserdu and Antibody-Drug Conjugates (ADCs) like Enhertu and Datroway provides new options for endocrine-resistant patients. Rapid uptake of high-premium agents like CDK4/6 inhibitors (e.g., Kisqali, Ibrance, Verzenio) has transformed the standard of care. Increasing use of genomic profiling to identify actionable mutations—such as ESR1, PIK3CA, and AKT1—is driving the use of personalized treatments like Truqap and Piqray (alpelisib). drugs moving from later-line metastatic settings into earlier, high-risk adjuvant settings. Over 300 drugs are currently under development, including next-generation ADCs and PROTAC protein degraders.
Restrain
The main restraints on the global metastatic HR+/HER2− breast cancer market include the development of new medical treatments includes CDK4/6 inhibitors and Antibody-Drug Conjugates (ADCs), which require annual expenses that exceed USD 100,000. Patients experience financial toxicity due to high out-of-pocket expenses for healthcare services in India, where more than 90% of the population lacks health insurance coverage. The increasing cost of patented cancer drugs does not result in an equivalent rise in their medical benefits, which creates difficulties for maintaining public health funding.
Competitive Analysis:
The report offers the appropriate analysis of the key organizations/companies involved within the metastatic HR+/HER2− breast cancer market, along with a comparative evaluation primarily based on their product of offering, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborative analysis focusing on the current news and developments of the companies, which includes product development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
Top Companies in Global Metastatic HR+/HER2− Breast Cancer Market
- Pfizer Inc.
- Novartis AG
- Eli Lilly and Company
- AstraZeneca PLC
- Gilead Sciences, Inc.
- F. Hoffmann-La Roche Ltd
- Daiichi Sankyo Company, Limited
- Menarini Group
- Merck & Co., Inc.
- Genentech, Inc.
Government Initiatives
|
Country |
Key Government Initiatives |
|
UK |
The UK’s National Institute for Health and Care Excellence approved capivasertib (AKT inhibitor) for NHS use in targets HR+/HER2− metastatic patients with specific mutations, Decision reversed earlier rejection → now publicly funded. |
|
US |
The U.S. Food and Drug Administration approved datopotamab deruxtecan for Metastatic HR+/HER2− breast cancer. FDA approved trastuzumab deruxtecan (Enhertu) for HR+ metastatic breast cancer with HER2-low or ultra-low expression |
|
Europe |
The European Medicines Agency continues evaluating new oncology combinations and targeted drugs. Regulatory reviews signal faster adoption of next-generation therapies, Increased alignment with FDA approvals. |
Market Segmentation
The metastatic HR+/HER2− breast cancer market share is classified into therapy type, drug class, and distribution channel
- The endocrine therapy segment dominated the market in 2025, and is projected to grow at a substantial CAGR of approximately 42% during the forecast period.
Based on the therapy type, the metastatic HR+/HER2− breast cancer market is divided into endocrine therapy, targeted therapy, chemotherapy, emerging therapy, and others. Among these, the endocrine therapy segment dominated the market in 2025 and is projected to grow at a substantial CAGR of approximately 42% during the forecast period.Due to all HR+ patients receiving endocrine therapy at some stage, which works by blocking estrogen product ion blocking estrogen receptors. Also, typical first-line treatment is endocrine therapy and targeted therapy.
- The CDK4/6 inhibitors segment accounted for the largest share in 2025, and is anticipated to grow at a significant CAGR of approximately 51% during the forecast period.
Based on the drug class, the metastatic HR+/HER2− breast cancer market is divided into CDK4/6 inhibitors, PI3K inhibitors, mTOR inhibitors, SERDs, PARP inhibitors, and ADCs. Among these, the CDK4/6 inhibitors segment accounted for the largest share in 2025, and is anticipated to grow at a significant CAGR of approximately 51% during the forecast period. The segmental growth is driven by the their role as first-line standard of care combined with endocrine therapy. They significantly improve progression-free survival, are widely prescribed across eligible patients, and have strong clinical evidence and adoption. Their broad use, high efficacy, and early-line positioning drive the largest revenue share.
- The hospitals segment dominated the market in 2025, and is projected to grow at a substantial CAGR during the forecast period.
Based on the distribution channel, the metastatic HR+/HER2− breast cancer market is divided into hospitals, oncology clinics, specialty cancer centers, and retail & specialty pharmacies. Among these, the hospitals segment dominated the market in 2025, and is projected to grow at a substantial CAGR during the forecast period. The growth of the segment is driven by the fact that they serve as primary centers for cancer diagnosis, treatment initiation, and management of metastatic cases. They provide access to multidisciplinary care, advanced therapies, and infusion services. Most patients begin treatment in hospitals, driving higher patient volume, drug administration, and overall revenue contribution compared to other settings.
What is the Reason for the Region Dominance?
The dominance of certain regions in the global metastatic HR+/HER2− breast cancer market is primarily driven by high healthcare expenditure, early adoption of novel therapies, and a robust infrastructure for clinical research and precision medicine. The U.S. has the highest per capita spending on cancer care globally—approximately $584 per person, which is twice the median of other high-income countries. North American markets are characterized by the rapid uptake of cutting-edge, high-cost treatments such as CDK4/6 inhibitors (e.g., Kisqali, Ibrance) and next-generation Antibody-Drug Conjugates (ADCs). The region is a leader in implementing precision medicine, with widespread use of biomarker-driven therapies and genomic profiling. Agencies like the U.S. Food and Drug Administration (FDA) provide expedited approval pathways for breakthrough oncology drugs, allowing novel therapies to enter the North American market sooner than in other regions.
Strategies to Implement for Growth of the Market in Non-Leading Regions
In non-leading regions (such as parts of Asia, Latin America, and the Middle East), the metastatic HR+/HER2− breast cancer market faces unique barriers, including high out-of-pocket costs, fragmented diagnostic infrastructure, and varying levels of clinician familiarity with newer targeted therapies. To drive growth in these markets, stakeholders are shifting toward value-based and access-oriented strategies. Implementing local diagnostic pathways is critical to accurately identify patients for targeted therapies like CDK4/6 inhibitors and newer ADCs. In regions like India, many laboratories use non-FDA-approved "homebrew" assays due to cost. Strategic growth requires investment in Quality Control (QC) programs and clinical workshops for pathology specialists to reduce false-negative results. Expanding Actionable Biomarkers: Beyond HR/HER2 status, increasing the availability of PIK3CA and ESR1 mutation testing is necessary to drive the adoption of recent launches like Orserdu and Truqap. In regions with limited infusion center capacity, emphasizing oral combinations can improve patient adherence and reduce the burden on local healthcare infrastructure
Regional Segment Analysis of the Metastatic HR+/HER2− Breast Cancer Market
- North America (U.S., Canada, Mexico)
- Europe (Germany, France, U.K., Italy, Spain, Rest of Europe)
- Asia-Pacific (China, Japan, India, Rest of APAC)
- South America (Brazil and the Rest of South America)
- The Middle East and Africa (UAE, South Africa, Rest of MEA)
North America is anticipated to hold the largest share of the metastatic HR+/HER2− breast cancer market over the predicted timeframe.
North America is anticipated to hold the largest share of the metastatic HR+/HER2− breast cancer market over the predicted timeframe. This market is accounting about approximately 41% of the total market share. The dominance of this market is driven by the high adoption of advanced targeted therapies, strong healthcare infrastructure and oncology centers, favorable reimbursement systems, presence of major pharmaceutical companies, early access to newly approved drugs, and high awareness and diagnosis rates. DATROWAY has been approved in the U.S. for the treatment of adult patients with unresectable or metastatic hormone receptor (HR) positive, HER2 negative (IHC 0, IHC 1+ or IHC 2+/ISH-) breast cancer who have received prior endocrine-based therapy and chemotherapy for unresectable or metastatic disease in 2025.
Asia Pacific is expected to grow at a rapid CAGR in the metastatic HR+/HER2− breast cancer market during the forecast period. Asia Pacific is poised to be the fastest-growing region in the global metastatic HR+/HER2− breast cancer market with a remarkable CAGR of 11% during the forecast period of 2025 to 2035. Due to the rapidly increasing breast cancer incidence, large patient population (e.g., China, India), improving healthcare infrastructure, rising access to targeted therapies, growing healthcare spending, expanding awareness and early diagnosis programs. Government initiatives to strengthen oncology care, rising clinical trial activity, and faster regulatory approvals are boosting market expansion. The increasing presence of global pharmaceutical companies and local manufacturers improves drug availability.
Europe is the 2nd largest region to grow in the Metastatic HR+/HER2− Breast Cancer market during the period. Europe is the second-largest region in the Metastatic HR+/HER2− Breast Cancer market, accounting for around 25–30% share. Growth is driven by strong healthcare infrastructure, widespread access to advanced targeted therapies, and supportive reimbursement systems across countries. Regulatory support from the European Medicines Agency and increasing clinical research activity further enhance treatment adoption, contributing to steady market expansion during the forecast period.
Future Market Trends in Global Metastatic HR+/HER2− Breast Cancer Market: -
- Shift Toward Precision Medicine
Shift toward precision medicine is transforming the metastatic HR+/HER2− breast cancer market, with over 40–50% of patients now undergoing biomarker testing (e.g., PIK3CA, ESR1). Targeted therapies improve progression-free survival by 30–50%, driving adoption. This trend is expected to contribute to a ~10% CAGR, significantly boosting personalized treatment demand globally.
- Expansion of Antibody-Drug Conjugates (ADCs)
Expansion of antibody-drug conjugates (ADCs) is accelerating in metastatic HR+/HER2− breast cancer, with ADCs showing ~40–60% response rates in later-line settings. Their market share is expected to exceed 15–20% by 2030, growing at ~12–15% CAGR. Increasing approvals and earlier-line use are significantly boosting adoption and revenue contribution globally.
- Rise of Oral Therapies
Rise of oral therapies is transforming the metastatic HR+/HER2− breast cancer market, with over 60% of new drugs being oral formulations. These therapies improve patient adherence by ~20–30% and reduce hospital visits. The oral segment is projected to grow at ~10–12% CAGR, driven by convenience, long-term use, and expanding pipelines.
Recent Development
In September 2025, Eli Lilly and Company announced that the U.S. Food and Drug Administration (FDA) has approved Inluriyo (imlunestrant, 200 mg tablets), an oral estrogen receptor antagonist, for the treatment of adults with estrogen receptor-positive (ER+), human epidermal growth factor receptor 2-negative (HER2–), ESR1-mutated advanced or metastatic breast cancer (MBC) whose disease progressed after at least one line of endocrine therapy (ET).
Market Segment
This study forecasts revenue at global, regional, and country levels from 2020 to 2035. Decision Advisors has segmented the metastatic HR+/HER2− breast cancer market based on the below-mentioned segments:
Global Metastatic HR+/HER2− Breast Cancer Market, By Therapy Type
- Endocrine Therapy
- Targeted Therapy
- Chemotherapy
- Emerging Therapy
- Others
Global Metastatic HR+/HER2− Breast Cancer Market, By Drug Class
- CDK4/6 inhibitors
- PI3K Inhibitors
- mTOR Inhibitors
- SERDs
- PARP Inhibitors
- ADCs
Global Metastatic HR+/HER2− Breast Cancer Market, By Distribution Channel
- Hospitals
- Oncology Clinics
- Specialty Cancer Centers
- Retail & Specialty Pharmacies
Global Metastatic HR+/HER2− Breast Cancer Market, By Regional Analysis
- North America
- US
- Canada
- Mexico
- Europe
- Germany
- UK
- France
- Italy
- Spain
- Russia
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- Australia
- Rest of Asia Pacific
- South America
- Brazil
- Argentina
- Rest of South America
- Middle East & Africa
- UAE
- Saudi Arabia
- Qatar
- South Africa
- Rest of the Middle East & Africa
Frequently Asked Questions (FAQ)
Q: What are the key treatment options available?
Treatment includes endocrine therapy, targeted therapy (like CDK4/6, PI3K, and mTOR inhibitors), and chemotherapy in advanced stages. Combination therapies are commonly used to improve survival outcomes. Treatment choice depends on patient condition, prior therapies, and genetic mutations identified through precision medicine approaches.
Q: What is the future outlook of this market?
The market is expected to grow steadily due to ongoing research, novel drug approvals, and increasing adoption of personalized medicine. Emerging therapies, including next-generation inhibitors and immunotherapies, are likely to improve outcomes. Expanding healthcare access and early diagnosis initiatives will further support long-term market expansion
Q: What role does personalized medicine play in this market?
Personalized medicine is crucial, as treatment decisions are increasingly based on genetic profiling and biomarkers. This approach improves treatment effectiveness and reduces unnecessary side effects. Advances in molecular diagnostics enable clinicians to tailor therapies, driving demand for targeted drugs and enhancing overall patient outcomes in metastatic HR+/HER2− breast cancer
Q: How do clinical trials impact market development?
Clinical trials are essential for introducing new therapies and expanding treatment options. They help evaluate safety and efficacy, leading to regulatory approvals. Ongoing trials in targeted therapies and combination regimens drive innovation, attract investments, and shape the competitive landscape of the metastatic breast cancer market.
Q: What trends are shaping the market?
Key trends include the rise of combination therapies, increased use of CDK4/6 inhibitors, and focus on overcoming drug resistance. Digital health tools and real-world evidence are gaining importance. Additionally, collaborations between biotech firms and pharmaceutical companies are accelerating drug development and commercialization efforts globally.
Q: How does healthcare policy influence the market?
Government policies, reimbursement frameworks, and regulatory approvals significantly impact market growth. Favorable reimbursement increases patient access to expensive therapies, while strict regulations can delay product launches. Policy support for cancer research and funding also plays a vital role in advancing treatment innovations.
Check Licence
Choose the plan that fits you best: Single User, Multi-User, or Enterprise solutions tailored for your needs.
We Have You Covered
- 24/7 Analyst Support
- Clients Across the Globe
- Tailored Insights
- Technology Tracking
- Competitive Intelligence
- Custom Research
- Syndicated Market Studies
- Market Overview
- Market Segmentation
- Growth Drivers
- Market Opportunities
- Regulatory Insights
- Innovation & Sustainability
Report Details
| Scope | Global |
| Pages | 210 |
| Delivery | PDF & Excel via Email |
| Language | English |
| Release | Mar 2026 |
| Access | Download from this page |
