Global Microbiome Therapeutics Market
Global Microbiome Therapeutics Market Size, Share, By Type (Transplantation and Drugs), By Disease Indication (Clostridium Difficile Infection (CDI), Oncology-Support Settings, Vaginal Health, Dermatology, Inflammatory Bowel Disease, and Others), By Route of Administration (Rectal, Oral, Topical, and Others), By Distribution Channel (Hospital Pharmacies, Specialty Pharmacies, and Others), and By Region (North America, Europe, Asia-Pacific, Latin America, Middle East & Africa), Analysis and Forecast 2026-2035
Report Overview
Table of Contents
Market Snapshot
- Global Microbiome Therapeutics Market Size (2025): USD 289.8Million
- Projected Global Microbiome Therapeutics Market Size (2035): USD 5789.1Million
- Global Microbiome Therapeutics Market Compound Annual Growth Rate (CAGR): 34.91%
- Largest Regional Market: North America
- Fastest Growing Region: Asia-Pacific
- Market 3rd Largest Region: Europe
- Base Year: 2025
- Historical Period: 2021–2024
- Forecast Period: 2026–2035
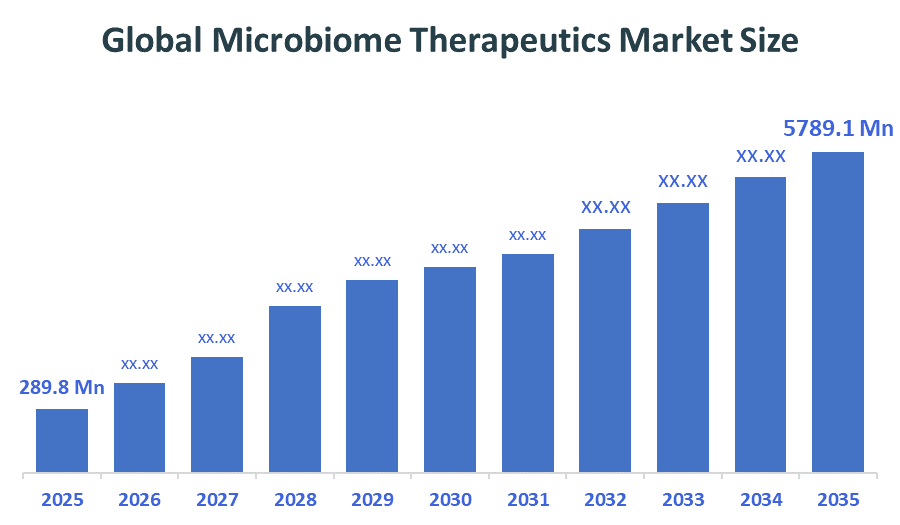
According to Decision Advisors, the Global Microbiome Therapeutics Market Size is expected to grow from USD 289.8 Million in 2025 to USD 5789.1 Million by 2035, at a CAGR of 34.91% during the forecast period 2026-2035. The global microbiome therapeutics market is projected to grow significantly over the next decade, driven by rising chronic diseases, advanced therapeutic developments like fecal microbiota transplantation and engineered probiotics, and increased understanding of the microbiomes role health.
Market Overview/ Introduction
The Microbiome Therapeutics Market refers to the global industry that develops and sells treatments that change the human microbiome for disease prevention and control, and disease treatment. Microbiome therapeutics serve as medical treatments that use live biotherapeutic products, fecal microbiota transplants, and microbiome-derived compounds to restore microbial balance in the body. The market experiences strong expansion because chronic diseases become more common, research on gut health increases, and regulatory frameworks provide support. The future provides new possibilities through personalized medicine and precision microbiome therapies and market development in oncology, dermatology, and metabolic disorders. The market experiences substantial global growth because sequencing technologies become more advanced, clinical trials achieve higher success rates, and pharmaceutical companies increase their funding.
- In the U.S. Food and Drug Administration (FDA) Microbiome Research Program, the U.S. government is currently advancing regulatory science for microbiome-based products, including live biotherapeutics and fecal microbiota transplantation. This initiative supports safety evaluation, clinical research, and product standardization, accelerating microbiome therapeutic development.
- The European Commission Biotech Act is currently being developed to strengthen biotechnology innovation, including microbiome therapeutics, through improved regulatory frameworks, stakeholder collaboration, and faster patient access to advanced biological treatments.
Notable Insights: -
- North America is anticipated to hold the largest share of approximately 45% in the microbiome therapeutics market over the forecast period.
- Asia Pacific is expected to grow at a rapid CAGR of approximately 35% in the microbiome therapeutics market during the forecast period.
- The drugs segment dominated the market in 2025 and is anticipated to grow at a significant CAGR of approximately 31% during the forecast period.
- The clostridium difficile infection (CDI) segment dominated the market in 2025 and is anticipated to grow at a significant CAGR of approximately 35% during the forecast period.
- The oral segment dominated the market in 2025 and is anticipated to grow at a significant CAGR of approximately 65% during the forecast period.
- The hospital pharmacies segment dominated the market in 2025 and is anticipated to grow at a significant CAGR of approximately 30% during the forecast period.
- The compound annual growth rate of the Global Microbiome Therapeutics Market is 34.91%.
- The market is likely to achieve a valuation of USD 5789.1Million by 2035.
What is role of technology in grooming the market?
Technology plays a critical role in shaping the global microbiome therapeutics market by enabling deeper understanding, precise analysis, and scalable development of microbiome-based treatments. Advanced sequencing technologies, such as next-generation sequencing (NGS), allow detailed identification of microbial communities and their functions. Artificial intelligence and bioinformatics tools help analyze complex microbiome data, accelerating drug discovery and target identification. Automation and high-throughput screening platforms improve efficiency in developing live biotherapeutic products. Additionally, innovations in synthetic biology enable the engineering of targeted microbial strains for therapeutic use. Improved manufacturing technologies ensure quality, consistency, and large-scale production. Overall, technological advancements are driving innovation, reducing development timelines, and supporting the commercialization of effective microbiome therapeutics globally.
Market Drivers
The Global Microbiome Therapeutics Market is presently expanding because the number of people who develop autoimmune diseases, metabolic disorders, and gastrointestinal tract diseases is increasing. The existing treatment methods for these diseases require the development of new therapeutic approaches. Scientists have developed an understanding of gut microbiomes, which enables them to create drugs that target specific pathogens and metabolic diseases. Biopharmaceutical companies currently receive increased funding, which enables them to collaborate on research initiatives that will result in new drug discovery. The market expansion occurs because regulatory authorities have established quicker procedures to approve fecal microbiota transplantation (FMT) and Clostridioides difficile treatment methods.
Restrain
The Global Microbiome Therapeutics Market faces restraints due to regulatory uncertainty, as clear guidelines for live biotherapeutic products are still evolving across regions. High research and development costs, complex microbiome variability among individuals, and challenges in standardization limit scalability. Additionally, limited clinical evidence for some indications and concerns regarding long-term safety hinder widespread adoption and commercialization of microbiome-based therapies.
Competitive Analysis:
The report offers the appropriate analysis of the key organizations/companies involved within the global microbiome therapeutics market, along with a comparative evaluation primarily based on their product offerings, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborative analysis focusing on the current news and developments of the companies, which includes product development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
Top Companies in Global Microbiome Therapeutics Market
- Seres Therapeutics
- Vedanta Biosciences
- Ferring Pharmaceuticals
- Rebiotix Inc.
- 4D Pharma plc
- Second Genome Inc.
- Synlogic Inc.
- Enterome SA
- Finch Therapeutics Group Inc.
- Assembly Biosciences Inc.
- AOBiome LLC
- MaaT Pharma
Government Initiatives
|
Country |
Key Government Initiatives |
|
US |
The U.S. government, through agencies like the NIH and FDA, is actively supporting microbiome therapeutics via funding programs and regulatory pathways for live biotherapeutic products. Recent initiatives emphasize fast-track approvals, clinical trial expansion, and microbiome-based drug standardization, accelerating commercialization and innovation in microbiome therapies. |
|
Europe |
The EU has strengthened regulatory frameworks under the Substances of Human Origin (SoHO) regulation, specifically addressing microbiome-based therapies. These policies support clinical development, safety standardization, and cross-border research funding, enabling faster adoption and commercialization of microbiome therapeutics across member states. |
|
India |
India is advancing microbiome research through national programs like the National Action Plan on Antimicrobial Resistance and government-backed microbiome and probiotic research initiatives. These efforts promote microbiome-based interventions to combat AMR and improve public health outcomes. |
Study on the Supply, Demand, Distribution, and Market Environment of the Microbiome Therapeutics Market
The Global Microbiome Therapeutics Market is shaped by a complex interplay of supply, demand, distribution, and market environment factors. Supply depends on advanced bioprocessing capabilities and availability of high-quality microbial strains, culture media, and biological raw materials such as donor stool (for FMT), bacterial consortia, and fermentation inputs. Demand is rising due to increasing prevalence of chronic and gastrointestinal diseases and growing interest in personalized medicine. Distribution relies on specialized cold-chain logistics and hospital or clinical channels to maintain product viability. The market environment is influenced by evolving regulatory frameworks, strong R&D investments, and partnerships between biotech and pharmaceutical companies. Overall, advancements in manufacturing and sourcing of reliable raw materials are crucial for scalable and consistent microbiome therapeutic production.
Price Analysis and Consumer Behaviour Analysis
Price analysis and consumer behaviour in the Global Microbiome Therapeutics Market reflect a premium, innovation-driven landscape. The high cost of microbiome therapies is driven by expensive R&D, complex manufacturing, and stringent regulatory requirements, with treatments such as fecal microbiota transplantation costing thousands per dose. At the same time, companies are working to reduce prices to improve accessibility and adoption. Consumer behaviour shows increasing preference for natural, preventive, and personalized healthcare solutions, especially for gut health and chronic diseases. Rising awareness of the microbiome’s role in overall health and demand for safer alternatives to conventional drugs are boosting acceptance. Overall, willingness to adopt innovative therapies is increasing despite high costs
Market Segmentation
The Microbiome Therapeutics Market share is classified into type, disease indication, route of administration and distribution channel
- The drugs segment dominated the market in 2025 and is anticipated to grow at a significant CAGR of approximately 31% during the forecast period.
Based on the type, the microbiome therapeutics market is divided into transplantation and drugs. Among these,the drugs segment dominated the market in 2025 and is anticipated to grow at a significant CAGR of approximately 31% during the forecast period. The drugs segment dominated due to increasing development and approval of microbiome-based pharmaceuticals, including live biotherapeutic products. Rising clinical trials, strong pipeline growth, and higher adoption of targeted therapies are expected to drive the segment’s significant CAGR of approximately 31% during the forecast period.
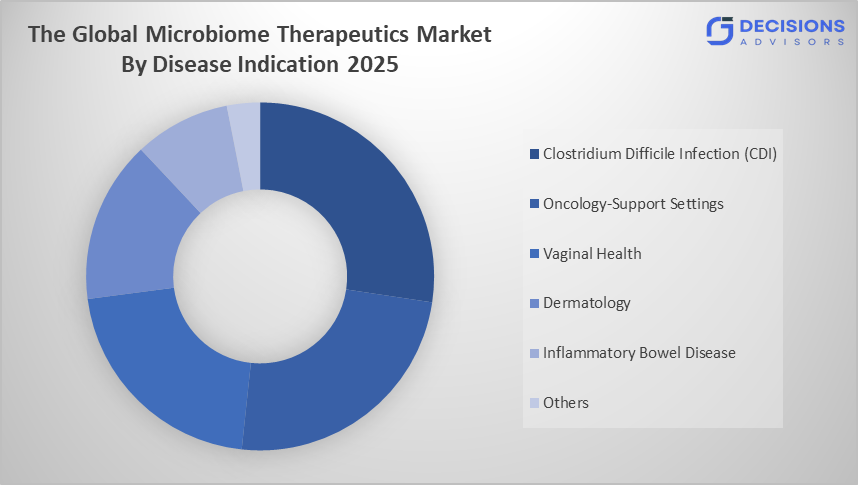
- The clostridium difficile infection (CDI) segment dominated the market in 2025 and is anticipated to grow at a significant CAGR of approximately 35% during the forecast period.
Based on the disease indication, the microbiome therapeutics market is divided into clostridium difficile infection (CDI), oncology-support settings, vaginal health, dermatology, inflammatory bowel disease, and others. Among these, the clostridium difficile infection (CDI) segment dominated the market in 2025 and is anticipated to grow at a significant CAGR of approximately 35% during the forecast period. The growth of the clostridium difficile infection (CDI) segment is growing due to high disease prevalence and strong clinical success of microbiome-based treatments such as fecal microbiota transplantation and live biotherapeutics. Increasing recurrence rates and effective therapeutic outcomes are expected to drive a significant CAGR of approximately 35% during the forecast period.
- The oral segment dominated the market in 2025 and is anticipated to grow at a significant CAGR of approximately 65% during the forecast period.
Based on the route of administration, the microbiome therapeutics market is divided into rectal, oral, topical, and others. Among these, the oral segment dominated the market in 2025 and is anticipated to grow at a significant CAGR of approximately 65% during the forecast period. The growth of the oral segment is growing due to its high patient compliance, ease of administration, and non-invasive nature compared to other delivery methods. Advancements in capsule-based microbiome formulations and increasing preference for convenient treatments.
- The hospital pharmacies segment dominated the market in 2025 and is anticipated to grow at a significant CAGR of approximately 30% during the forecast period.
Based on the distribution channel, the microbiome therapeutics market is divided into hospital pharmacies, specialty pharmacies, and others. Among these, the hospital pharmacies segment dominated the market in 2025 and is anticipated to grow at a significant CAGR of approximately 30% during the forecast period. The growth of the hospital pharmacies segment is growing due to the increasing administration of microbiome-based treatments in clinical settings and hospitals. Higher patient inflow, availability of specialized care, and proper storage facilities.
Strategies to Implement for Growth of the Market in Non-Leading Regions
To drive growth of the Global Microbiome Therapeutics Market in non-leading regions, companies should focus on increasing awareness through education campaigns on microbiome health and its clinical benefits. Expanding collaborations with local healthcare providers and research institutions can strengthen clinical adoption and regional innovation. Cost-reduction strategies, including localized manufacturing and scalable production technologies, are essential to improve affordability. Governments and private players should support funding and regulatory clarity to encourage market entry. Developing region-specific therapies based on local microbiome profiles can enhance effectiveness and acceptance. Additionally, improving distribution networks, including cold-chain logistics and digital health platforms, will ensure better accessibility. Strategic partnerships, clinical trials in emerging markets, and investment in infrastructure are key to unlocking growth potential in these regions.
Regional Segment Analysis of the Microbiome Therapeutics Market
- North America (U.S., Canada, Mexico)
- Europe (Germany, France, U.K., Italy, Spain, Rest of Europe)
- Asia-Pacific (China, Japan, India, Rest of APAC)
- South America (Brazil and the Rest of South America)
- The Middle East and Africa (UAE, South Africa, Rest of MEA)
North America is anticipated to hold the largest share of approximately 45% the microbiome therapeutics market over the forecast period.
North America is anticipated to hold the largest share of approximately 45% the microbiome therapeutics market over the forecast period. The dominance of this region is primarily driven by strong presence of leading biotech companies, advanced healthcare infrastructure, and significant R&D investments. Additionally, supportive regulatory frameworks, increasing clinical trials, and high awareness of microbiome-based therapies are driving rapid adoption and market growth in the region.
Asia Pacific is expected to grow at a rapid CAGR of approximately 35% in the microbiome therapeutics market during the forecast period.
Asia Pacific is expected to grow at a rapid CAGR of approximately 35% in the microbiome therapeutics market during the forecast period. This growth is driven by increasing healthcare investments, rising awareness of gut health, and expanding biotechnology research. Additionally, large patient populations, improving healthcare infrastructure, and supportive government initiatives are driving clinical research and adoption of microbiome-based therapies across the region.
Europe is the 3rd largest region to grow in the microbiome therapeutics market during the period.
Europe is the third-largest region in the microbiome therapeutics market due to strong government support, well-established healthcare systems, and increasing investment in biotechnology research. Additionally, supportive regulatory frameworks, growing clinical trials, and rising awareness of microbiome-based therapies are contributing to steady market growth across the region.
Future Market Trends in Global Microbiome Therapeutics Market: -
1. Growth of Personalized Microbiome Therapies
Personalized medicine is emerging as a key trend, with therapies tailored to individual microbiome profiles using advanced sequencing and bioinformatics tools. Increasing demand for targeted treatments and better clinical outcomes is driving this shift, enabling precise disease management and improving patient response rates significantly.
2. Expansion into New Therapeutic Areas
Microbiome therapeutics are expanding beyond gastrointestinal diseases into oncology, neurology, and metabolic disorders. Growing scientific evidence linking the microbiome to immune response and brain health is increasing applications, significantly widening the patient base and boosting market growth potential across multiple disease segments.
3. Advancements in Live Biotherapeutics & Manufacturing
Next-generation live biotherapeutic products and improved manufacturing technologies are shaping the market. Increased investment, standardization of microbial consortia, and scalable production methods are enhancing product efficacy, safety, and accessibility, supporting faster commercialization and large-scale adoption globally.
Recent Development
- In March 2026, MaaT Pharma announced that its microbiome therapy MaaT013 (Xervyteg) is under European Medicines Agency (EMA) review, positioning it as a potential first-in-class microbiome drug in oncology, signaling major regulatory progress in Europe.
- In January 2026, the U.S. Food and Drug Administration granted Investigational New Drug (IND) clearance to a microbiome-based therapy (MH002), enabling a Phase 2b clinical trial for ulcerative colitis. This highlights strong regulatory support and accelerating clinical development in microbiome therapeutics.
- In January 2026, a new microbiome research center was launched to focus on gut microbiome-based therapeutics for diseases like IBD, obesity, and liver disorders, emphasizing country-specific microbiome data for precision medicine and future therapeutic development.
How is Recent Developments Helping the Market?
Recent developments are significantly accelerating growth in the global microbiome therapeutics market by improving clinical validation, regulatory clarity, and product innovation. Advancements such as successful Phase 2 and Phase 3 clinical trials, increasing FDA and EMA approvals for live biotherapeutic products, and progress in fecal microbiota transplantation are boosting confidence among stakeholders. Strategic collaborations between biotech firms and pharmaceutical companies are enhancing research capabilities and speeding up commercialization. Additionally, innovations in sequencing technologies, artificial intelligence, and synthetic biology are enabling precise microbiome targeting and personalized treatments. Growing investment and expansion of microbiome pipelines are further strengthening the market, leading to faster product development, improved treatment outcomes, and increased global adoption of microbiome-based therapies.
Market Segment
This study forecasts revenue at global, regional, and country levels from 2020 to 2035. Decision Advisors has segmented the Microbiome therapeutics market based on the below-mentioned segments:
Global Microbiome Therapeutics Market, By Type
- Transplantation
- Drugs
Global Microbiome Therapeutics Market, By Disease Indication
- Clostridium Difficile Infection (CDI)
- Oncology-Support Settings, Vaginal Health
- Dermatology
- Inflammatory Bowel Disease
- Others
Global Microbiome Therapeutics Market, By Route of Administration
- Rectal
- Oral
- Topical
- Others
Global Microbiome Therapeutics Market, By Distribution Channel
- Hospital Pharmacies
- Specialty Pharmacies
- Others
Global Microbiome Therapeutics Market, By Regional Analysis
- North America
- US
- Canada
- Mexico
- Europe
- Germany
- UK
- France
- Italy
- Spain
- Russia
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- Australia
- Rest of Asia Pacific
- South America
- Brazil
- Argentina
- Rest of South America
- Middle East & Africa
- UAE
- Saudi Arabia
- Qatar
- South Africa
- Rest of the Middle East & Africa
Frequently Asked Questions (FAQ)
Q What are the key challenges in commercializing microbiome therapeutics?
A. The commercialization of microbiome therapeutics is challenged by complex regulatory pathways, variability in patient microbiomes, and difficulties in standardizing live biological products. Additionally, high manufacturing costs and limited long-term clinical data create barriers for large-scale adoption and market penetration.
Q. How do partnerships impact the microbiome therapeutics market?
A. Strategic partnerships between biotechnology firms and pharmaceutical companies enhance research capabilities, funding access, and clinical trial expansion. These collaborations accelerate drug development timelines, improve technological innovation, and increase the chances of successful commercialization of microbiome-based therapies globally.
Q. What role does patient awareness play in market growth?
A. Increasing patient awareness about gut health and the role of microbiomes in overall wellness is significantly driving demand. Educational initiatives and digital health platforms are encouraging early diagnosis and acceptance of microbiome-based therapies, boosting adoption rates across both developed and emerging markets.
Q. How is the competitive landscape evolving in this market?
A. The competitive landscape is becoming more dynamic with the entry of new biotech startups, increased venture capital investments, and rising mergers and acquisitions. Companies are focusing on pipeline expansion, innovative product development, and geographic expansion to strengthen their market position and gain competitive advantage.
Check Licence
Choose the plan that fits you best: Single User, Multi-User, or Enterprise solutions tailored for your needs.
We Have You Covered
- 24/7 Analyst Support
- Clients Across the Globe
- Tailored Insights
- Technology Tracking
- Competitive Intelligence
- Custom Research
- Syndicated Market Studies
- Market Overview
- Market Segmentation
- Growth Drivers
- Market Opportunities
- Regulatory Insights
- Innovation & Sustainability
Report Details
| Scope | Global |
| Pages | 240 |
| Delivery | PDF & Excel via Email |
| Language | English |
| Release | Apr 2026 |
| Access | Download from this page |
