Global Narcolepsy Market
Global Narcolepsy Market Size, Share, and COVID-19 Impact Analysis, By Type of Narcolepsy (Type 1 Narcolepsy, and Type 2 Narcolepsy), By Treatment (Narcolepsy with Cataplexy, Narcolepsy Without Cataplexy), By Product (Central Nervous System Stimulants, Sodium Oxybate, Selective Serotonin Reuptake Inhibitor, Tricyclic Antidepressants, and Others), and By Region (North America, Europe, Asia-Pacific, Latin America, Middle East, and Africa), Analysis and Forecast 2025-2035
Report Overview
Table of Contents
Global Narcolepsy Market Size Insights Forecasts to 2035
- The Global Narcolepsy Market Size Was Estimated at USD 3.52 Billion in 2024
- The Market Size is Expected to Grow at a CAGR of around 6.65 % from 2025 to 2035
- The Worldwide Narcolepsy Market Size is Expected to Reach USD 7.15 Billion by 2035
- North America is expected to grow the fastest during the forecast period.
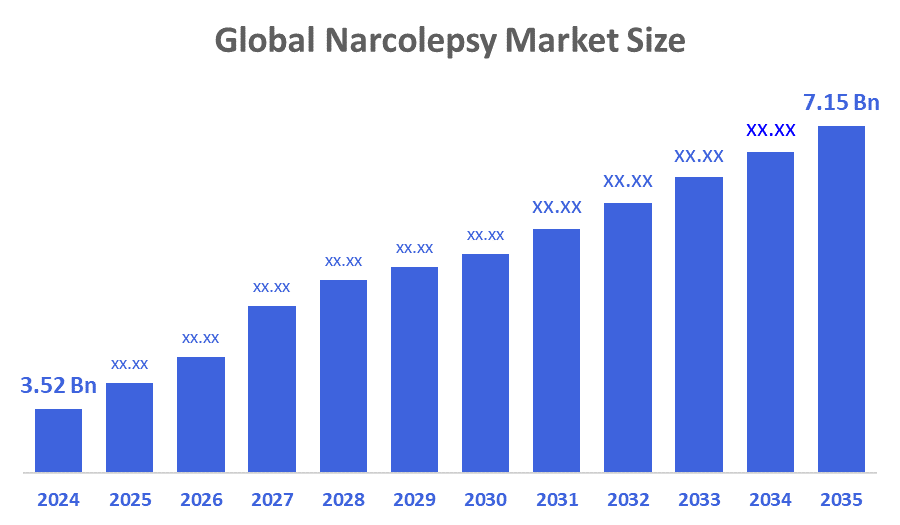
According to a research report published by Decisions Advisors and Consulting, The Global Narcolepsy Market Size Was Worth Around USD 3.52 Billion In 2024 And Is Predicted To Grow To Around USD 7.15 Billion By 2035 With A Compound Annual Growth Rate (CAGR) Of 6.65 % From 2025 To 2035. Growing awareness of sleep disorders, higher diagnosis rates, and a move toward individualised and successful treatment plans are the main factors driving the growing demand for narcolepsy treatments. Among the main factors propelling market expansion are the rising incidence of illness, rising R&D expenditures and activities, and the launch of innovative medicinal products.
Market Overview
The narcolepsy market refers to the pharmaceutical and healthcare sectors devoted to the diagnosis, treatment, and management of narcolepsy, a chronic neurological disorder marked by excessive daytime sleepiness, cataplexy, sleep paralysis, and disturbed nighttime sleep. Narcolepsy constitutes a lifelong disorder that affects the brain and causes disturbances in the normal patterns of sleep and wakefulness because of the destruction or impairment of orexin-producing neurons in the hypothalamus. The biological deficiency now present in the brain blocks natural processes that should create balanced periods of wakefulness and sleep, thus resulting in excessive daytime sleepiness, cataplexy and disrupted nighttime sleep. The clinical condition of narcolepsy occurs at a low frequency but results in major daily functional impairments that affect cognitive abilities and overall life quality. The field of neurobiology, together with sleep research, has developed better molecular diagnostic methods, which now allow doctors to identify the disease and create specific treatments using orexin-based drugs. The medical field faces persistent issues because people do not understand narcolepsy, and patients experience prolonged wait times for their diagnosis. The field of narcolepsy research remains active because it provides new developments that will influence sleep medicine and individualised treatment methods.
According to the World Health Organisation (WHO), which considers narcolepsy to be a rare neurological disorder, new estimates indicate that it affects about 3 million people globally. Despite rates varying by region due to variations in diagnosis and reporting, the global prevalence is typically reported to be 25–50 cases per 100,000 people.
Alkermes announced a $2.1 billion acquisition of Avadel Pharmaceuticals, marking its entry into the sleep medicine market. The deal centres on Lumryz, Avadel’s extended-release sodium oxybate therapy for narcolepsy. This acquisition positions Alkermes as a major player in sleep medicine, combining a commercially successful oxybate therapy with a pipeline of orexin-targeted drugs. It also reflects the growing competition in narcolepsy treatments.
Report Coverage
This research report categorizes the narcolepsy market based on various segments and regions, forecasts revenue growth, and analyzes trends in each submarket. The report analyses the key growth drivers, opportunities, and challenges influencing the narcolepsy market. Recent market developments and competitive strategies, including expansion, product launches, development, partnerships, mergers, and acquisitions, have been incorporated to illustrate the competitive landscape in the market. The report strategically identifies and profiles the key market players and analyzes their core competencies in each sub-segment of the narcolepsy market.
Driving Factors
The narcolepsy market is going through a significant transformation because of new treatment methods and a better understanding of the condition. The increased public knowledge about the condition will lead to getting diagnosed because they will seek out effective treatment options. The research dedicated to understanding narcolepsy's fundamental causes will create new treatment methods that will boost patient health results and life quality. The narcolepsy market today shows two main changes because of new drug treatments and the rising popularity of custom-tailored medical solutions. The technique of matching treatments with specific patient characteristics shows potential benefits because it will increase treatment success rates while reducing treatment-related problems. The introduction of technology for narcolepsy treatment will transform patient treatment because it includes mobile apps that track symptoms and telemedicine services. The market demands that stakeholders continue their watch over new developments while they change their business plans to satisfy the specific requirements of this particular patient group.
Alkermes officially launched its Phase II Vibrance-2 trial of ALKS 2680 for narcolepsy type 2 (NT2). This is a major step in developing new therapies for NT2, which is characterised by severe daytime sleepiness but lacks the cataplexy seen in narcolepsy type 1.
Restraining Factors
The application of narcolepsy treatments is restricted by safety issues, legal restrictions, and expensive medication prices. Despite novel treatments, patient trust, access, and adherence are impacted by cardiovascular and gastrointestinal risks, delayed approvals, inadequate monitoring in emerging markets, and financial burdens.
Market Segmentation
The narcolepsy market share is classified into type, treatment, and product.
- The type 1 narcolepsy segment accounted for the largest market share in 2024 and is anticipated to grow at a substantial CAGR during the forecast period.
Based on the type, the narcolepsy market is segmented into type 1 narcolepsy, and type 2 narcolepsy. Among these, the type 1 narcolepsy segment accounted for the largest market share in 2024 and is anticipated to grow at a substantial CAGR during the forecast period. This segment growth is driven by the increased incidence of cataplexy and narcolepsy worldwide. In addition to the severe daytime sleepiness and cataplexy symptoms that greatly affect quality of life, Type 1 treatment is given priority by clinicians. Furthermore, therapies for Type 1 are the main focus of current research and product launches, strengthening their market share.
For instance, Takeda reported breakthrough Phase 3 results for its investigational drug Oveporexton (TAK-861), a first-in-class oral orexin receptor 2 (OX2R)-selective agonist, in patients with narcolepsy type 1 (NT1).
- The narcolepsy with cataplexy segment accounted for the highest market revenue in 2024 and is anticipated to grow at a notable CAGR during the forecast period.
Based on the treatment, the narcolepsy market is divided into narcolepsy with cataplexy, narcolepsy without cataplexy. Among these, the narcolepsy with cataplexy segment accounted for the highest market revenue in 2024 and is anticipated to grow at a notable CAGR during the forecast period. The narcolepsy therapeutics market shows its highest market share in cases of narcolepsy with cataplexy because this condition has distinct features. The clinical presentation of this narcolepsy type shows higher severity because patients experience sudden muscle weakness that their emotional reactions trigger. Patients who experience cataplexy need more advanced treatment methods, which include sodium oxybate-based medications.
For instance, the FDA approved WAKIX (pitolisant) for the treatment of cataplexy in pediatric patients aged 6 years and older with narcolepsy. This expands its use beyond adults, making it the first and only FDA-approved treatment for both pediatric and adult narcolepsy patients with or without cataplexy that is not scheduled as a controlled substance.
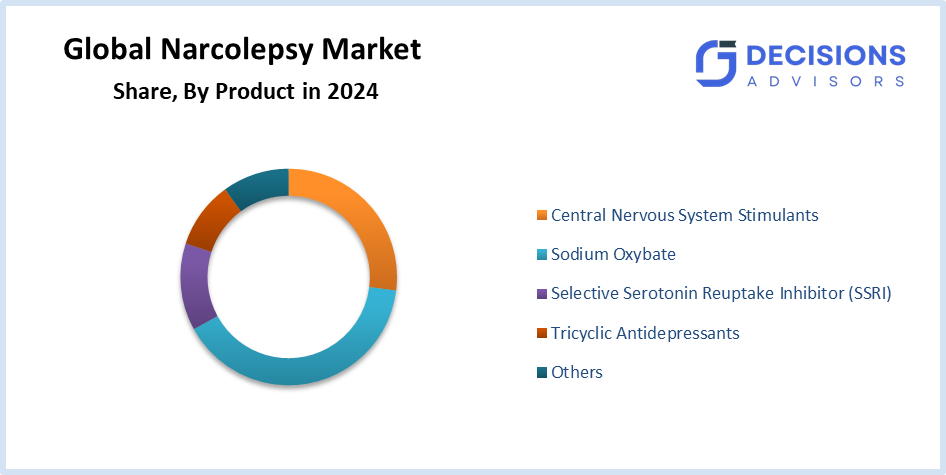
- The sodium oxybate segment accounted for the highest market revenue in 2024 and is anticipated to grow at a significant CAGR during the forecast period.
Based on the product, the narcolepsy market is differentiated into central nervous system stimulants, sodium oxybate, selective serotonin reuptake inhibitor, tricyclic antidepressants, and others. Among these, the sodium oxybate segment accounted for the highest market revenue in 2024 and is anticipated to grow at a significant CAGR during the forecast period. The segment growth will increase because of three main factors, which include high prescription rates, the expensive nature of drugs and their protective reimbursement policies, and the growing public knowledge about diagnosis and treatment methods.
For instance, Tris Pharma announced that the FDA has accepted its New Drug Application (NDA) for TRN-257, a once-nightly, low-sodium oxybate product designed to treat narcolepsy and idiopathic hypersomnia. This is a significant development in sleep medicine, as TRN-257 aims to reduce the cardiovascular risks associated with high-sodium oxybate therapies.
Regional Segment Analysis of the Narcolepsy Market
- North America (U.S., Canada, Mexico)
- Europe (Germany, France, U.K., Italy, Spain, Rest of Europe)
- Asia-Pacific (China, Japan, India, Rest of APAC)
- South America (Brazil and the Rest of South America)
- The Middle East and Africa (UAE, South Africa, Rest of MEA)
Asia Pacific is anticipated to hold the largest share of the narcolepsy market over the predicted timeframe.
Asia Pacific is anticipated to hold the largest share of the narcolepsy market over the predicted timeframe. Increasing public awareness about sleep disorders, together with higher healthcare funding levels and more people looking for effective treatments, are driving the growth of this market. The therapeutic needs of narcolepsy patients receive better treatment because regulatory authorities now classify narcolepsy as an important health condition. Japan and Australia lead this expansion because of their established healthcare systems and dedication to developing new medical solutions. The market competition is changing because more organisations are working together to make treatments more accessible and efficient for patients.
Eisai announced that Japan’s Ministry of Health, Labour and Welfare (MHLW) has granted orphan drug designation to its investigational therapy E2086, a novel orexin receptor agonist, for the treatment of narcolepsy. This move strengthens Eisai’s presence in sleep medicine and highlights the growing race to develop orexin agonists — therapies that could go beyond symptom management to address the root cause of narcolepsy.
North America is expected to grow at a rapid CAGR in the narcolepsy market during the forecast period. The region's growth is driven by increasing awareness of narcolepsy and advancements in treatment options and supportive regulatory frameworks. The demand for innovative therapies is further fueled by rising healthcare expenditures and a growing patient population seeking effective management solutions. The U.S. stands out as the leading country, with a well-established healthcare infrastructure and a focus on research and development. This environment allows companies to create new therapies that address the unique requirements of narcolepsy patients. The pharmaceutical industry's presence in major companies creates market competition, which maintains access to efficient medical treatments.
Takeda announced that the U.S. Food and Drug Administration (FDA) accepted its New Drug Application (NDA) for Oveporexton (TAK-861) and granted Priority Review for the treatment of narcolepsy type 1 (NT1). The first orexin agonist therapy available for NT1 represents a major shift from current treatments (like oxybates and pitolisant) that primarily manage symptoms rather than directly addressing orexin deficiency.
Competitive Analysis:
The report offers the appropriate analysis of the key organizations/companies involved within the narcolepsy market, along with a comparative evaluation primarily based on their product offering, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborative analysis focusing on the current news and developments of the companies, which includes product development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
List of Key Companies
- Amneal Pharmaceuticals LLC
- Avadel
- Eisai Co., Ltd.
- Eli Lilly and Company
- Harmony Biosciences
- Harmony Biosciences Holdings, Inc.
- Janssen Global Services, LLC
- Jazz Pharmaceuticals, Inc.
- Jazz Pharmaceuticals, Inc.
- Novartis AG
- Rhodes Pharmaceuticals L.P.
- Takeda Pharmaceutical Company Limited
- Teva Pharmaceutical Industries Ltd.
- UCB S.A.
- Others
Key Target Audience
- Market Players
- Investors
- End-users
- Government Authorities
- Consulting and Research Firm
- Venture capitalists
- Value-Added Resellers (VARs)
Recent Development
- In January 2026, Alkermes introduced its narcolepsy research and development (R&D) as a launchpad for broader fatigue therapies, signalling a strategic expansion beyond sleep disorders into conditions marked by chronic tiredness and impaired wakefulness. This approach represents a paradigm shift: Alkermes wants to reinterpret orexin agonists as a platform technology for a variety of fatigue disorders rather than just narcolepsy.
- In September 2025, Amneal Pharmaceuticals received FDA approval for its sodium oxybate oral solution, expanding treatment options for narcolepsy patients in the U.S. This approval strengthens Amneal’s position in the speciality pharmaceutical space and adds competition in the narcolepsy treatment market, which is rapidly evolving with new orexin agonists (like Takeda’s Oveporexton and Alkermes’ ALKS 2680) on the horizon.
- In October 2024, Avadel Pharmaceuticals received FDA approval to expand the use of Lumryz, its extended-release sodium oxybate oral suspension, for pediatric patients aged 7 years and older with narcolepsy. This approval is a milestone for pediatric sleep medicine. Children with narcolepsy now have access to a once-nightly therapy, reducing treatment burden and potentially improving adherence compared to traditional oxybate regimens.
Market Segment
This study forecasts revenue at global, regional, and country levels from 2020 to 2035. Decisions Advisors has segmented the narcolepsy market based on the below-mentioned segments:
Global Narcolepsy Market, By Type
- Type 1 Narcolepsy
- Type 2 Narcolepsy
Global Narcolepsy Market, By Treatment
- Narcolepsy with Cataplexy
- Narcolepsy Without Cataplexy
Global Narcolepsy Market, By Product
- Central Nervous System Stimulants
- Sodium Oxybate
- Selective Serotonin Reuptake Inhibitor
- Tricyclic Antidepressants
- Others
Global Narcolepsy Market, By Regional Analysis
- North America
- US
- Canada
- Mexico
- Europe
- Germany
- UK
- France
- Italy
- Spain
- Russia
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- Australia
- Rest of Asia Pacific
- South America
- Brazil
- Argentina
- Rest of South America
- Middle East & Africa
- UAE
- Saudi Arabia
- Qatar
- South Africa
- Rest of the Middle East & Africa
Frequently Asked Questions (FAQ)
1. How is the shift toward once-nightly therapies impacting the competitive dynamics of the narcolepsy market?
The introduction of once-nightly formulations is reshaping competition by improving patient adherence and convenience compared to twice-nightly dosing regimens. Companies that successfully develop simplified dosing options may gain stronger brand loyalty and expanded prescribing preference among sleep specialists.
2. What role do patient advocacy groups play in shaping the narcolepsy market?
Patient advocacy organizations significantly influence awareness campaigns, early diagnosis initiatives, and reimbursement discussions. Their efforts help reduce stigma, support research funding, and encourage faster regulatory pathways, indirectly driving market expansion.
3. How does delayed diagnosis affect overall market revenue potential?
Narcolepsy is often underdiagnosed or misdiagnosed for years, which limits early treatment adoption. Improved screening tools, better physician education, and digital symptom tracking platforms could shorten diagnosis timelines and unlock untapped patient populations, positively impacting market revenue.
4. Are biosimilars or generic entrants expected to influence pricing trends in the narcolepsy market?
As patents for certain sodium oxybate and stimulant therapies expire, generic competition is likely to intensify. This could lower drug costs, increase patient access in cost-sensitive markets, and shift revenue distribution among branded and generic manufacturers.
5. How is digital health integration contributing to narcolepsy disease management?
Wearable sleep trackers, AI-driven sleep pattern analysis, and telehealth consultations are enhancing long-term disease monitoring. These digital tools may create new service-based revenue models alongside pharmaceutical therapies, expanding the overall market ecosystem.
6. What opportunities exist in emerging markets for narcolepsy treatment providers?
Emerging economies present untapped growth potential due to increasing healthcare infrastructure investments and improving neurological care services. Rising awareness and regulatory recognition of sleep disorders could gradually increase treatment penetration in these regions.
7. How could pipeline orexin-based therapies change long-term market structure?
Orexin-targeted therapies aim to address the underlying neurochemical deficiency rather than only managing symptoms. If proven effective and safe, these therapies may redefine the standard of care, shift prescribing patterns, and potentially reduce reliance on older stimulant-based treatments.
Check Licence
Choose the plan that fits you best: Single User, Multi-User, or Enterprise solutions tailored for your needs.
We Have You Covered
- 24/7 Analyst Support
- Clients Across the Globe
- Tailored Insights
- Technology Tracking
- Competitive Intelligence
- Custom Research
- Syndicated Market Studies
- Market Overview
- Market Segmentation
- Growth Drivers
- Market Opportunities
- Regulatory Insights
- Innovation & Sustainability
Report Details
| Scope | Global |
| Pages | 230 |
| Delivery | PDF & Excel via Email |
| Language | English |
| Release | Feb 2026 |
| Access | Download from this page |
