Global Paroxysmal Nocturnal Hemoglobinuria Therapeutics Market
Global Paroxysmal Nocturnal Hemoglobinuria Therapeutics Market Size, Share, By Therapeutic Approach (Complement Inhibitors, Gene Therapy, Monoclonal Antibodies, Supportive Care), By Application (Aplastic Anemia Associated PNH, Classical PNH, Subclinical PNH), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), and By Region (North America, Europe, Asia-Pacific, Latin America, Middle East & Africa), Analysis and Forecast 2026?2035
Report Overview
Table of Contents
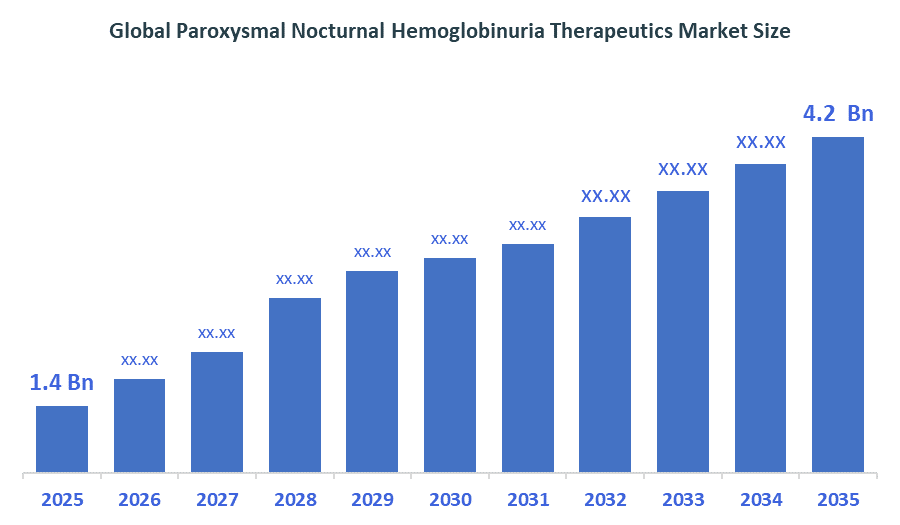
The Global Paroxysmal Nocturnal Hemoglobinuria Therapeutics Market size is forecast to grow from USD 1.4 Billion in 2025 to approximately USD 4.2 Billion by 2035. According to Decision Advisors, a detailed research report on the paroxysmal nocturnal hemoglobinuria therapeutics market indicates that the rapid expansion of the complement inhibitor therapies trend dominates the market, accounting for approximately 60-63% of the total share globally. Alexion Pharmaceuticals (AstraZeneca) leads the market with an estimated 35-40% global market share. The company reported a 2025 revenue of approximately USD 45+ Billion, making it one of the most influential forces shaping industry trends and overall market growth.
Market Snapshot
- Global Paroxysmal Nocturnal Hemoglobinuria Therapeutics Market Size (2025): USD 1.4Billion
- Projected Global Paroxysmal Nocturnal Hemoglobinuria Therapeutics Market Size (2035): USD 4.2Billion
- Global Paroxysmal Nocturnal Hemoglobinuria Therapeutics Market Compound Annual Growth Rate (CAGR): 11.61%
- Largest Regional Market: North America
- Fastest Growing Region: Asia-Pacific
- Market 3rd Largest Region: Europe
- Base Year: 2025
- Historical Period: 2021–2024
- Forecast Period: 2026–2035
Market Overview/ Introduction
The Global Paroxysmal Nocturnal Hemoglobinuria (PNH) Therapeutics Market refers to the industry that develops, produces, and sells PNH treatments, which treat a dangerous blood disorder that occurs in rare instances. Paroxysmal Nocturnal Hemoglobinuria therapeutics include complement inhibitors and monoclonal antibodies, and emerging oral and gene-based therapies, which prevent red blood cell destruction while helping to control disease symptoms. The market expands because people will gain more disease knowledge, diagnostic methods will advance, and biologic treatments will become more common. Next-generation C3 inhibitors and long-acting formulations, biosimilars, and personalized medicine approaches will create future opportunities for development. The market development depends on major factors, which include increased clinical research activities, regulatory approval achievements, and pharmaceutical companies funding of rare disease research.
- The German Arzneimittelmarkt-Neuordnungsgesetz (AMNOG), orphan drugs for PNH are legally deemed to have "added benefit." This allows immediate market access and triggers unique price negotiations with the G-BA to balance innovation with cost control.
- Canada finalized bilateral funding agreements with all provinces to create a "common list" of rare disease drugs. This aims to replace the previous patchwork of provincial programs with a unified federal strategy to improve access to PNH therapeutics.
Notable Insights: -
- North America is anticipated to hold the largest share of approximately 45% in the paroxysmal nocturnal hemoglobinuria therapeutics market over the forecast period.
- Asia Pacific is expected to grow at a rapid CAGR of approximately 11.5 % in the paroxysmal nocturnal hemoglobinuria therapeutics market during the forecast period.
- The complement inhibitors segment dominated the market in 2025, approximately 63.1 %, and is projected to grow at a substantial CAGR during the forecast period.
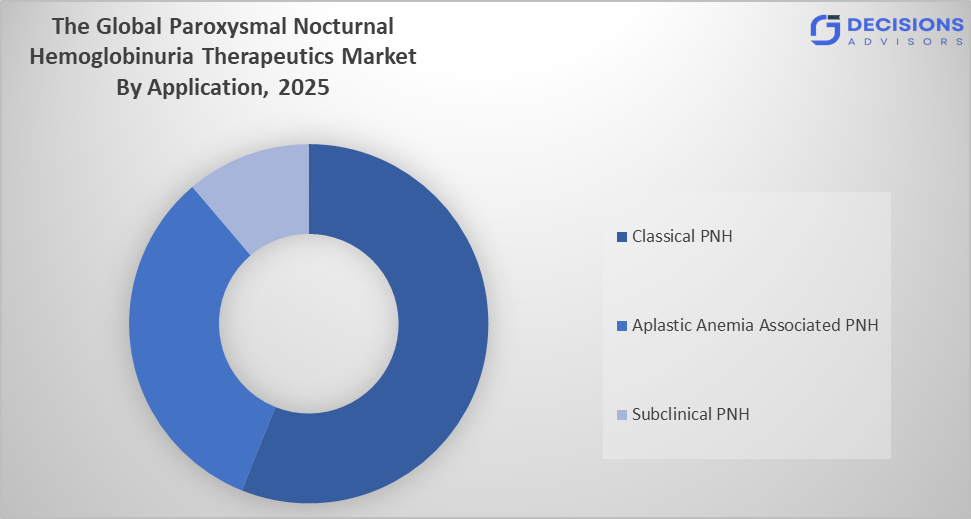
- The classical PNH segment dominated the market in 2025, approximately 60%, and is projected to grow at a substantial CAGR during the forecast period.
- The hospital pharmacies segment dominated the market in 2025, approximately 50%, and is projected to grow at a substantial CAGR during the forecast period.
- The compound annual growth rate of the Global Paroxysmal Nocturnal Hemoglobinuria Therapeutics Market is 11.61%.
- The market is likely to achieve a valuation of USD 4.2 Billion by 2035.
What is role of technology in grooming the market?
Technology plays a crucial role in shaping the Global Paroxysmal Nocturnal Hemoglobinuria (PNH) Therapeutics Market by enhancing diagnosis, treatment, and drug development. Advanced flow cytometry and genetic testing technologies enable early and accurate detection of PNH, improving patient outcomes. Biotechnology innovations support the development of next-generation complement inhibitors, including C3 and oral therapies, offering better efficacy and convenience. Artificial intelligence and data analytics accelerate drug discovery and clinical trial optimization, reducing development time and costs. Additionally, digital health platforms and telemedicine improve patient monitoring and long-term disease management. Improved biomanufacturing technologies ensure scalable production of complex biologics, increasing global availability. Overall, technology significantly enhances precision, accessibility, and efficiency in PNH treatment, driving market growth.
Market Drivers
The Global Paroxysmal Nocturnal Hemoglobinuria (PNH) Therapeutics Market grows because better disease understanding and better diagnostic methods enable doctors to find more patients with the disease throughout the globe. The clinical results from C5 and C3 inhibitor therapies show significant enhancements, which drive more healthcare professionals to use these treatments. The combination of rising healthcare costs and reimbursement systems in developed countries allows patients to access expensive biologic treatments more easily. The treatment landscape is currently expanding because of the rising number of new treatment approvals and the active clinical research that is being conducted. The market experiences continuous growth because personalized medicine approaches and long-acting treatment options lead to better patient adherence throughout the world.
Restrain
The global paroxysmal nocturnal hemoglobinuria therapeutics market faces restraints due to the extremely high cost of complement inhibitor therapies, limiting patient access in low- and middle-income regions. Additionally, limited disease awareness, delayed diagnosis, strict regulatory requirements, and dependence on long-term treatment further hinder broader market adoption and growth.
Competitive Analysis:
The report offers the appropriate analysis of the key organizations/companies involved within the global Paroxysmal nocturnal hemoglobinuria therapeutics market, along with a comparative evaluation primarily based on their product offerings, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborative analysis focusing on the current news and developments of the companies, which includes product development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
Top Companies in Global Paroxysmal Nocturnal Hemoglobinuria Therapeutics Market
- Alexion Pharmaceuticals (AstraZeneca)
- Apellis Pharmaceuticals
- Novartis AG
- F. Hoffmann-La Roche Ltd
- Genentech Inc.
- Pfizer Inc.
- Amgen Inc.
- Sanofi S.A.
- Regeneron Pharmaceuticals Inc.
- BioCryst Pharmaceuticals Inc.
- Alnylam Pharmaceuticals Inc.
- Swedish Orphan Biovitrum (Sobi)
Government Initiatives
|
Country |
Key Government Initiatives |
|
USA |
The FDA has significantly expanded the PNH treatment landscape through priority reviews and approvals of next-generation therapies, including the first oral monotherapy (Iptacopan) and subcutaneous Crovalimab. The agency also maintains a robust Risk Evaluation and Mitigation Strategy (REMS) for specific PNH drugs to manage risks like serious infections while ensuring patient safety. |
|
India |
The CDSCO recently granted permission for the import and marketing of Eculizumab (Soliris), the first anti-complement medicine in India for PNH. These moves align with India’s evolving National Policy for Rare Diseases, which facilitates local clinical trials for orphan drugs and emphasizes Post-Marketing Surveillance (PMS) to assess real-world safety in Indian patients. |
|
China |
The NMPA is leading global first-time approvals, making China the first country to approve Crovalimab (Piasky) for PNH. Under the "Healthy China 2030" plan, the government uses priority review pathways and National Reimbursement Drug List (NRDL) negotiations to drastically reduce the time to market and cost burden for rare disease therapies. |
Study on the Supply, Demand, Distribution, and Market Environment of the Paroxysmal Nocturnal Hemoglobinuria Therapeutics Market
The Global Paroxysmal Nocturnal Hemoglobinuria (PNH) Therapeutics Market shows a tightly linked supply–demand and distribution ecosystem shaped by rare disease prevalence and high-cost biologics. On the supply side, the market is dominated by a few specialized pharmaceutical companies producing complement inhibitors, resulting in concentrated manufacturing capacity and strong entry barriers. Demand is steadily rising due to improved diagnostic accuracy, increasing disease awareness, and higher patient identification rates. However, demand remains inelastic because PNH is life-threatening and requires continuous therapy. Distribution is primarily controlled through hospital pharmacies and specialty clinics, as treatments are complex biologics requiring strict storage, administration, and monitoring, though online pharmacies are gradually expanding access in remote regions. The market environment is highly regulated, innovation-driven, and reimbursement-dependent, with strong influence from insurance systems and government healthcare programs. Increasing competition from biosimilars, clinical partnerships, and emerging therapies is gradually improving accessibility and reshaping global market dynamics.
Price Analysis and Consumer Behaviour Analysis
Price analysis and consumer behaviour in the Global Paroxysmal Nocturnal Hemoglobinuria (PNH) Therapeutics Market is strongly influenced by the extremely high cost of complement inhibitor therapies such as eculizumab, ravulizumab, and newer biologics, which can reach several hundred thousand dollars annually per patient. This creates a value-driven pricing environment where manufacturers adopt premium pricing supported by strong clinical efficacy and life-saving outcomes. However, increasing entry of biosimilars and next-generation inhibitors is gradually introducing price competition and encouraging value-based and outcome-based reimbursement models. From a consumer behaviour perspective, treatment adoption is highly dependent on insurance coverage, reimbursement policies, and hospital access, as patients prioritize therapies that reduce transfusion dependence and hospital visits. Physicians also play a central role in prescribing decisions due to the rarity and complexity of PNH. Overall, despite high costs, demand remains inelastic because PNH is life-threatening and requires continuous long-term treatment.
Market Segmentation
The Paroxysmal Nocturnal Hemoglobinuria Therapeutics Market share is classified into therapeutic approach, application, and distribution channel.
- The complement inhibitors segment dominated the market in 2025, approximately 63.1%, and is projected to grow at a substantial CAGR during the forecast period.
Based on the therapeutic approach, the paroxysmal nocturnal hemoglobinuria therapeutics market is divided into complement inhibitors, gene therapy, monoclonal antibodies, and supportive care. Among these, the complement inhibitors segment dominated the market in 2025, approximately 63.1%, and is projected to grow at a substantial CAGR during the forecast period. The growth of the complement inhibitors segment is growing due to their strong clinical efficacy in controlling hemolysis and improving patient survival outcomes. These therapies directly target the complement system, reducing red blood cell destruction and minimizing complications such as thrombosis and anemia. Increasing regulatory approvals and strong physician preference have further strengthened their adoption as the standard of care. Additionally, the availability of long-acting formulations and expanding clinical evidence supporting improved quality of life have driven widespread use. High unmet medical need in PNH and limited alternative curative options also reinforce reliance on complement inhibitors.
- The classical PNH segment dominated the market in 2025, approximately 60%, and is projected to grow at a substantial CAGR during the forecast period.
Based on the application, the paroxysmal nocturnal hemoglobinuria therapeutics market is divided into aplastic anemia associated PNH, classical PNH, and subclinical PNH. Among these, the classical PNH segment dominated the market in 2025, approximately 60%, and is projected to grow at a substantial CAGR during the forecast period. The classical PNH segment dominated due to its higher clinical severity and strong symptom burden compared to other subtypes. Patients in this segment experience frequent hemolysis, anemia, thrombosis risk, and fatigue, which increases the need for immediate and continuous treatment. This leads to higher diagnosis rates and earlier therapeutic intervention. The availability of advanced complement inhibitor therapies and strong physician preference for aggressive disease management further supports market dominance.
- The hospital pharmacies drugs segment dominated the market in 2025, approximately 50%, and is projected to grow at a substantial CAGR during the forecast period.
Based on the distribution channel, the paroxysmal nocturnal hemoglobinuria therapeutics market is divided into hospital pharmacies, retail pharmacies, and online pharmacies. Among these, the hospital pharmacies segment dominated the market in 2025, approximately 50%, and is projected to grow at a substantial CAGR during the forecast period. The growth of the hospital pharmacies segment is due to the complex nature of treatment administration and the requirement for specialist supervision. Most PNH therapies, particularly complement inhibitors, are high-cost biologics that require intravenous infusion, careful monitoring, and controlled storage conditions, making hospitals the primary distribution channel.
Strategies to Implement for Growth of the Market in Non-Leading Regions
To drive growth of the Global Paroxysmal Nocturnal Hemoglobinuria (PNH) Therapeutics Market in non-leading regions, companies should focus on improving diagnosis rates and healthcare awareness, as underdiagnosis remains a major barrier. Expanding physician education programs and training hematologists in rare disease identification can support earlier detection and treatment initiation. Strengthening partnerships with local governments and NGOs can help improve reimbursement frameworks and patient access to high-cost biologics. Introducing cost-effective biosimilars and tiered pricing strategies can enhance affordability in low- and middle-income countries. Establishing regional distribution networks and cold-chain infrastructure will ensure efficient drug availability. Additionally, clinical trial expansion in emerging markets can increase patient inclusion and accelerate regulatory approvals. Digital health tools, such as telemedicine and remote monitoring, can further support long-term disease management, ultimately improving outcomes and expanding the market footprint in underserved regions globally.
Regional Segment Analysis of the Paroxysmal Nocturnal Hemoglobinuria Therapeutics Market
- North America (U.S., Canada, Mexico)
- Europe (Germany, France, U.K., Italy, Spain, Rest of Europe)
- Asia-Pacific (China, Japan, India, Rest of APAC)
- South America (Brazil and the Rest of South America)
- The Middle East and Africa (UAE, South Africa, Rest of MEA)
North America is anticipated to hold the largest share of approximately 45% the paroxysmal nocturnal hemoglobinuria therapeutics market over the forecast period.
North America is anticipated to hold the largest share of approximately 45% the paroxysmal nocturnal hemoglobinuria therapeutics market over the forecast period. The dominance of this region is primarily driven by advanced healthcare infrastructure, high diagnosis rates, strong reimbursement policies, and early adoption of complement inhibitors. Presence of leading pharmaceutical companies and robust clinical trial activity further supports regional dominance.
Asia Pacific is expected to grow at a rapid CAGR of approximately 11.5% in the paroxysmal nocturnal hemoglobinuria therapeutics market during the forecast period.
Asia Pacific is expected to grow at a rapid CAGR of approximately 11.5% in the paroxysmal nocturnal hemoglobinuria therapeutics market during the forecast period. This growth is driven by improving healthcare access, rising awareness of rare diseases, expanding diagnostic capabilities, and increasing healthcare expenditure. Growing patient population, government initiatives, and gradual adoption of advanced biologics are accelerating market expansion across emerging economies.
Europe is the 3rd largest region to grow in the paroxysmal nocturnal hemoglobinuria therapeutics market during the period.
Europe is the third largest region in the Global Paroxysmal Nocturnal Hemoglobinuria (PNH) Therapeutics Market, driven by well-established healthcare systems, strong rare disease awareness, and supportive regulatory frameworks. The region benefits from increasing adoption of complement inhibitor therapies and expanding access through public healthcare reimbursement. Growth is also supported by ongoing clinical research collaborations and rising diagnosis rates across major European countries.
Future Market Trends in Global Paroxysmal Nocturnal Hemoglobinuria Therapeutics Market: -
1. Growth of Complement Inhibitor Therapies
The market is shifting toward advanced complement inhibitors, including C5 and next-generation C3 inhibitors, due to their ability to effectively control intravascular and extravascular hemolysis. These therapies improve survival rates, reduce transfusion dependency, and offer better disease management, making them the preferred standard of care for PNH patients globally.
2. Rise of Long-Acting and Oral Treatments
A key trend is the development of long-acting injectables and oral therapies that improve patient convenience and adherence. Frequent hospital visits for intravenous infusions are being replaced by extended dosing intervals or oral options, enhancing quality of life and driving higher adoption in both developed and emerging healthcare markets.
3. Expansion of Personalized and Early Diagnosis Approaches
The market is increasingly focusing on precision medicine and early diagnosis using advanced biomarkers and genetic testing. Early detection allows timely treatment initiation, reducing complications and disease progression. This shift supports better clinical outcomes and encourages pharmaceutical companies to invest in targeted and individualized therapeutic solutions for PNH patients.
Recent Development
- In July 2025, Apellis Pharmaceuticals strengthened its partnership with Sobi by entering into a $275 million royalty repurchase agreement (with up to $300 million total potential value) related to the commercialization of Empaveli/Aspaveli (pegcetacoplan). Under the agreement, Sobi acquired rights to 90% of Apellis’ future ex-U.S. royalties, reinforcing their collaboration in rare disease treatments, including paroxysmal nocturnal hemoglobinuria (PNH).
- In December 2024, Novartis expanded its collaboration with The Max Foundation and a network of healthcare partners to improve access to Fabhalta (iptacopan) for patients with paroxysmal nocturnal hemoglobinuria (PNH). The initiative focused on working with medical institutions and specialists to address unmet needs such as residual anemia, while also strengthening treatment pathways and patient management across multiple regions.
- In April 2024, AstraZeneca and its rare disease unit Alexion Pharmaceuticals announced that Voydeya (danicopan) had been approved by the U.S. FDA as an add-on therapy to ravulizumab or eculizumab for adults with paroxysmal nocturnal hemoglobinuria (PNH) experiencing extravascular hemolysis (EVH). The therapy was designed to address persistent anemia in patients already receiving C5 inhibitor treatments, marking a significant advancement in complement-targeted therapies.
- In February 2024, Chugai Pharmaceutical and Roche announced that Piasky/Epysqli (crovalimab) had received priority review and approval in China, becoming the first country globally to authorize the therapy for patients with paroxysmal nocturnal hemoglobinuria (PNH)
How is Recent Developments Helping the Market?
Recent developments are significantly driving growth in the Global Paroxysmal Nocturnal Hemoglobinuria (PNH) Therapeutics Market by improving treatment effectiveness and expanding patient access. The introduction of complement inhibitors such as C5 and emerging C3-targeting therapies has transformed disease management by reducing hemolysis, lowering transfusion dependence, and improving survival outcomes. Ongoing clinical trials and pipeline innovations are expanding the range of next-generation oral and long-acting therapies, enhancing convenience compared to traditional intravenous options. Regulatory approvals for novel biologics and biosimilars are increasing competition and making treatments more affordable in several regions. Additionally, advancements in precision medicine and biomarker-based diagnosis are enabling earlier detection and personalized treatment approaches. Growing awareness among healthcare providers and improved reimbursement frameworks in developed markets are also supporting adoption. Together, these developments are strengthening market expansion, improving quality of life for patients, and encouraging pharmaceutical investment in rare hematologic disorder therapies globally.
Market Segment
This study forecasts revenue at global, regional, and country levels from 2020 to 2035. Decision Advisors has segmented the Global Paroxysmal Nocturnal Hemoglobinuria Therapeutics Market based on the below-mentioned segments:
Global Paroxysmal Nocturnal Hemoglobinuria Therapeutics Market, Therapeutic Approach
- Complement Inhibitors
- Gene Therapy
- Monoclonal Antibodies
- Supportive Care
Global Paroxysmal Nocturnal Hemoglobinuria Therapeutics Market, By Application
- Aplastic Anemia Associated PNH
- Classical PNH
- Subclinical PNH
Global Paroxysmal Nocturnal Hemoglobinuria Therapeutics Market, By Distribution Channel
- Hospital Pharmacies
- Retail Pharmacies
- Online Pharmacies
Global Paroxysmal Nocturnal Hemoglobinuria Therapeutics Market, By Regional Analysis
- North America
- US
- Canada
- Mexico
- Europe
- Germany
- UK
- France
- Italy
- Spain
- Russia
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- Australia
- Rest of Asia Pacific
- South America
- Brazil
- Argentina
- Rest of South America
- Middle East & Africa
- UAE
- Saudi Arabia
- Qatar
- South Africa
- Rest of the Middle East & Africa
Frequently Asked Questions (FAQ)
Q What is the key unmet need in the Global PNH Therapeutics Market?
A. The major unmet need is the lack of a definitive cure for Paroxysmal Nocturnal Hemoglobinuria. Current therapies mainly focus on symptom control and complement inhibition, but they do not fully eliminate disease progression, creating demand for gene therapies and curative treatment options.
Q. How does patient awareness impact the PNH therapeutics market?
A. Low initial awareness often leads to delayed diagnosis, which worsens patient outcomes. However, increasing education campaigns and rare disease screening programs are improving early detection rates, thereby expanding treatment adoption and driving market growth globally.
Q. Why is combination therapy gaining attention in PNH treatment?
A. Combination therapy is gaining traction because it can address both intravascular and extravascular hemolysis more effectively. Using multiple targeted agents improves disease control, reduces breakthrough hemolysis, and enhances patient quality of life compared to single-drug approaches.
Q. What role do clinical trials play in shaping the PNH therapeutics market?
A. Clinical trials are critical for introducing next-generation therapies, including oral complement inhibitors and gene-based treatments. They help validate safety and efficacy, accelerate regulatory approvals, and expand treatment options, thereby significantly influencing market innovation and competition.
Check Licence
Choose the plan that fits you best: Single User, Multi-User, or Enterprise solutions tailored for your needs.
We Have You Covered
- 24/7 Analyst Support
- Clients Across the Globe
- Tailored Insights
- Technology Tracking
- Competitive Intelligence
- Custom Research
- Syndicated Market Studies
- Market Overview
- Market Segmentation
- Growth Drivers
- Market Opportunities
- Regulatory Insights
- Innovation & Sustainability
Report Details
| Scope | Global |
| Pages | 210 |
| Delivery | PDF & Excel via Email |
| Language | English |
| Release | Apr 2026 |
| Access | Download from this page |
