Global Pharmaceutical Membrane Filtration Technologies Market
Global Pharmaceutical Membrane Filtration Technologies Market Size, Share, By Material (Polyethersulfone (PES), Mixed Cellulose Ester & Cellulose Acetate, Polyvinylidene Difluoride (PVDF), Nylon Membrane Filters) By Technique (Microfiltration, Ultrafiltration, Nanofiltration), By Application (Final Product Processing, Raw Material Filtration, Cell Separation, Water Purification, Air Purification), and By Region (North America, Europe, Asia-Pacific, Latin America, Middle East & Africa), Analysis and Forecast 2026?2035
Report Overview
Table of Contents
Market Snapshot
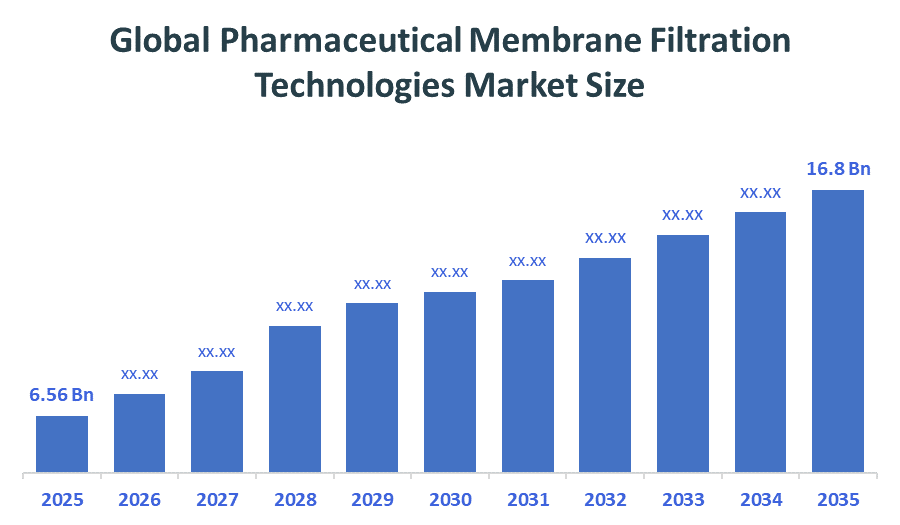
- Global Pharmaceutical Membrane Filtration Technologies Market Size (2025): USD 6.56Billion
- Projected Global Pharmaceutical Membrane Filtration Technologies Market Size (2035): USD 16.8Billion
- Global Pharmaceutical Membrane Filtration Technologies Market Compound Annual Growth Rate (CAGR): 9.96%
- Largest Regional Market: North America
- Fastest Growing Region: Asia-Pacific
- Market 3rd Largest Region: Europe
- Base Year: 2025
- Historical Period: 2021–2024
- Forecast Period: 2026–2035
Market Overview/ Introduction
The global pharmaceutical membrane filtration technologies market refers to the industry involved in manufacturing and supplying glass-based containers such as vials, ampoules, and syringes used for storing medicines. Pharmaceutical glass packaging uses non-reactive containers, which preserve drug safety, drug stability, and drug sterility. The market is witnessing strong growth because of increasing demand for packaging solutions that support biologics and vaccines, and injectable drugs. The future will bring opportunities through the creation of new glass materials and the development of lightweight packaging and sustainable recycling methods. The market expansion receives support from major factors, which include rising healthcare infrastructure investments and the implementation of stricter regulatory requirements. The pharmaceutical membrane filtration technologies market experiences continuous growth because people increasingly choose safe and environmentally friendly packaging materials instead of plastic throughout the world.
- The Production Linked Incentive (PLI) Scheme for Pharmaceuticals, launched by the Government of India, aims to boost domestic manufacturing and investment in high-value pharmaceutical products, including biopharmaceuticals, APIs, and complex generics. These segments significantly drive demand for membrane filtration technologies, particularly in sterile processing, biologics purification, and contamination control, thereby supporting growth in the global pharmaceutical membrane filtration technologies market.
- India’s bulk drug policy initiatives focus on strengthening domestic API manufacturing and reducing import dependency through financial support measures and the development of bulk drug parks. The expansion of API production capacity under these initiatives indirectly increases demand for membrane filtration technologies, particularly in upstream and downstream processing, purification, and contamination control, thereby supporting growth in the global pharmaceutical membrane filtration technologies market.
Notable Insights: -
- North America is anticipated to hold the largest share of approximately 45% in the pharmaceutical membrane filtration technologies market over the forecast period.
- Asia Pacific is expected to grow at a rapid CAGR of approximately 11.9% in the pharmaceutical membrane filtration technologies market during the forecast period.
- The polyethersulfone (PES) segment dominated the market in 2025, approximately 45 %, and is projected to grow at a substantial CAGR during the forecast period.
- The microfiltration segment dominated the market in 2025, approximately 48%, and is projected to grow at a substantial CAGR during the forecast period.
- The final product processing segment dominated the market in 2025, approximately 42%, and is projected to grow at a substantial CAGR during the forecast period.
- The compound annual growth rate of the Global Pharmaceutical membrane filtration technologies market is 9.96%.
- The market is likely to achieve a valuation of USD 16.8Billion by 2035.
What is role of technology in grooming the market?
Technology plays a crucial role in driving the growth of the global pharmaceutical membrane filtration technologies market by improving efficiency, precision, and product safety in drug manufacturing. Advanced membrane materials such as high-performance polymers and ceramic composites enhance filtration accuracy, durability, and resistance to harsh chemical conditions. Automation and real-time monitoring systems enable better process control, reducing human error and ensuring consistent product quality. The integration of single-use technologies minimizes contamination risks and improves operational flexibility, especially in biologics and vaccine production. Digitalization and Industry 4.0 solutions, including AI-based predictive maintenance and smart sensors, optimize system performance and reduce downtime. Innovations in nanofiltration and ultrafiltration technologies further support high-purity separation processes. Overall, technological advancements are making filtration processes more efficient, scalable, and compliant with strict global pharmaceutical regulations, thereby accelerating market expansion.
Market Drivers
The Pharmaceutical Membrane Filtration Technologies Market drive by major factors, the introduction of disposable filtration systems leads to faster production processes and better system cleanliness, and decreased risk of contamination between different processes, the FDA and EMA regulations establish strict drug purity standards, which require high-performance filtration systems needed for biologics and parenteral medicines, the rapid development of monoclonal antibodies, together with vaccines and recombinant proteins, demands purification processes that utilize specialized membrane filters for cell harvesting, the industry needs membrane technologies that can provide reliable performance at scale while maintaining high operational efficiency during its transition to continuous processing methods, and the increasing need for pharmaceutical infrastructure development in developing countries, together with the expanding generic drug market, drives demand for the product.
Restrain
The global pharmaceutical membrane filtration technologies market faces restraints due to high installation and operational costs, frequent membrane fouling issues, and the need for regular replacement. Additionally, stringent regulatory validation processes and limited affordability in developing regions hinder adoption, while technical complexity and skilled workforce requirements further restrict market expansion.
Competitive Analysis:
The report offers the appropriate analysis of the key organizations/companies involved within the global Pharmaceutical membrane filtration technologies market, along with a comparative evaluation primarily based on their product offerings, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborative analysis focusing on the current news and developments of the companies, which includes product development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
Top Companies in Global Pharmaceutical Membrane Filtration Technologies Market
- Merck KGaA (MilliporeSigma)
- Danaher Corporation (Pall Corporation, Cytiva)
- Sartorius AG
- Thermo Fisher Scientific Inc.
- 3M Company
- Parker Hannifin Corporation
- Repligen Corporation
- Koch Membrane Systems (Koch Industries)
- Donaldson Company, Inc.
- Alfa Laval AB
- Porvair plc
- GEA Group AG
Government Initiatives
|
Country |
Key Government Initiatives |
|
US |
The U.S. Food and Drug Administration enforces strict Current Good Manufacturing Practices (cGMP) requiring validated sterile filtration processes in drug manufacturing. Guidelines such as Sterile Drug Products Produced by Aseptic Processing mandate filtration validation, pressure monitoring, and microbial control to ensure product sterility and safety. |
|
Europe |
The European Medicines Agency regulates membrane filtration through EU GMP Annex 1, emphasizing sterilizing filtration, integrity testing, and contamination control in aseptic processing. Policies require validated 0.2–0.22 µm membrane filters and strict documentation for biologics and sterile drugs. |
|
India |
The Central Drugs Standard Control Organization aligns with WHO-GMP standards, mandating sterile filtration and validation protocols in pharmaceutical manufacturing. Government initiatives under “Make in India” and biologics expansion programs are driving adoption of membrane filtration systems in vaccine and injectable drug production. |
Study on the Supply, Demand, Distribution, and Market Environment of the Pharmaceutical Membrane Filtration Technologies Market
The pharmaceutical membrane filtration technologies market operates through a complex supply chain involving raw material suppliers, membrane manufacturers, equipment providers, and pharmaceutical end-users. Key raw materials include polymeric compounds such as polyethersulfone (PES), polyvinylidene fluoride (PVDF), polypropylene, and cellulose-based materials, along with ceramic components used in high-performance filtration systems. On the supply side, production is concentrated in technologically advanced regions due to the need for precision manufacturing and strict quality standards. Demand is strongly driven by biologics, vaccines, and injectable drug production, where high-purity separation is essential. Distribution networks include direct sales, specialized distributors, and partnerships with biopharmaceutical companies. The market environment is shaped by strict regulatory requirements, rising investments in bioprocessing, and increasing adoption of single-use systems. Overall, growing pharmaceutical production and continuous innovation in membrane materials are strengthening global supply-demand balance and expanding market opportunities.
Price Analysis and Consumer Behaviour Analysis
The pricing in the global pharmaceutical membrane filtration technologies market is influenced by high material costs, advanced membrane design, and stringent regulatory validation requirements. Technologies such as ultrafiltration and nanofiltration systems are relatively expensive due to complex manufacturing, single-use system integration, and the need for high sterility assurance. Frequent replacement of membranes and operational maintenance further increase overall lifecycle costs, making pricing a critical factor for adoption, especially among small and mid-sized pharmaceutical firms. From a consumer behavior perspective, pharmaceutical companies prioritize product quality, regulatory compliance, and contamination-free processing over cost alone. Demand is strongly driven by biopharmaceutical manufacturers focusing on biologics, vaccines, and injectables, where high-purity filtration is essential. Additionally, end-users are increasingly adopting advanced filtration systems that offer automation, scalability, and reduced downtime, even at higher costs, reflecting a shift toward value-based purchasing decisions rather than price-sensitive choices.
Market Segmentation
The Pharmaceutical Membrane Filtration Technologies Market share is classified into material, technique, and application.
- The polyethersulfone (PES) segment dominated the market in 2025, approximately 45%, and is projected to grow at a substantial CAGR during the forecast period.
Based on the material, the pharmaceutical membrane filtration technologies market is divided into polyethersulfone (PES), mixed cellulose ester & cellulose acetate, polyvinylidene difluoride (PVDF), and nylon membrane filters. Among these, the polyethersulfone (PES) segment dominated the market in 2025, approximately 45%, and is projected to grow at a substantial CAGR during the forecast period. The growth of the polyethersulfone (PES) segment is growing due to the product demonstrates outstanding thermal stability and exceptional chemical resistance, together with its ability to filter at high efficiency. PES membranes deliver protein binding strength and fast flow capacities, which make them perfect for biopharmaceutical processes that involve vaccine production, biologic development, and injectable drug manufacturing. The product achieves market dominance because it combines durability with cost efficiency and compatibility across various pharmaceutical manufacturing processes.
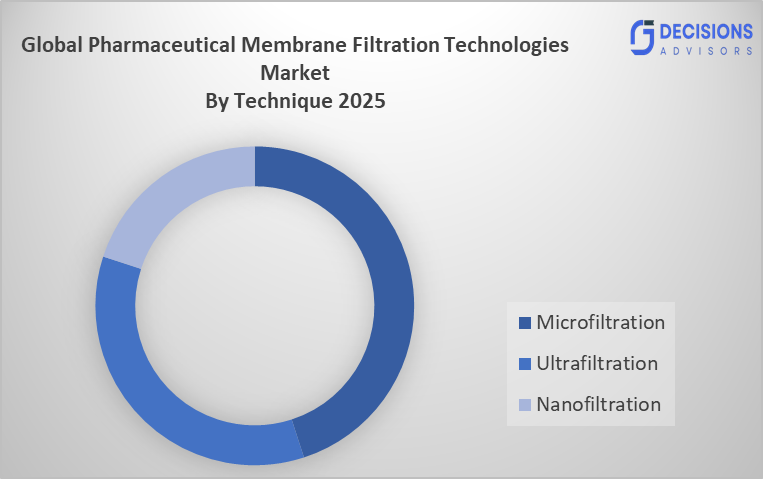
- The microfiltration segment dominated the market in 2025, approximately 48%, and is projected to grow at a substantial CAGR during the forecast period.
Based on the technique, the pharmaceutical membrane filtration technologies market is divided into microfiltration, ultrafiltration, and nanofiltration. Among these, the microfiltration segment dominated the market in 2025, approximately 48%, and is projected to grow at a substantial CAGR during the forecast period. The microfiltration segment dominated due the system achieves exceptional efficiency for eliminating bacteria and particles while keeping product integrity intact. The system serves both biopharmaceutical and sterile drug production because it provides affordable solutions that can handle extensive manufacturing operations. The rising need for biologics, vaccines, and injectable medications makes this filtration method the most common choice used in pharmaceutical manufacturing processes
- The final product processing drugs segment dominated the market in 2025, approximately 42%, and is projected to grow at a substantial CAGR during the forecast period.
Based on the application, the pharmaceutical membrane filtration technologies market is divided into final product processing, raw material filtration, cell separation, water purification, and air purification. Among these, the final product processing segment dominated the market in 2025, approximately 42%, and is projected to grow at a substantial CAGR during the forecast period. The growth of the final product processing segment is growing due to the system demonstrates its capacity to eliminate bacteria, particles, and impurities from products while keeping their original state. The system is commonly used in biopharmaceutical and sterile drug production because it offers affordable solutions and scalable operations and can handle high-volume manufacturing. The rising need for biologics, vaccines, and injectable drugs establishes the system as the main filtration choice for pharmaceutical manufacturing operations.
Strategies to Implement for Growth of the Market in Non-Leading Regions
To drive growth of the global pharmaceutical membrane filtration technologies market in non-leading regions, companies should focus on strengthening local manufacturing capabilities to reduce dependency on imports and lower production costs. Establishing strategic partnerships with regional pharmaceutical firms and biotech startups can improve market penetration and technology adoption. Investment in affordable and scalable filtration solutions tailored to local production needs will enhance accessibility. Training programs and technical support should be introduced to build skilled manpower for operating advanced filtration systems. Governments and private players should collaborate to improve regulatory frameworks and encourage foreign direct investment in pharmaceutical infrastructure. Additionally, increasing awareness about the benefits of membrane filtration in ensuring drug purity and safety will support adoption. Expanding distribution networks and leveraging digital platforms for sales and service support will further accelerate market growth in emerging and underserved regions.
Regional Segment Analysis of the Pharmaceutical Membrane Filtration Technologies Market
- North America (U.S., Canada, Mexico)
- Europe (Germany, France, U.K., Italy, Spain, Rest of Europe)
- Asia-Pacific (China, Japan, India, Rest of APAC)
- South America (Brazil and the Rest of South America)
- The Middle East and Africa (UAE, South Africa, Rest of MEA)
North America is anticipated to hold the largest share of approximately 45% the pharmaceutical membrane filtration technologies market over the forecast period.
North America is anticipated to hold the largest share of approximately 45% the pharmaceutical membrane filtration technologies market over the forecast period. The dominance of this region is primarily driven by the biopharmaceutical industry in this region operates through established methods while leading pharmaceutical companies maintain their presence, and advanced healthcare facilities provide support. The region demonstrates three main advantages through its extensive use of modern filtration systems, its strict quality control regulations, and its substantial financial commitment to biologics development, vaccine production, and single-use system implementation. The company's continuous research and development programs,, together with its technological advancements enable it to maintain its position as the market leader.
Asia Pacific is expected to grow at a rapid CAGR of approximately 11.9% in the pharmaceutical membrane filtration technologies market during the forecast period.
Asia Pacific is expected to grow at a rapid CAGR of approximately 11.9% in the pharmaceutical membrane filtration technologies market during the forecast period. This growth is driven by the biopharmaceutical manufacturing sector is experiencing rapid growth, while healthcare facilities receive increased funding and contract manufacturing organizations show substantial expansion. The market for biologics, vaccines, and injectable drugs experiences accelerated growth in emerging markets like China and India because of rising demand, government support, and affordable production methods.
Europe is the 3rd largest region to grow in the pharmaceutical membrane filtration technologies market during the period.
Europe holds the position of the third-largest region in the global pharmaceutical membrane filtration technologies market, supported by the area possesses two essential elements, which include its established pharmaceutical manufacturing facilities and its regulatory systems that guarantee product excellence and safety. The area sees extensive use of modern bioprocessing methods and advanced filtration systems, which particularly support biologics and vaccine manufacturing processes. The European market shows continuous growth because of increasing research and development investments and rising requirements for high-purity drug production.
Future Market Trends in Global Pharmaceutical Membrane Filtration Technologies Market: -
- Rise of Single-Use Filtration Systems
Single-use filtration systems will dominate future growth due to their ability to reduce cross-contamination risks and eliminate cleaning validation processes. They improve operational flexibility, lower downtime, and support rapid production of biologics and vaccines. Increasing demand for cost-efficient and scalable manufacturing further accelerates their adoption in pharmaceutical facilities globally.
- Advancements in Nanofiltration and High-Precision Membranes
Nanofiltration technologies will expand rapidly due to their superior ability to separate viruses, proteins, and small molecules with high accuracy. Continuous innovation in membrane materials improves permeability, selectivity, and durability. Growing use in gene therapy, monoclonal antibodies, and complex biologics drives demand for more efficient and high-performance filtration solutions.
- Integration of Automation and Smart Monitoring Systems
Automation and digital monitoring will become key trends as pharmaceutical manufacturers adopt real-time filtration tracking, AI-based quality control, and predictive maintenance. These technologies enhance process reliability, reduce human error, and ensure regulatory compliance. Increasing focus on data-driven manufacturing and Industry 4.0 integration further supports this transformation.
Recent Development
- In March 2026, the introduction of compact and modular membrane filtration systems enabled more efficient pharmaceutical manufacturing by reducing facility footprint and enhancing process flexibility. These advanced configurations, including single-use and scalable systems, support small-batch production and continuous manufacturing, thereby contributing to the growth of the global pharmaceutical membrane filtration technologies market.
- In February 2026, the development of next-generation membrane materials with enhanced permeability and selectivity has enabled higher flux rates and improved protein recovery in pharmaceutical manufacturing. These advancements, supported by innovations in membrane design and surface chemistry, are driving efficiency in downstream bioprocessing and contributing to the growth of the global pharmaceutical membrane filtration technologies market.
- In July 2025, the growing adoption of sterile, single-use membrane filter capsules in pharmaceutical manufacturing was driven by their ability to eliminate cleaning validation requirements, reduce operational costs, and minimize cross-contamination risks. This trend, supported by the increasing use of disposable technologies in aseptic processing, contributed significantly to the growth of the global pharmaceutical membrane filtration technologies market.
How is Recent Developments Helping the Market?
Recent developments are significantly driving the growth of the global pharmaceutical membrane filtration technologies market by improving efficiency, product purity, and regulatory compliance in drug manufacturing. Advances in filtration materials such as high-performance polymer and ceramic membranes have enhanced durability, chemical resistance, and filtration accuracy, enabling better removal of contaminants. The integration of single-use filtration systems has reduced cross-contamination risks and lowered operational costs, especially in biologics and vaccine production. Automation and digital monitoring systems are improving process control, ensuring consistent product quality and faster production cycles. Additionally, rising investments in biopharmaceutical R&D and increasing demand for sterile injectable drugs are accelerating the adoption of advanced filtration technologies. Continuous innovation in nanofiltration and ultrafiltration techniques is further supporting high-yield production, making the market more efficient, scalable, and compliant with stringent global pharmaceutical standards.
Market Segment
This study forecasts revenue at global, regional, and country levels from 2020 to 2035. Decision Advisors has segmented the Pharmaceutical membrane filtration technologies market based on the below-mentioned segments:
Global Pharmaceutical Membrane Filtration Technologies Market, By Material Type
- Polyethersulfone (PES)
- Cellulose Ester & Cellulose Acetate
- Polyvinylidene Difluoride (PVDF)
- Nylon Membrane Filters
Global Pharmaceutical Membrane Filtration Technologies Market, By Technique
- Microfiltration
- Ultrafiltration
- Nanofiltration
Global Pharmaceutical Membrane Filtration Technologies Market, By Application
- Final Product Processing
- Raw Material Filtration
- Cell Separation
- Water Purification
- Air Purification
Global Pharmaceutical Membrane Filtration Technologies Market, By Regional Analysis
- North America
- US
- Canada
- Mexico
- Europe
- Germany
- UK
- France
- Italy
- Spain
- Russia
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- Australia
- Rest of Asia Pacific
- South America
- Brazil
- Argentina
- Rest of South America
- Middle East & Africa
- UAE
- Saudi Arabia
- Qatar
- South Africa
- Rest of the Middle East & Africa
Frequently Asked Questions (FAQ)
Q What factors are driving the shift toward advanced membrane filtration systems in pharmaceutical manufacturing?
A. The shift toward advanced membrane filtration systems is driven by increasing demand for high-purity biologics, stricter global regulatory standards, and the need for contamination-free drug production. Additionally, the rise of continuous manufacturing and single-use technologies encourages adoption of more efficient, scalable, and reliable filtration solutions that improve product consistency and reduce operational risks.
Q. How does membrane filtration improve drug development in biopharmaceutical industries?
A. Membrane filtration enhances drug development by enabling precise separation of impurities, cells, and microorganisms during production. It supports the production of sensitive biologics such as monoclonal antibodies by maintaining molecular stability. This leads to higher yield, improved safety profiles, and faster development cycles, making it essential for modern biopharmaceutical innovation.
Q. What challenges do companies face when adopting membrane filtration technologies?
A. Companies face challenges such as high capital investment costs, membrane fouling issues that reduce efficiency, and the need for frequent membrane replacement. Additionally, maintaining compliance with strict validation protocols and training skilled personnel for system operation can slow down adoption, especially in cost-sensitive and emerging markets.
Q. Why is sustainability becoming important in pharmaceutical membrane filtration technologies?
A. Sustainability is gaining importance due to increasing pressure to reduce waste, energy consumption, and environmental impact in pharmaceutical manufacturing. Manufacturers are focusing on recyclable membrane materials, energy-efficient filtration systems, and single-use technologies that minimize cleaning chemicals. These efforts align with global green manufacturing goals and regulatory expectations for environmentally responsible production.
Check Licence
Choose the plan that fits you best: Single User, Multi-User, or Enterprise solutions tailored for your needs.
We Have You Covered
- 24/7 Analyst Support
- Clients Across the Globe
- Tailored Insights
- Technology Tracking
- Competitive Intelligence
- Custom Research
- Syndicated Market Studies
- Market Overview
- Market Segmentation
- Growth Drivers
- Market Opportunities
- Regulatory Insights
- Innovation & Sustainability
Report Details
| Scope | Global |
| Pages | 225 |
| Delivery | PDF & Excel via Email |
| Language | English |
| Release | Apr 2026 |
| Access | Download from this page |
