Global Polycythemia Vera Market
Global Polycythemia Vera Market Size, Share, By Indication (Primary Polycythemia Vera, Secondary Polycythemia Vera) By Mutation Status (JAK2 V617F Mutation, CALR Mutation, MPL Mutation) By Treatment Type (Phlebotomy, Cytoreductive Medications, Supportive Care, and Other) and By Region (North America, Europe, Asia-Pacific, Latin America, Middle East, and Africa), Analysis and Forecast 2025 ? 2035.
Report Overview
Table of Contents
Market Snapshot
- Market Size (2025): USD 1.35 Billion
- Projected Market Size (2035): USD 2.34 Billion
- Compound Annual Growth Rate (CAGR): 2.34%
- Largest Regional Market: North America
- Fastest Growing Region: Asia Pacific
- 3rd Largest Region: Europe
- Base Year: 2025
- Historical Period: 2021–2024
- Forecast Period: 2025–2035
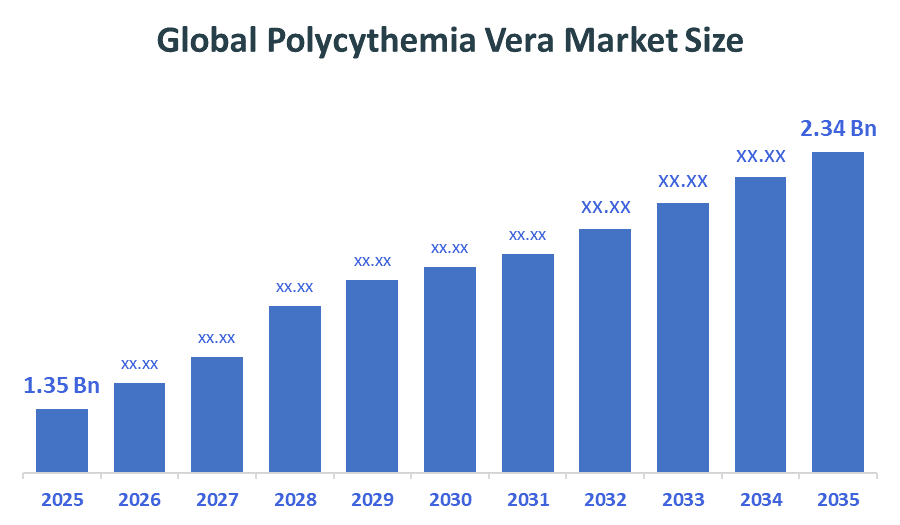
According to Decision Advisors, the Global Polycythemia Vera Market size is expected to grow from USD 1.35 billion in 2025 to USD 2.34 billion by 2035, at a CAGR of 5.65% during the forecast period 2025-2035. The global polycythemia vera market is driven by rising diagnosis rates, increasing adoption of targeted therapies such as JAK inhibitors, strong rare disease research investments, improving healthcare infrastructure, favorable reimbursement policies, and growing awareness supporting early detection and long-term disease management demand.
Market Overview/ Introduction
The global polycythemia vera market refers to the global industry focused on the diagnosis, treatment, and management of polycythemia vera, a rare chronic myeloproliferative disorder characterized by excessive production of red blood cells. Polycythemia Vera is primarily associated with mutations such as JAK2, leading to increased blood viscosity and risk of thrombosis, requiring long-term therapeutic intervention. The market encompasses a range of treatment options including phlebotomy, cytoreductive therapies, JAK inhibitors, and interferon-based biologics, addressing both low-risk and high-risk patient populations across hospitals, specialty clinics, and research institutions. The scope of the market includes drug development, diagnostics, patient management solutions, and ongoing clinical research aimed at improving treatment outcomes and disease control. Increasing investment in targeted therapies, expansion of rare disease frameworks, and advancements in molecular diagnostics are strengthening the treatment landscape. Future opportunities lie in the development of next-generation therapies, personalized medicine approaches, expansion in emerging markets, and integration of digital health technologies to enhance early diagnosis, monitoring, and long-term patient care.
- The National Policy for Rare Diseases (2021) in India provides financial assistance of up to ?50 lakh per patient, enabling access to high-cost Polycythemia Vera therapies such as ruxolitinib developed by Incyte Corporation and co-marketed by Novartis AG, along with interferon-based treatments from PharmaEssentia Corporation, thereby improving affordability, diagnosis, and long-term disease management through designated Centres of Excellence.
- The Life Saving Drugs Program in Australia offers government-funded access to expensive therapies for rare diseases, including Polycythemia Vera, supporting patient access to advanced treatments such as ruxolitinib from Incyte Corporation and ropeginterferon alfa-2b developed by PharmaEssentia Corporation, which reduces out-of-pocket costs and ensures continuity of care for chronic disease management.
- Japan’s Medical Care Program for Specific Diseases (Nanbyo) provides financial subsidies and long-term support for patients with Polycythemia Vera, facilitating access to innovative therapies such as ropeginterferon alfa-2b from PharmaEssentia Corporation and interferon-based treatments from AOP Orphan Pharmaceuticals, thereby enhancing early diagnosis, treatment adherence, and sustained clinical outcomes.
Notable Insights: -
- North America holds the largest regional market share approximately 41.65% in the global polycythemia vera market.
- Asia-Pacific is the fastest growing region in the global polycythemia vera market.
- Europe holds a significant position accounting for approximately 28% of the global polycythemia vera market share in 2024.
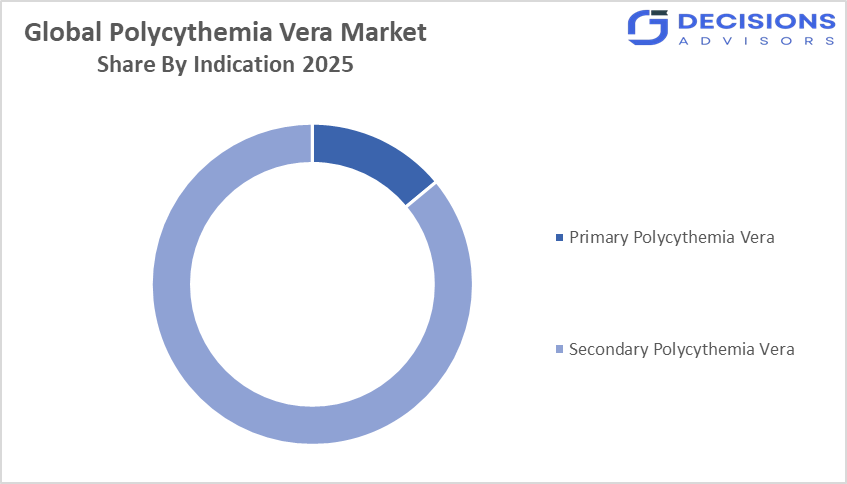
- By indication, the primary polycythemia vera segment held a dominant position with approximately 62% in terms of market share in 2025.
- By mutation status, the JAK2 V617F segment held a dominant position with presence in over 8.2% of patients.
- By treatment type, cytoreductive medications segment is the dominating accounting for over 62% of the global market share in 2025.
- The compound annual growth rate of the global Polycythemia Vera market is 5.65%.
- The market is likely to achieve a valuation of USD 2.34 Billion by 2035.
What is role of technology in grooming the market?
Technology plays a critical role in advancing the global polycythemia vera market by improving diagnosis, treatment precision, and patient management. Molecular diagnostics, particularly JAK2 mutation testing, enable early and accurate detection of the disease, supporting timely intervention. Artificial intelligence and data analytics are increasingly used to identify high-risk patients, optimize treatment selection, and enhance clinical decision-making. Additionally, advancements in targeted therapies, including JAK inhibitors and interferon-based biologics, are improving treatment outcomes and patient quality of life. Digital health platforms and telemedicine are further supporting remote monitoring, adherence tracking, and long-term disease management, especially for chronic PV patients. These technological innovations are enhancing treatment efficiency, expanding access to advanced care, and driving overall market growth.
How is Recent Developments Helping the Market?
Recent developments in the global polycythemia vera market are primarily driven by innovation in targeted therapies, expanding clinical pipelines, and increasing regulatory approvals. The introduction and growing adoption of JAK inhibitors and long-acting interferon therapies have significantly improved disease control and reduced symptom burden. Ongoing clinical trials for novel agents such as hepcidin mimetics and next-generation biologics are further strengthening the treatment landscape. Additionally, increased investment in rare disease research, improved diagnostic awareness, and expansion of specialty care centers are enhancing early detection and treatment rates. Collaborations between pharmaceutical companies and research institutions are accelerating drug development, while regulatory support for orphan drugs is facilitating faster approvals, collectively driving market expansion.
Market Drivers
The global polycythemia vera market is driven by increasing prevalence of myeloproliferative disorders, rising awareness and diagnosis rates, and growing adoption of targeted therapies. The availability of advanced treatments such as JAK inhibitors and interferon therapies is significantly improving patient outcomes and driving treatment demand. Strong healthcare infrastructure, particularly in North America and Europe, supports early diagnosis and long-term disease management. Additionally, favorable reimbursement policies, increasing investment in rare disease research, and expanding clinical pipelines are accelerating market growth. The chronic nature of the disease further ensures continuous treatment demand, contributing to sustained revenue generation.
Restrain
The global polycythemia vera market faces several restraints, including high cost of advanced therapies, limited accessibility in developing regions, and stringent regulatory requirements for drug approvals. Delayed diagnosis due to low awareness in emerging markets restricts timely treatment initiation.
Competitive Analysis:
The report offers the appropriate analysis of the key organizations/companies involved within the global polycythemia vera market, along with a comparative evaluation primarily based on their product of offering, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborative analysis focusing on the current news and developments of the companies, which includes product development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
Top Companies in Global Polycythemia Vera Market
- Incyte Corporation
- Novartis AG
- PharmaEssentia Corporation
- Bristol Myers Squibb Company
- Protagonist Therapeutics
- Disc Medicine
- Italfarmaco S.p.A.
- AOP Orphan Pharmaceuticals
- Ionis Pharmaceuticals
- Silence Therapeutics
- Merck & Co., Inc.
- F. Hoffmann-La Roche Ltd.
Government Initiatives
|
Country |
Key Government Initiatives |
|
US |
The Orphan Drug Act (1983) provides tax credits, grant funding, regulatory fee waivers, and seven-year market exclusivity to encourage development of therapies for rare diseases such as Polycythemia Vera, significantly boosting innovation and availability of targeted treatment options. |
|
EU |
The Orphan Medicinal Products Regulation (EC No. 141/2000) supports rare disease treatment development through incentives, including market exclusivity and protocol assistance, enabling faster approval and wider availability of innovative Polycythemia Vera therapies across member states. |
|
Japan |
Japan’s Orphan Drug Regulation framework provides R&D subsidies, priority review, and tax incentives to pharmaceutical companies, encouraging development and commercialization of therapies for rare diseases such as Polycythemia Vera, thereby improving treatment accessibility and innovation within the country. |
|
China |
China’s national rare disease initiatives focus on establishing patient registries, improving diagnostic capabilities, and accelerating approval pathways for orphan drugs, supporting earlier detection and increased availability of advanced therapies for Polycythemia Vera patients across the country. |
Study on the Supply, Demand, Distribution, and Market Environment of Polycythemia Vera Market
The polycythemia vera market is characterized by limited but specialized supply, with key pharmaceutical companies producing targeted therapies such as JAK inhibitors and interferons, alongside widely available conventional treatments like hydroxyurea. Demand is steadily increasing due to rising diagnosis rates, aging populations, and improved awareness of rare hematological disorders. Distribution is primarily hospital-centric, supported by specialty pharmacies and controlled drug supply chains to ensure appropriate usage. The market environment is highly regulated, with strong influence from reimbursement policies, clinical guidelines, and ongoing research advancements, which collectively shape therapy adoption, pricing dynamics, and overall market expansion across developed and emerging regions.
Price Analysis and Consumer Behaviour Analysis
The polycythemia vera market demonstrates a dual pricing structure, where low-cost treatments such as phlebotomy and hydroxyurea dominate volume, while high-cost targeted therapies like JAK inhibitors drive revenue growth. Pricing is influenced by reimbursement frameworks, hospital procurement systems, and regulatory approvals, limiting access in cost-sensitive regions. Consumer behavior is largely physician-driven, with treatment decisions based on disease severity and clinical guidelines. Patients in developed regions prefer advanced therapies due to better insurance coverage, whereas emerging markets rely on conventional treatments. Long-term adherence is affected by therapy cost, accessibility, and side effects, while growing awareness is gradually increasing the adoption of targeted treatments.
Market Segmentation
The global polycythemia vera market share is classified into indication, mutation status, and treatment type.
- The primary polycythemia vera segment dominated the market in 2025, and is projected to grow at a substantial CAGR of approximately 62% during the forecast period.
Based on indication, the global polycythemia vera market is divided into primary polycythemia vera and secondary polycythemia vera. Among these, the primary polycythemia vera segment dominated the market in 2025, accounting for 14% of cases, and is projected to grow at a substantial CAGR of approximately 62% during the forecast period. This is because it represents the majority of diagnosed PV cases, with well-established diagnostic criteria and long-term management protocols. The dominance is driven by increasing adoption of cytoreductive therapies, advanced diagnostic tools, and rising awareness among clinicians for early intervention, especially in developed regions.
- The JAK2 V617F mutation segment dominated the market in 2025, and is projected to grow at a substantial CAGR of approximately 8.2 % during the forecast period.
Based on mutation status, the global polycythemia vera market is divided into JAK2 V617F, CALR, and MPL mutations. Among these, the JAK2 V617F mutation segment dominated the market in 2025, present in 94% of all PV patients, and is projected to grow at a substantial CAGR of approximately 8.2% during the forecast period. This is because it is the primary genetic driver of PV, widely used in diagnostics, and is a key focus of targeted therapies. The dominance is driven by growing genetic testing, availability of targeted therapies, and physician awareness for early diagnosis.
- The cytoreductive medications segment dominated the market in 2025, and is projected to grow at a substantial CAGR of approximately 62% during the forecast period.
Based on treatment type, the global polycythemia vera market is divided into phlebotomy, cytoreductive medications, supportive care, and other/rare treatments. Among these, the cytoreductive medications segment dominated the market in 2025, with an estimated 62% of high-risk PV patients receiving Hydroxyurea, Ruxolitinib, or Interferon, and is projected to grow at a substantial CAGR during the forecast period. This is because these medications are the mainstay for high-risk patients, reducing complications and improving outcomes. The dominance is driven by adoption of personalized medicine, rising prevalence of PV, and supportive reimbursement policies.
What is the Reason of the Region Dominance?
The dominance of certain regions in the global polycythemia vera market is primarily driven by differences in disease diagnosis rates, treatment accessibility, and availability of advanced therapeutics. North America leads the market due to a high prevalence of diagnosed cases, strong hematology infrastructure, and early adoption of targeted therapies such as JAK inhibitors and interferon-based treatments. The presence of well-established reimbursement systems and specialized treatment centers further strengthens regional dominance. Additionally, robust clinical research activity, regulatory approvals of novel therapies, and high patient awareness contribute to increased treatment uptake. In contrast, Europe maintains a strong position due to structured rare disease frameworks and standardized treatment protocols, while Asia Pacific is expanding due to improving healthcare systems and rising diagnosis rates.
Strategies to Implement for Growth of the Market in Non-Leading Regions
Growth in non-leading regions such as Middle East & Africa can be accelerated through targeted healthcare investments, improved diagnostic access, and expansion of treatment availability. Increasing awareness among healthcare professionals and patients regarding early symptoms and disease progression can significantly enhance diagnosis rates. Expanding access to advanced therapies, including JAK inhibitors and biologics, through pricing strategies and inclusion in national reimbursement frameworks is critical for market penetration. Strengthening healthcare infrastructure, particularly hematology and oncology specialty centers, will support effective disease management. Collaborations with local healthcare providers, government bodies, and research institutions can improve clinical trial participation and accelerate therapy adoption. Additionally, investment in local manufacturing, supply chain optimization, and digital health platforms can enhance treatment accessibility and long-term patient monitoring, driving sustained market growth.
Regional Segment Analysis of the Polycythemia Vera Market
- North America (U.S., Canada, Mexico)
- Europe (Germany, France, U.K., Italy, Spain, Rest of Europe)
- Asia-Pacific (China, Japan, India, Rest of APAC)
- South America (Brazil and the Rest of South America)
- The Middle East and Africa (UAE, South Africa, Rest of MEA)
North America is anticipated to hold the largest share of the global polycythemia vera market over the predicted timeframe.
North America is the largest regional market for polycythemia vera, capturing approximately 41.65% of the global market share in 2024. This dominant position is supported by a well?established healthcare infrastructure with widespread access to advanced hematology care and high diagnostic awareness among clinicians and patients. The region’s growth is specifically fueled by rapid adoption of innovative targeted therapies such as JAK inhibitors and ropeginterferon alfa?2b, strong reimbursement coverage across the United States and Canada, extensive clinical research initiatives, and a large base of specialty clinics and hospitals focused on long?term PV management.
Asia Pacific is expected to grow at a rapid CAGR in the global polycythemia vera market during the forecast period. Asia Pacific accounts for approximately 20.3% of the global polycythemia vera market in 2024 and is expected to grow at the fastest CAGR through the forecast period. Expansion of the regional market is due to rising healthcare expenditure, improvements in diagnostic infrastructure, and growing patient awareness and screening efforts in China, India, and Japan. The Asia Pacific growth is specifically propelled by increasing investments in hematology research, expanding specialty care facilities, policy reforms that enhance access to advanced therapies, and heightened public health initiatives aimed at early detection and treatment uptake.
Europe is the 3rd largest region to grow in the global polycythemia vera market during the region. Europe holds about 28% of the global polycythemia vera market in 2024, making it the second?largest regional contributor. The market in countries such as Germany, France, and the United Kingdom benefits from high physician awareness of PV, structured rare disease management programs, and supportive healthcare policies that enable access to both established and novel treatments. Europe’s share growth is driven by comprehensive public and private reimbursement frameworks, early diagnosis initiatives, growing adoption of personalized treatment regimens, and strong participation in multinational clinical trials for next?generation therapies.
Future Market Trends in Global Polycythemia Vera Market: -
- Shift from Symptom Management to Disease Modification:
Future therapies are increasingly targeting molecular-level mechanisms to slow disease progression and achieve long-term hematocrit control. This represents a fundamental shift in PV treatment, moving beyond merely managing symptoms toward modifying disease course, which will drive both adoption of novel therapies and long-term market growth.
- Pipeline Dominance of Novel Mechanisms:
Emerging therapies like rusfertide, sapablursen, bomedemstat, and divesiran are designed to reduce the need for phlebotomy and provide better hematocrit control. The presence of multiple promising agents in advanced clinical stages indicates strong future market expansion and the potential for paradigm-shifting treatment options.
- Adoption of Targeted and Combination Therapies:
JAK inhibitors such as Jakafi (ruxolitinib) remain the standard of care, but combination strategies (e.g., Incyte’s LIMBER initiative) are gaining traction to overcome resistance and improve response rates. This trend reflects a movement toward personalized medicine and combination approaches, boosting clinical outcomes and driving market demand.
Recent Development
- In March 2026, Takeda submitted the New Drug Application (NDA) for rusfertide to the FDA, marking a major regulatory milestone for potential market approval. This submission underscores the progress toward making a novel therapy available to PV patients and reflects the growing investment in expanding treatment options within the market.
- In December 2025, 52?week extended VERIFY trial data confirmed sustained hematocrit control for rusfertide, supporting durable clinical response over long-term use. These results strengthened confidence among physicians and stakeholders regarding the therapy’s safety, efficacy, and commercial prospects in the PV market.
- In August 2025, the FDA granted Breakthrough Therapy Designation to rusfertide, accelerating regulatory review for its use in treating erythrocytosis associated with PV. This designation recognized the therapy’s potential clinical benefit and positioned rusfertide to significantly influence treatment protocols and market adoption.
- In June 2025, Takeda presented Phase?3 VERIFY clinical trial results for rusfertide, showing improved hematocrit control and reduced reliance on phlebotomy in PV patients. This pivotal study reinforced rusfertide’s potential as a disease-modifying therapy and demonstrated its capacity to address unmet clinical needs in Polycythemia Vera management.
- In February 2025, a major polycythemia vera market insight and forecast report was published, highlighting JAKAFI (ruxolitinib) and BESREMi as leading therapies. The report emphasized emerging treatments and diagnostic advances, providing a comprehensive outlook on market trends, future growth drivers, and competitive dynamics shaping the global PV landscape.
Market Segment
This study forecasts revenue at global, regional, and country levels from 2020 to 2035. Decision Advisor has segmented the global polycythemia vera market based on the below-mentioned segments:
Global Polycythemia Vera Market, By Indication
- Primary Polycythemia Vera
- Secondary Polycythemia Vera
Global Polycythemia Vera Market, By Mutation Status
- JAK2 V617F Mutation
- CALR Mutation
- MPL Mutation
Global Polycythemia Vera Market, By Treatment Type
- Phlebotomy
- Cytoreductive Medications
- Supportive Care
- Other
Global Polycythemia Vera Market, By Regional Analysis
- North America
- US
- Canada
- Mexico
- Europe
- Germany
- UK
- France
- Italy
- Spain
- Russia
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- Australia
- Rest of Asia Pacific
- South America
- Brazil
- Argentina
- Rest of South America
- Middle East & Africa
- UAE
- Saudi Arabia
- Qatar
- South Africa
- Rest of the Middle East & Africa
Frequently Asked Questions (FAQ)
Q: How does disease risk stratification influence treatment selection in polycythemia vera?
A: Risk stratification based on age, thrombotic history, and hematocrit levels determines treatment intensity. High-risk patients require cytoreductive therapies, while low-risk patients are often managed with phlebotomy and aspirin, optimizing clinical outcomes and reducing complications.
Q: What role do emerging pipeline therapies play in reshaping long-term treatment approaches?
A: Emerging therapies targeting novel pathways such as hepcidin regulation and epigenetic modulation aim to reduce treatment burden, minimize phlebotomy dependence, and potentially alter disease progression, offering more durable and patient-centric treatment outcomes.
Q: How does patient adherence impact the overall effectiveness of polycythemia vera treatments?
A: Long-term adherence is critical due to the chronic nature of the disease. Poor adherence can lead to uncontrolled hematocrit levels and increased thrombotic risk, while consistent treatment improves survival rates and enhances quality of life.
Q: What challenges do pharmaceutical companies face in expanding polycythemia vera treatments globally?
A: Companies face challenges including limited patient populations, high clinical trial costs, regulatory complexities, pricing pressures, and uneven healthcare access across regions, which collectively impact commercialization strategies and global market expansion.
Check Licence
Choose the plan that fits you best: Single User, Multi-User, or Enterprise solutions tailored for your needs.
We Have You Covered
- 24/7 Analyst Support
- Clients Across the Globe
- Tailored Insights
- Technology Tracking
- Competitive Intelligence
- Custom Research
- Syndicated Market Studies
- Market Overview
- Market Segmentation
- Growth Drivers
- Market Opportunities
- Regulatory Insights
- Innovation & Sustainability
Report Details
| Scope | Global |
| Pages | 210 |
| Delivery | PDF & Excel via Email |
| Language | English |
| Release | Apr 2026 |
| Access | Download from this page |
