Global Pompe Disease Market
Global Pompe Disease Market Size, Share, and COVID-19 Impact Analysis, By Therapy Type (Enzyme Replacement Therapy (ERT), Combination Therapy, and Other Therapies), By Route of Administration (Oral and Parenteral) and By Region (North America, Europe, Asia-Pacific, Latin America, Middle East, and Africa), Analysis and Forecast 2025 - 2035
Report Overview
Table of Contents
Global Pompe Disease Market Size Insights Forecasts to 2035
- The Global Pompe Disease Market Size Was valued at USD 1.78 Billion in 2024
- The Global Pompe Disease Market Size is Expected to Grow at a CAGR of around 7.03% from 2025 to 2035
- The Worldwide Pompe Disease Market Size is Expected to Reach USD 3.76 Billion by 2035
- Asia-Pacific is expected to Grow the fastest during the forecast period.
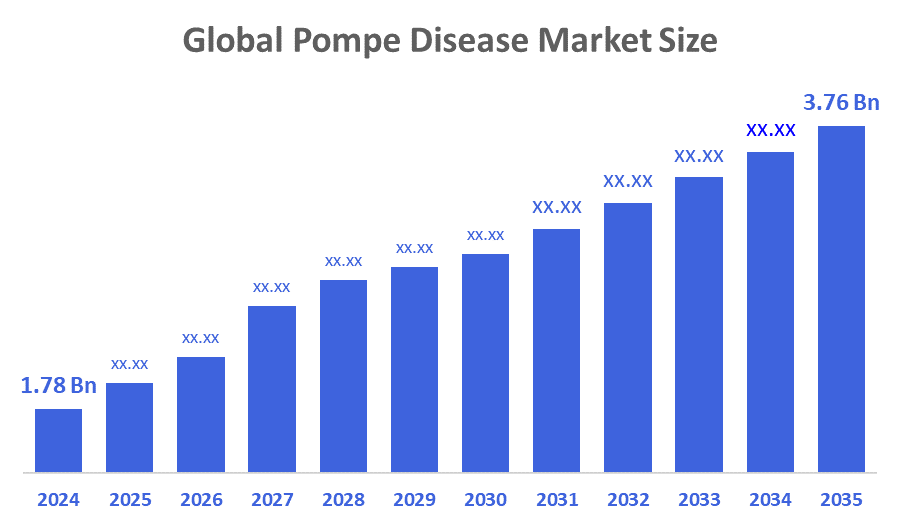
According to a Research Report Published by Decisions Advisors and Consulting, The Global Pompe Disease Market Size was worth around USD 1.78 Billion in 2024 and is predicted to Grow to around USD 3.76 Billion by 2035 with a compound annual growth rate (CAGR) of 7.03% from 2025 to 2035. The global pompe disease market presents future opportunities which will emerge from advanced gene therapies and enzyme replacement developments and the expansion of newborn screening programs and the increasing awareness of rare diseases and the development of precise diagnostic methods and strategic partnerships and the existence of supporting orphan drug regulations and better treatment accessibility in developing countries.
Market Overview
The global pompe disease market describes the entire international healthcare sector which concentrates on the process of diagnosing and treating and managing Pompe disease. This rare genetic disorder occurs because of a deficiency in the acid alpha-glucosidase enzyme which results in progressive muscular degeneration together with breathing difficulties. The market experiences growth because of both new enzyme replacement therapies and ongoing development into gene therapy and better newborn screening systems which help detect diseases early. Government initiatives provide essential support through their implementation of rare disease policies and their provision of research funding together with their establishment of expanded public healthcare services. The U.S. Food and Drug Administration together with the European Medicines Agency uses orphan drug designations and priority review pathways to speed up the approval process for new drugs. The healthcare sector gains from increased funding while patient advocacy groups and public research institutes and biopharmaceutical companies work together to improve treatment access and drive worldwide market growth.
Report Coverage
This research report categorizes the pompe disease market based on various segments and regions, forecasts revenue growth, and analyzes trends in each submarket. The report analyses the key growth drivers, opportunities, and challenges influencing the pompe disease market. Recent market developments and competitive strategies such as expansion, type launch, development, partnership, merger, and acquisition, have been included to draw the competitive landscape in the market. The report strategically identifies and profiles the key market players and analyses their core competencies in each sub-segment of the pompe disease market.
Driving Factors
The global pompe disease market experiences growth because of three factors which include increasing awareness about rare diseases and more newborn screening programs and higher rates of diagnosis. The development of next-generation enzyme replacement therapies together with gene therapy platforms and CRISPR-based research and AI-assisted diagnostics and precision medicine methods leads to better treatment results. The market expands because of strong research and development funding which benefits from orphan drug policies and patient advocacy efforts and expanding biopharmaceutical research programs.
Restraining Factors
The global pompe disease market faces restraints through multiple factors which include high therapy costs, limited patient population, delayed diagnosis, complex regulatory approvals and treatment accessibility gaps and long-term therapy dependence and insufficient specialized care infrastructure within developing regions.
Market Segmentation
The pompe disease market share is classified into therapy type and route of administration.
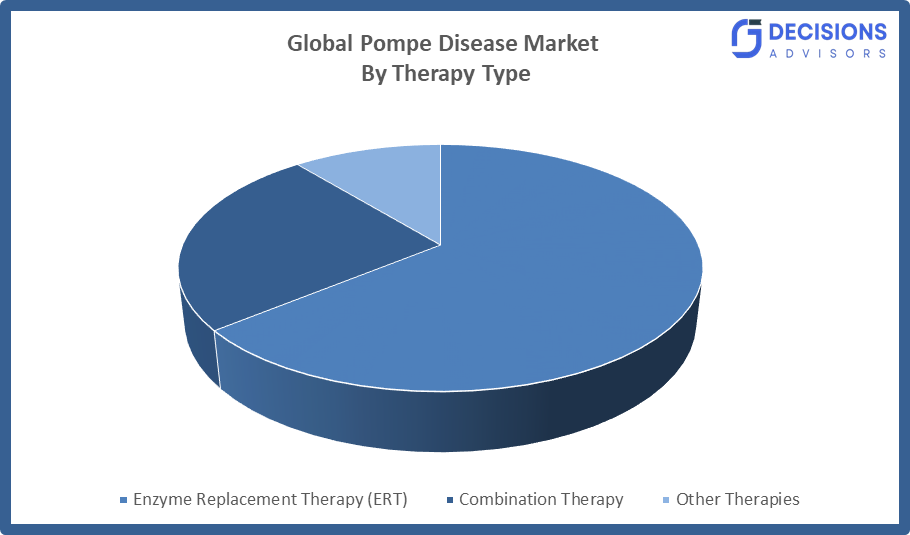
- The enzyme replacement therapy (ERT) segment dominated the market in 2024, approximately 86% and is projected to grow at a substantial CAGR during the forecast period.
Based on the therapy type, the pompe disease market is divided into enzyme replacement therapy (ERT), combination therapy, and other therapies. Among these, the enzyme replacement therapy (ERT) segment dominated the market in 2024, approximately 86% and is projected to grow at a substantial CAGR during the forecast period. The treatment shows established clinical effectiveness for Pompe disease because it successfully handles patient symptoms while it prevents disease progression. The treatment serves as the primary first-choice medication because doctors prefer it and regulatory bodies have approved it. The treatment experiences increased demand because multiple factors, such as product updates and wider access, provide reimbursement assistance and newborn screening programs improve early detection capabilities. The segment continues to grow worldwide through these factors.
- The parenteral segment accounted for the largest share in 2024, approximately 90.88% and is anticipated to grow at a significant CAGR during the forecast period.
Based on the route of administration, the pompe disease market is divided into oral and parenteral. Among these, the parenteral segment accounted for the largest share in 2024, approximately 90.88% and is anticipated to grow at a significant CAGR during the forecast period. The majority of Pompe disease therapies which receive approval need intravenous infusion to deliver their active components through the body. The method provides better drug absorption because it maintains precise medication amounts while delivering treatment effects at a quicker speed. The combination of hospital facilities, physician choices, standardized treatment procedures, and progress in developing new injectable biologic treatments will continue to drive demand for this market segment.
Regional Segment Analysis of the Pompe Disease Market
- North America (U.S., Canada, Mexico)
- Europe (Germany, France, U.K., Italy, Spain, Rest of Europe)
- Asia-Pacific (China, Japan, India, Rest of APAC)
- South America (Brazil and the Rest of South America)
- The Middle East and Africa (UAE, South Africa, Rest of MEA)
North America is anticipated to hold the largest share of the pompe disease market over the predicted timeframe.
North America is anticipated to hold the largest share of the pompe disease market over the predicted timeframe. The majority of active phase III gene-therapy studies in this area test new therapies which receive funding from its developed financial markets and its skilled contract-manufacturing facilities. The Inflation Reduction Act price negotiations create market instability through their policy changes yet these changes will not affect the high clinical value assessments which researchers conduct for life-threatening rare diseases.
Asia-Pacific is expected to grow at a rapid CAGR in the pompe disease market during the forecast period. The genomic newborn screening program in China requires all newborns to undergo testing which discovered new cases of genetic conditions that led to government funding for enzyme replacement therapy treatment. The Pharmaceuticals and Medical Devices Agency of Japan operates a fast-track system which completes orphan drug reviews within a median period of nine months while granting seven years of market exclusivity to approved medications which creates an incentive for sponsors to submit their global registration documents at the earliest opportunity. The adoption of avalglucosidase alfa by Australia and South Korea into their national reimbursement programs demonstrates how next-generation therapies have begun to achieve wider adoption throughout the entire region. The company established local production capabilities for its supply chain operations which provide both import cost reductions and operational efficiency improvements that support ongoing business development.
Competitive Analysis:
The report offers the appropriate analysis of the key organizations/companies involved within the pompe disease market, along with a comparative evaluation primarily based on their type of offering, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborative analysis focusing on the current news and developments of the companies, which includes type development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
List of Key Companies
- Sanofi
- Amicus Therapeutics
- Audentes Therapeutics (Astellas Pharma)
- Asklepios BioPharmaceutical (AskBio)
- Genethon
- Orchard Therapeutics
- Shionogi & Co., Ltd.
- Beijing GeneCradle Technology Co., Ltd.
- Maze Therapeutics
- Exerkine Corporation
- Others
Key Target Audience
- Market Players
- Investors
- End-users
- Government Authorities
- Consulting and Research Firm
- Venture capitalists
- Value-Added Resellers (VARs)
Recent Development
- In May 2025, Amicus Therapeutics reported Q1 2025 revenue of USD 125.2 million with 90% year-over-year growth for Pombiliti + Opfolda, guided toward GAAP profitability in H2 2025, and confirmed launches in up to 10 new countries during 2025.
Market Segment
This study forecasts revenue at global, regional, and country levels from 2020 to 2035. Decisions Advisors has segmented the pompe disease market based on the below-mentioned segments:
Global Pompe Disease Market, By Therapy Type
- Enzyme Replacement Therapy (ERT)
- Combination Therapy
- Other Therapies
Global Pompe Disease Market, By Route of Administration
- Oral
- Parenteral
Global Pompe Disease Market, By Regional Analysis
- North America
- US
- Canada
- Mexico
- Europe
- Germany
- UK
- France
- Italy
- Spain
- Russia
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- Australia
- Rest of Asia Pacific
- South America
- Brazil
- Argentina
- Rest of South America
- Middle East & Africa
- UAE
- Saudi Arabia
- Qatar
- South Africa
- Rest of the Middle East & Africa
Frequently Asked Questions (FAQ)
1. What is the projected growth of the global Pompe disease market?
A: The market is projected to grow from USD 1.78 billion in 2024 to USD 3.76 billion by 2035, registering a CAGR of 7.03% during the forecast period.
2. Which therapy type leads the Pompe disease market?
A: Enzyme Replacement Therapy dominates due to proven clinical effectiveness, regulatory approvals, physician preference, reimbursement support, continuous product improvements, and increased early diagnosis through expanding newborn screening programs worldwide.
3. Why does the parenteral route dominate treatment?
A: Parenteral administration leads because approved therapies require intravenous infusion for effective systemic delivery, ensuring better bioavailability, accurate dosing, faster therapeutic response, strong hospital support, standardized protocols, and growing injectable biologics development.
4. Which region holds the largest Pompe disease market share?
A: North America leads due to active Phase III gene therapy trials, strong financial markets, advanced manufacturing infrastructure, skilled research workforce, and high clinical value assessments supporting rare disease innovations.
5. Why is Asia-Pacific the fastest-growing region?
A: Mandatory newborn genomic screening in China, accelerated orphan drug approvals in Japan, expanding national reimbursements, adoption of advanced therapies, and localized biopharmaceutical manufacturing collectively accelerate rapid regional market growth.
Check Licence
Choose the plan that fits you best: Single User, Multi-User, or Enterprise solutions tailored for your needs.
We Have You Covered
- 24/7 Analyst Support
- Clients Across the Globe
- Tailored Insights
- Technology Tracking
- Competitive Intelligence
- Custom Research
- Syndicated Market Studies
- Market Overview
- Market Segmentation
- Growth Drivers
- Market Opportunities
- Regulatory Insights
- Innovation & Sustainability
Report Details
| Scope | Global |
| Pages | 259 |
| Delivery | PDF & Excel via Email |
| Language | English |
| Release | Mar 2026 |
| Access | Download from this page |
