Global Primary Biliary Cirrhosis Market
Global Primary Biliary Cirrhosis Market Size, Share, and COVID-19 Impact Analysis, By Drug Type (Ursodeoxycholic acid (USDA) and Obeticholic acid), By Stages (Portal, Periportal, Septal, Cirrhotic, and Others) and By Region (North America, Europe, Asia-Pacific, Latin America, Middle East, and Africa), Analysis and Forecast 2025 - 2035
Report Overview
Table of Contents
Global Primary Biliary Cirrhosis Market Insights Forecasts to 2035
- The Global Primary Biliary Cirrhosis Market Size Was valued at USD 552.1 Million in 2024
- The Global Primary Biliary Cirrhosis Market Size is Expected to Grow at a CAGR of around 8.34% from 2025 to 2035
- The Worldwide Primary Biliary Cirrhosis Market Size is Expected to Reach USD 1332.9 Million by 2035
- Asia-Pacific is expected to grow the fastest during the forecast period.
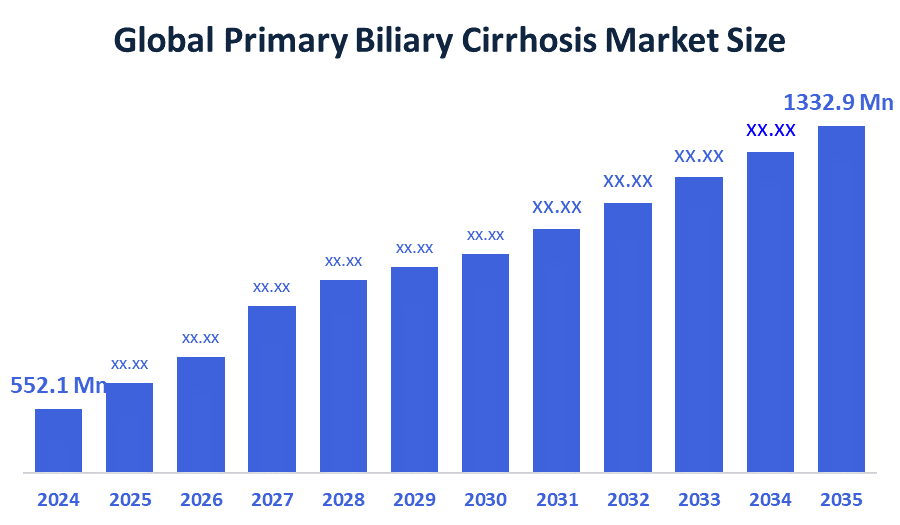
According to a research report published by Decisions Advisors and Consulting, the global primary biliary cirrhosis market size was worth around USD 552.1 Million in 2024 and is predicted to grow to around USD 1332.9 Million by 2035 with a compound annual growth rate (CAGR) of 8.34% from 2025 to 2035. The global primary biliary cirrhosis market develops future prospects through five factors which include better patient diagnosis because of increased disease awareness and the introduction of new targeted therapies and combination treatment options and the expansion of developing markets and the development of superior diagnostic tools which will sustain long-term treatment growth.
Market Overview
The global primary biliary cirrhosis market refers to the worldwide industry focused on the diagnosis, treatment, and management of primary biliary cirrhosis—now commonly known as primary biliary cholangitis a chronic autoimmune liver disease that slowly damages bile ducts and can lead to liver failure. The market experiences expansion because people have started to understand the disease better and doctors can now diagnose patients and hospitals have begun to use advanced treatments which include targeted therapies and biologic medicines. The early detection and treatment of diseases receives financial support from governments and health agencies through their funding programs and rare disease policies and expedited drug approval processes. The U.S. Food and Drug Administration and the European Medicines Agency enable companies to create new products through their orphan drug designations while the World Health Organization leads public health initiatives that elevate liver disease knowledge and provide worldwide treatment facilities.
Report Coverage
This research report categorizes the primary biliary cirrhosis market based on various segments and regions, forecasts revenue growth, and analyzes trends in each submarket. The report analyses the key growth drivers, opportunities, and challenges influencing the primary biliary cirrhosis market. Recent market developments and competitive strategies such as expansion, type launch, development, partnership, merger, and acquisition, have been included to draw the competitive landscape in the market. The report strategically identifies and profiles the key market players and analyses their core competencies in each sub-segment of the primary biliary cirrhosis market.
Driving Factors
The primary biliary cirrhosis market size will expand through the development of new treatments which drive research into all existing rare diseases. For instance, in October 2018, CymaBay Therapeutics, Inc., started placebo-controlled and randomized phase 3 clinical trial for seladelpar 5-10mg. The primary goal of the study involved evaluating the safety and effectiveness of seladelpar treatment for patients with primary biliary cirrhosis who had shown insufficient results from ursodeoxycholic acid (UDCA) treatment. The study is projected to complete in December 2021. Thus, this studies are contributing to the market growth.
Restraining Factors
The global primary biliary cirrhosis market faces restrictions because treatment expenses remain high and people lack understanding of the disease and doctors encounter difficulties with diagnosis and medications cause harmful effects and regulations become more difficult and new treatment methods take time to reach areas that lack adequate medical resources.
Market Segmentation
The primary biliary cirrhosis market share is classified into drug type and stages
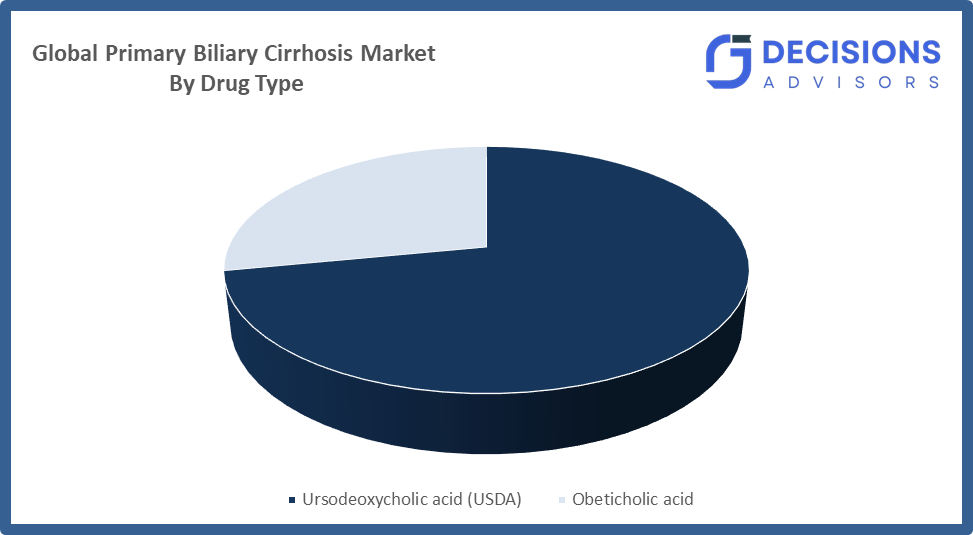
- The ursodeoxycholic acid (USDA) segment dominated the market in 2024, approximately 45% and is projected to grow at a substantial CAGR during the forecast period.
Based on the drug type, the primary biliary cirrhosis market is divided into ursodeoxycholic acid (USDA) and obeticholic acid. Among these, the ursodeoxycholic acid (USDA) segment dominated the market in 2024, approximately 45% and is projected to grow at a substantial CAGR during the forecast period. The treatment maintains its first-line status because doctors have proven its safety and effectiveness which they prefer and clinical guidelines support its use. The treatment provides greater accessibility because it costs less than newer treatments which results in higher patient acceptance. The increasing need for early diagnosis and long-term maintenance treatment will continue to drive its global market demand.
- The cirrhotic segment accounted for the largest share in 2024, approximately 35% and is anticipated to grow at a significant CAGR during the forecast period.
Based on the stages, the primary biliary cirrhosis market is divided into portal, periportal, septal, cirrhotic, and others. Among these, the cirrhotic segment accounted for the largest share in 2024, approximately 35% and is anticipated to grow at a significant CAGR during the forecast period. The advanced disease stage of patients needs continuous medical treatment because they need ongoing observation and extended medical care for their condition. The healthcare system incurs expenses because patients need to stay in hospitals more often and they face greater medical complications and need advanced medical treatments. The prevalence of late-stage diagnosis continues to increase, which expands the number of patients who require treatment in this area and helps the market maintain its ongoing growth.
Regional Segment Analysis of the Primary Biliary Cirrhosis Market
- North America (U.S., Canada, Mexico)
- Europe (Germany, France, U.K., Italy, Spain, Rest of Europe)
- Asia-Pacific (China, Japan, India, Rest of APAC)
- South America (Brazil and the Rest of South America)
- The Middle East and Africa (UAE, South Africa, Rest of MEA)
North America is anticipated to hold the largest share of the primary biliary cirrhosis market over the predicted timeframe.
North America is anticipated to hold the largest share of the primary biliary cirrhosis market over the predicted timeframe. The advanced healthcare infrastructure together with the strong specialist availability and the high disease awareness in the region creates optimal conditions for medical treatment. The combination of favorable reimbursement systems with the early adoption of innovative therapies enables patients to access their required medical treatments. The region benefits from extensive research funding together with the operations of major pharmaceutical companies and the existence of supportive regulatory pathways which boost drug development and market growth.
Asia-Pacific is expected to grow at a rapid CAGR in the primary biliary cirrhosis market during the forecast period. The healthcare system experiences challenges because healthcare costs continue to increase, hospitals acquire better diagnostic tools, and patients obtain greater access to specialized medical services. The combination of increased disease knowledge, existence of unserved patient needs, and development of government health programs enables earlier disease identification and treatment solutions. The regional market experiences substantial growth because pharmaceutical companies establish themselves in the area and medical facilities quickly adopt new treatment methods.
Competitive Analysis:
The report offers the appropriate analysis of the key organizations/companies involved within the primary biliary cirrhosis market, along with a comparative evaluation primarily based on their type of offering, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborative analysis focusing on the current news and developments of the companies, which includes type development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
List of Key Companies
- F. Hoffmann-La Roche Ltd.
- Mylan N.V.
- Pfizer Inc.
- Novartis AG
- AstraZeneca
- Johnson & Johnson Private Limited
- Sun Pharmaceutical Industries Ltd.
- Bristol-Myers Squibb Company
- Lilly
- Amgen Inc.
- Others
Key Target Audience
- Market Players
- Investors
- End-users
- Government Authorities
- Consulting and Research Firm
- Venture capitalists
- Value-Added Resellers (VARs)
Recent Development
- In July 2024, GENFIT announced that the European Medicines Agency's (EMA) Committee for Medicinal Products for Human Use (CHMP) had issued a positive opinion for Ipsen's Iqirvo (elafibranor) for the treatment of primary biliary cholangitis in combination with ursodeoxycholic acid (UDCA) in adults who had an inadequate response to UDCA or as a monotherapy in patients who could not tolerate UDCA.
Market Segment
This study forecasts revenue at global, regional, and country levels from 2020 to 2035. Decisions Advisors has segmented the primary biliary cirrhosis market based on the below-mentioned segments:
Global Primary Biliary Cirrhosis Market, By Drug Type
- Ursodeoxycholic acid (USDA)
- Obeticholic acid
Global Primary Biliary Cirrhosis Market, By Stages
- Portal
- Periportal
- Septal
- Cirrhotic
- Others
Global Primary Biliary Cirrhosis Market, By Regional Analysis
- North America
- US
- Canada
- Mexico
- Europe
- Germany
- UK
- France
- Italy
- Spain
- Russia
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- Australia
- Rest of Asia Pacific
- South America
- Brazil
- Argentina
- Rest of South America
- Middle East & Africa
- UAE
- Saudi Arabia
- Qatar
- South Africa
- Rest of the Middle East & Africa
Frequently Asked Questions (FAQ)
1. What is the projected growth of the global primary biliary cirrhosis market by 2035?
A: The market is projected to grow from USD 552.1 Million in 2024 to USD 1332.9 Million by 2035, registering a CAGR of 8.34% due to improving diagnosis and treatment innovations.
2. Which drug type segment dominates the primary biliary cirrhosis market?
A: The ursodeoxycholic acid segment dominates with a 45% share in 2024, supported by first-line treatment status, strong clinical guidelines, affordability, proven safety, effectiveness, and widespread patient and physician preference.
3. Why does the cirrhotic stage hold the largest market share?
A: Cirrhotic patients require intensive treatment, frequent hospitalization, and long-term care. Higher complication risks, increased healthcare spending, and common late-stage diagnosis significantly expand this segment’s patient pool and demand.
4. Which region leads the primary biliary cirrhosis market and why?
A: North America leads due to advanced healthcare systems, strong reimbursement coverage, high awareness, major pharmaceutical presence, significant research funding, specialist availability, and supportive regulatory pathways encouraging faster therapy approvals.
5. Why is the Asia-Pacific region growing fastest in this market?
A: Rapid growth is driven by rising healthcare investments, expanding diagnostic infrastructure, large untreated populations, improving access to specialty care, supportive government programs, and increasing adoption of advanced therapies by regional healthcare providers.
Check Licence
Choose the plan that fits you best: Single User, Multi-User, or Enterprise solutions tailored for your needs.
We Have You Covered
- 24/7 Analyst Support
- Clients Across the Globe
- Tailored Insights
- Technology Tracking
- Competitive Intelligence
- Custom Research
- Syndicated Market Studies
- Market Overview
- Market Segmentation
- Growth Drivers
- Market Opportunities
- Regulatory Insights
- Innovation & Sustainability
Report Details
| Scope | Global |
| Pages | 200 |
| Delivery | PDF & Excel via Email |
| Language | English |
| Release | Mar 2026 |
| Access | Download from this page |
