Global Ranitidine Market
Global Ranitidine Market Size, Share, By Dosage Form (Tablets, Syrup / Oral solution, and Injectable solution), By Indication (Gastroesophageal Reflux Disease (GERD), Intestinal & Stomach Ulcers (Peptic/Gastric ulcers), Esophagitis, Zollinger?Ellison Syndrome and Other) By Dosage Strength (75 mg, 150 mg, and 300 mg) By End User (Hospitals, , Specialty clinics, and Homecare / outpatient settings), and By Region (North America, Europe, Asia-Pacific, Latin America, Middle East, and Africa), Analysis and Forecast 2026-2035.
Report Overview
Table of Contents
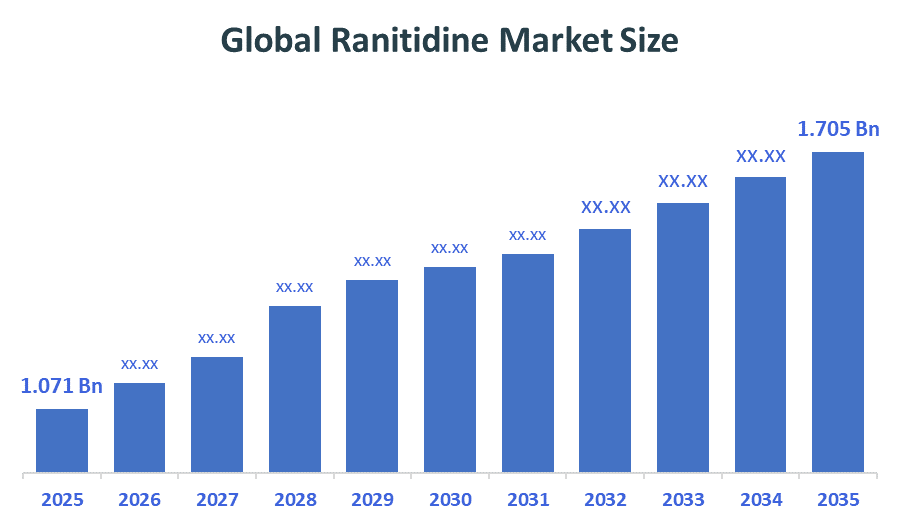
The Global Ranitidine Market Size is projected USD 1.071 Billion in 2025 and is forecasted to reach around USD 1.705 Billion by 2035. According to Decision Advisors, a detailed report on analysis of the Global Ranitidine Market indicates that the Shift from Ranitidine to Proton Pump Inhibitors (PPIs) trend dominates the Market, accounting for approx. 70–80% of the total Global demand worldwide. GlaxoSmithKline dominates the Global Ranitidine Market, supported by revenue of approx. USD 1+ billion, due to global recalls and regulatory restrictions after NDMA impurity concerns, leading to safety warnings and widespread market withdrawal.
Market Snapshot
- Global Ranitidine Market Size (2025): USD 1.071 Billion
- Projected Global Ranitidine Market Size (2035): USD 1.705 Billion
- Global Ranitidine Market Compound Annual Growth Rate (CAGR): 4.76%
- Largest Regional Market: Asia pacific
- Fastest Growing Region: North America
- 3rd Largest Region: Europe
- Base Year: 2025
- Historical Period: 2021–2024
- Forecast Period: 2026–2035
Market Overview/ Introduction
The Global Ranitidine Market is a term used to describe the international business environment for drugs containing ranitidine and employed mainly for the treatment of acid-related gastrointestinal ailments. The medication is an H2-receptor antagonist that helps in the reduction of stomach acid formation, thus serving well in GERD, peptic ulcer, Zollinger-Ellison syndrome, and esophagitis treatment. Traditionally, the growth of this market has been influenced by factors such as an increase in the number of digestive problems, poor eating practices, stress-induced illnesses, and the growth of the elderly demographic on a global scale. Recently, however, there has been a decline in the size of the market because of strict regulations related to the presence of NDMA contamination in the drug formulation. Safety measures by government bodies like the US FDA, EMA, and others have been implemented through the form of warnings and recalls as well as stringent quality parameters for the benefit of patients. There has been an implementation of programs related to pharmacovigilance, drugs quality management, and the generic drugs market. In general, the market is undergoing a shift from a time period of heavy usage to a strictly regulated period of decline. In November 2025, US FDA approved reformulated ranitidine after enhanced NDMA impurity controls, enabling limited reintroduction in regulated markets with strict safety monitoring and manufacturing standards to restore therapeutic confidence.
- In March 2026, India government’s PLI scheme for bulk drugs (?6,940 crore outlay) achieved ?4,814 crore investment and 56,800 MT API capacity by Dec 2025, strengthening domestic pharma supply chains.
- In April 2026, India’s National Pharmaceutical Pricing Authority revised ceiling prices of scheduled medicines with approximately 0.65% WPI-linked increase, impacting ranitidine formulations under price control.
- In July 2025, India’s CDSCO mandated NDMA monitoring in ranitidine API and formulations, requiring risk-based measures like reduced shelf life, following expert review of elevated impurity levels in some batches.
Notable Insights: -
- Asia-Pacific holds the largest regional Market share approximately 40% in the Global Ranitidine Market.
- North America is the fastest growing region Market share approximately 17% in the Global Ranitidine Market.
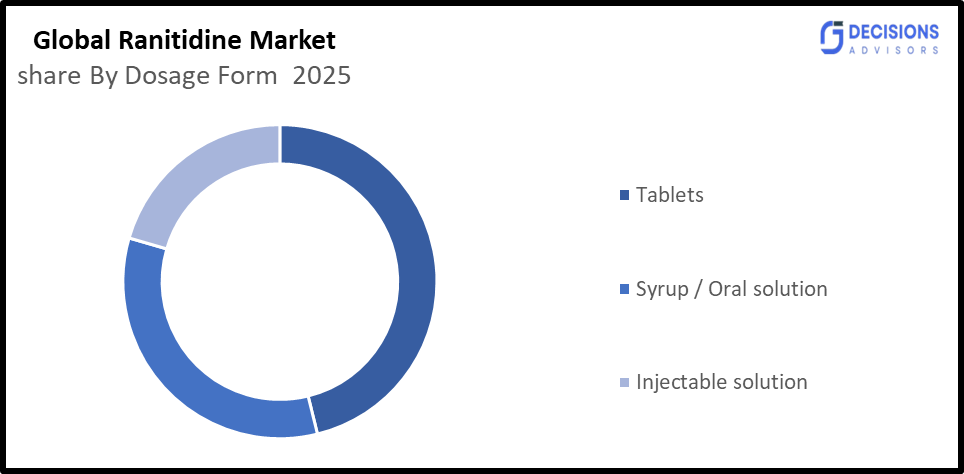
- By Dosage Form, the Tablets segment held a dominant position with 68% in terms of Market share in 2025.
- By Indication, Gastroesophageal Reflux Disease (GERD) segment is the dominating accounting for Market is approximately 43%.
- By end user, Hospitals segment held a dominant position Market is approximately 53%.
- By Dosage Strength, 150 mg segment held a dominant position Market is approximately 58%.
- The Market is likely to achieve a valuation of USD 1.705 Billion by 2035.
What is role of technology in grooming the Market?
Technology has been essential in forming and refining the global ranitidine market through improvements in drug safety, manufacturing efficiency, and regulation. Pharmaceutical technology advancements like HPLC and mass spectrometry helped to accurately detect impurities such as NDMA, which increased safety and recall efforts for the drug. Manufacturing automation and Process Analytical Technology (PAT) helped to create better efficiency and decreased contamination. Also, digital pharmacovigilance helped in reporting adverse effects to the FDA and ensured proper regulation. Formulation technology innovations also provided benefits in controlled release. The end result was that technology moved the market away from volume manufacturing to a more regulated and safer environment.
How is Recent Developments Helping the Market?
Some recent advancements that are positively impacting the global ranitidine market are related to increased safety, improved regulatory confidence, and the controlled use of the drug in certain parts of the world. Following global recalls of the drug because of the presence of an impurity known as NDMA, advancements in technology in the form of sophisticated testing technologies, including high-performance liquid chromatography, coupled with stringent manufacturing practices, have contributed considerably toward better product quality. Increased pharmacovigilance along with more stringent impurity limits have restored some level of confidence for ranitidine producers. The new formulation is being used in certain emerging countries under limited conditions. Nevertheless, the demand is weak owing to low consumption of ranitidine.
Market Drivers
The world ranitidine market is affected by various factors that act as drivers even in the face of overall decline owing to safety issues. The first driver is the high global prevalence of disorders of the digestive system, such as GERD, peptic ulcers, and acid reflux, which continues to provide the base for demand. Second, the low price and widespread availability of ranitidine across the globe have resulted in increased demand. Other drivers include the presence of geriatrics and problems associated with their lifestyles such as poor diet, stressful situations, and excessive drinking. The production of generics along with distribution channels also plays an important role in some developing nations in sustaining demand. Additionally, ranitidine remains prevalent in some areas because of its use in the past and familiarity of doctors with this drug. Moreover, the introduction of better regulation and more sophisticated technology for detecting impurities allows for careful use of the drug in selected markets.
Restrain
The global ranitidine market is restrained mainly by safety concerns related to NDMA impurities, leading to widespread recalls and regulatory bans. Strong substitution by proton pump inhibitors (PPIs), declining physician preference, and reduced consumer trust further limit demand. Additionally, strict regulatory scrutiny and limited product approvals in major markets significantly restrict market growth and expansion opportunities.
Competitive Analysis:
The report offers the appropriate analysis of the key organizations/companies involved within the Global Ranitidine Market, along with a comparative evaluation primarily based on their Product of offering, business overviews, geographic presence, enterprise strategies, segment Market share, and SWOT analysis. The report also provides an elaborative analysis focusing on the current news and developments of the companies, which includes Product development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and Others. This allows for the evaluation of the overall competition within the Market.
Top Companies in Global Ranitidine Market
- GlaxoSmithKline
- Sanofi
- Pfizer
- Novartis (Sandoz)
- Boehringer Ingelheim
- Teva Pharmaceutical Industries
- Sun Pharmaceutical Industries
- Dr. Reddy’s Laboratories
- Cipla
- Aurobindo Pharma
Government Initiatives
|
Country |
Key Government Initiatives |
|
U.S.
|
In November 2025, U.S. Food and Drug Administration approved reformulated ranitidine with updated storage and manufacturing controls to reduce NDMA risks, supporting global nitrosamine monitoring efforts. |
|
New Zealand |
In January 2025, Government granted consent for distribution of ranitidine (Zantac) tablets via Gazette notice, supporting cautious reintroduction of reformulated products meeting updated safety standards. |
Study on the Supply, Demand, Distribution, and Market Environment of Ranitidine Market
The market for ranitidine worldwide functions within an intensely regulated and shrinking industry, owing to safety issues that have been raised regarding NDMA contamination of this product. With regards to supply, there has been a reduction in the supply of this product due to the many recalls that have taken place across the globe. In relation to demand, the demand for this product has fallen sharply owing to the substitution of this drug by proton pump inhibitors (PPIs). Nonetheless, there is still some demand in emerging nations owing to their cheaper nature. In terms of distribution, distribution takes place in hospitals and pharmacies but in reduced amounts.
Price Analysis and Consumer Behaviour Analysis
The worldwide ranitidine market experiences falling prices and stringent regulation related to the safety and demand of the product. The pricing is generally low for tablets, which can be purchased for roughly USD 0.02-0.10 per tablet in generics, as a result of increased competition among the remaining manufacturers. Price sensitivity is very high for ranitidine tablets, particularly in emerging nations due to affordability influencing consumer decision-making processes. There have been notable changes in consumer behaviour after 2020, whereby 60-70% of customers opted to use products such as PPIs because of concerns raised about safety and professional advice from their doctors.
Market Segmentation
The Global Ranitidine Market share is classified into mounting dosage form, indication, dosage strength, and end user
- The Tablets segment dominated the Market in 2025, and is projected to grow at a substantial CAGR of approximately 5.0% during the forecast period.
Based on the dosage form, the global ranitidine market is divided into tablets, syrup / oral solution, and injectable solution. Among these, tablets are the most dominant segment in the global ranitidine market, accounting for the largest share in 2025 at approximately 65–70% of total market revenue, driven by wide outpatient use, low cost, and ease of administration. Syrup/oral solutions hold a moderate share due to paediatric and elderly demand, while injectables remain limited to hospital settings. The tablet segment is expected to grow steadily with a CAGR of around 4.0–5.2% during the forecast period, supported by high prescription volume and chronic acid disorder prevalence.
- The Gastroesophageal Reflux Disease (GERD) segment accounted for the largest share in 2025, and is anticipated to grow at a significant CAGR of approximately 5.3% during the forecast period.
based on the indication, the global ranitidine market is divided into gastroesophageal reflux disease (GERD), intestinal & stomach ulcers (peptic/gastric ulcers), esophagitis, Zollinger–Ellison syndrome, and other. Among these, Gastroesophageal Reflux Disease (GERD) is the most dominant segment in the global ranitidine market, accounting for approximately 40–45% of total market revenue in 2025. This dominance is driven by the rising global prevalence of acid reflux, unhealthy dietary habits, and increasing stress levels. Peptic and gastric ulcers follow as the second-largest segment. GERD is expected to grow at a CAGR of around 4.2–5.5%, supported by high diagnosis rates, long-term treatment needs, and continued use of acid-suppressing therapies in clinical practice.
- The Hospitals segment dominated the Market in 2025, and is projected to grow at a substantial CAGR of approximately 4.5% during the forecast period.
Based on the end user, the global ranitidine market is divided into hospitals, specialty clinics, and homecare / outpatient settings. Among these, hospitals are the most dominant segment in the global ranitidine market, accounting for approximately 50–55% of total market revenue in 2025. This dominance is due to high inpatient admissions for severe acid-related disorders, intravenous administration of ranitidine, and availability of advanced diagnostic and treatment facilities. Specialty clinics follow with moderate share, while homecare/outpatient settings are growing steadily. The hospital segment is expected to expand at a CAGR of around 3.8–4.8%, supported by rising gastrointestinal disease burden and emergency care utilization.
- The 150 mg segment dominated the Market in 2025, and is projected to grow at a substantial CAGR of approximately 5.7% during the forecast period.
Based on the dosage strength, the global ranitidine market is divided into 75 mg, 150 mg, and 300 mg. Among these, the 150 mg segment is the most dominant in the global ranitidine market, accounting for approximately 55–60% of total market revenue in 2025. This dominance is due to its widespread prescription as the standard maintenance dose for GERD, peptic ulcers, and other acid-related disorders. The 75 mg segment is mainly used for mild symptoms, while 300 mg is reserved for severe conditions. The 150 mg segment is expected to grow at a CAGR of around 4.0–5.3%, supported by high clinical preference and long-term therapy use.
Top of FormBottom of FormStrategies to Implement for Growth of the Market in Non-Leading Regions
The world ranitidine market reflects low pricing trends that continue to drop owing to stiff competition and falling demand following global withdrawals. Market size, as reflected in revenue, stood at about $1.19 billion in 2025, although there are still pressures arising from safety risks and government limitations. Price undercutting often happens in developing economies due to economic considerations, particularly in Asia Pacific, where the market has the highest revenue share of roughly 38.2%. There are notable changes in consumer behaviour, with up to 60-70% of consumers moving away to other medications, such as PPIs, due to safety risks linked to the presence of NDMA and doctors' choices. The rest of the consumers represent those from economically poor nations using their availability in pharmacies and hospitals. Generally, consumer behaviour revolves around being highly sensitive to prices, lack of confidence in ranitidine, and substituting for safer drugs.
Regional Segment Analysis of the Global Ranitidine Market
- North America (U.S., Canada, Mexico)
- Europe (Germany, France, U.K., Italy, Spain, Rest of Europe)
- Asia-Pacific (China, Japan, India, Rest of APAC)
- South America (Brazil and the Rest of South America)
- The Middle East and Africa (UAE, South Africa, Rest of MEA)
Asia pacific is anticipated to hold the largest share of the Global Ranitidine Market over the predicted timeframe.
Asia Pacific is anticipated to hold the largest share of the global ranitidine market in 2025, accounting for approximately 38–42% of total market revenue. This dominance is driven by a high prevalence of gastrointestinal disorders, large population base, increasing OTC drug usage, and strong demand for low-cost generic medicines. Countries like India and China contribute significantly due to extensive pharmaceutical manufacturing and distribution networks. The region is expected to grow at a CAGR of around 3.5–4.5%, supported by healthcare accessibility improvements and continued use in cost-sensitive markets despite global safety concerns and substitution with PPIs.
North America is expected to grow at a rapid CAGR in the Global Ranitidine Market during the forecast period. North America is expected to show limited but relatively stable activity in the global ranitidine market, accounting for around 15–18% of market revenue in 2025. Growth is driven by residual hospital use, strict regulatory monitoring, and controlled prescriptions in specific clinical cases. However, demand remains restricted due to FDA recalls and strong substitution with proton pump inhibitors. The region is projected to grow at a CAGR of approximately 2.5–3.5%, mainly supported by niche medical use, pharmacovigilance-driven reintroductions, and limited generic availability rather than broad consumer demand.
Europe is the 3rd largest region to grow in the Global Ranitidine Market during the region. Europe is the third-largest region in the global ranitidine market, accounting for approximately 18–22% of market revenue in 2025. Growth is limited due to strict regulatory actions by the EMA following NDMA impurity concerns, leading to reduced prescriptions and widespread substitution with proton pump inhibitors. However, demand persists in certain Eastern European and cost-sensitive segments where generic drugs remain in use. The region is expected to grow at a CAGR of around 2.0–3.0%, driven mainly by controlled hospital usage, legacy stock utilization, and selective approvals under stringent safety monitoring frameworks.
Future Market Trends in Global Ranitidine Market: -
- Continued Shift Toward Safer Alternatives
The dominant future trend in the global ranitidine market is the ongoing replacement of ranitidine with proton pump inhibitors (PPIs) due to safety concerns related to NDMA impurities. Physicians and hospitals are increasingly avoiding ranitidine, leading to a long-term decline in prescriptions. This substitution is expected to persist globally, especially in developed markets with strict regulatory frameworks. As a result, ranitidine’s market share continues to shrink while PPIs dominate acid-suppressant therapy, reshaping treatment standards worldwide.
2. Strict Regulatory Surveillance and Limited Re-Entry
Regulatory bodies like the FDA and EMA are maintaining strict monitoring of ranitidine products, requiring advanced impurity testing and pharmacovigilance systems. In some developing regions, controlled re-evaluation and limited re-entry of reformulated products may occur under stringent conditions. However, approvals remain highly restricted. This trend ensures patient safety but prevents large-scale market recovery. The market is increasingly shaped by compliance-driven manufacturing, batch testing, and tighter quality assurance standards globally.
- Growth in Emerging Market Niche Demand
Despite global decline, small-scale demand persists in emerging economies due to affordability and availability of generic formulations. Rural and cost-sensitive populations still rely on ranitidine as an OTC acid-relief option. Local manufacturers continue supplying low-cost versions under regulatory limits. This creates a niche but stable demand segment. However, growth remains modest as awareness of safer alternatives increases. Overall, emerging markets sustain residual consumption rather than driving significant global expansion.a
Recent Development
In November 2025, U.S. Food and Drug Administration approved VKT Pharma/SMS Pharma reformulated ranitidine tablets (150 mg, 300 mg), marking major re-entry after five-year withdrawal and addressing NDMA safety concerns.
In March 2024, India Government launched RPTUAS with ?300.10 crore outlay to support pharma MSMEs in upgrading to WHO-GMP and Schedule-M standards, enhancing quality and global competitiveness.
Market Segment
This study forecasts revenue at Global, regional, and country levels from 2021 to 2035. Decision Advisors has segmented the Global Ranitidine Market based on the below-mentioned segments:
Global Ranitidine Market, By Dosage Form
- Tablets
- Syrup / Oral solution
- Injectable solution
Global Ranitidine Market, By Indication
- Gastroesophageal Reflux Disease (GERD)
- Intestinal & Stomach Ulcers (Peptic/Gastric ulcers)
- Esophagitis
- Zollinger–Ellison Syndrome
- Other
Global Ranitidine Market, By Dosage Strength
- 75 mg
- 150 mg
- 300 mg
Global Ranitidine Market, By End User
- Hospitals
- Specialty clinics
- Homecare / outpatient settings
Global Ranitidine Market, By Regional Analysis
- North America
- US
- Canada
- Mexico
- Europe
- Germany
- UK
- France
- Italy
- Spain
- Russia
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- Australia
- Rest of Asia Pacific
- South America
- Brazil
- Argentina
- Rest of South America
- Middle East & Africa
- UAE
- Saudi Arabia
- Qatar
- South Africa
- Rest of the Middle East & Africa
Frequently Asked Questions (FAQ)
Q1. What factors influence the pricing of ranitidine products globally?
A1. Pricing is influenced by raw material costs, regulatory compliance requirements, manufacturing quality standards, and competition among generic drug manufacturers, especially in emerging markets.
Q2. How does the supply chain of ranitidine ensure drug availability in developing regions?
A2. The supply chain relies on generic manufacturers, local distributors, and hospital procurement systems that help maintain limited but steady availability in cost-sensitive regions.
Q3. What role do physicians play in shaping ranitidine usage patterns?
A3. Physicians significantly influence usage through prescription practices, often preferring safer alternatives and limiting ranitidine use based on updated clinical guidelines and safety advisories.
Q4. How do patient awareness levels impact demand trends?
A4. Increasing patient awareness about drug safety and side effects has reduced demand for ranitidine, encouraging patients to switch to alternative acid-reducing therapies.
Q5. What challenges do manufacturers face in maintaining ranitidine production?
A5. Manufacturers face challenges such as strict regulatory compliance, impurity control requirements, declining market demand, and increased competition from alternative therapies.
Check Licence
Choose the plan that fits you best: Single User, Multi-User, or Enterprise solutions tailored for your needs.
We Have You Covered
- 24/7 Analyst Support
- Clients Across the Globe
- Tailored Insights
- Technology Tracking
- Competitive Intelligence
- Custom Research
- Syndicated Market Studies
- Market Overview
- Market Segmentation
- Growth Drivers
- Market Opportunities
- Regulatory Insights
- Innovation & Sustainability
Report Details
| Scope | Global |
| Pages | 210 |
| Delivery | PDF & Excel via Email |
| Language | English |
| Release | Apr 2026 |
| Access | Download from this page |
