Global Recombinant Therapeutic Antibodies and Proteins Market
Global Recombinant Therapeutic Antibodies and Proteins Market Size, Share, By Drug Class (Plasma Proteins, Growth Factors, Fusion Proteins, Monoclonal Antibodies, Hormones, Enzymes, Coagulation Factors), By Host Cell Type (Mammalian Cells, Bacterial Cells, Yeast Cells), By End User (Biotechnology and Pharmaceutical Companies, Research Organizations and Laboratories, Diagnostic Centers), and By Region (North America, Europe, Asia-Pacific, Latin America, Middle East & Africa), Analysis and Forecast 2026-2035
Report Overview
Table of Contents
Market Snapshot
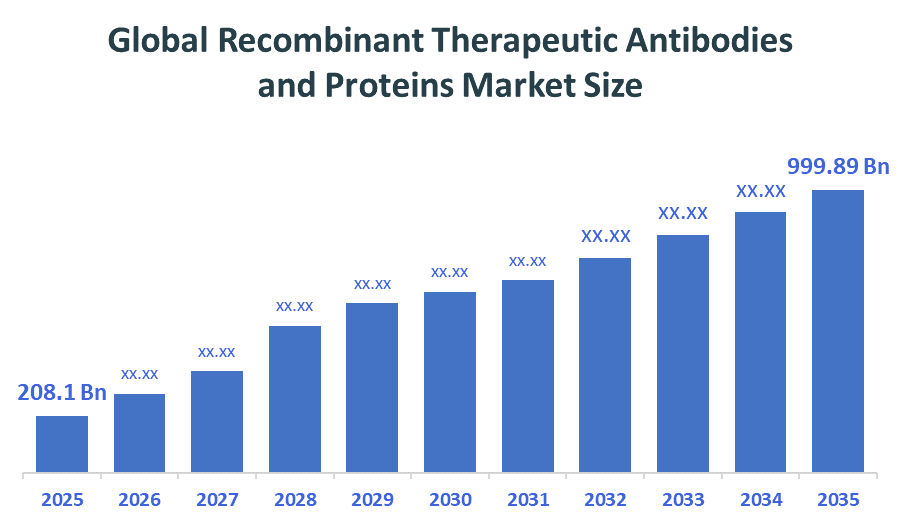
- Global Recombinant Therapeutic Antibodies and Proteins Market Size (2025): USD 208.1 Billion
- Projected Global Recombinant Therapeutic Antibodies and Proteins Market Size (2035): USD 999.89Billion
- Global Recombinant Therapeutic Antibodies and Proteins Market Compound Annual Growth Rate (CAGR): 17%
- Largest Regional Market: North America
- Fastest Growing Region: Asia-Pacific
- Market 3rd Largest Region: Europe
- Base Year: 2025
- Historical Period: 2021–2024
- Forecast Period: 2026–2035
Market Overview/ Introduction
The Global Recombinant Therapeutic Antibodies and Proteins Market refers to the industry dedicated to creating and manufacturing biologically engineered proteins and antibodies that serve as therapeutic solutions to medical conditions. Scientists create recombinant therapeutic antibodies and proteins through laboratory processes that use genetic engineering to produce specific proteins that treat medical conditions. The market is growing because more individuals are developing chronic illnesses, creating demand for precise medical solutions and biotechnological advancements. The market will create new business possibilities through its development of personalized medicine and biosimilar products, and its entry into developing countries, which are building advanced medical facilities. The market expansion occurs from protein engineering advancements and AI-based drug discovery methods, and affordable manufacturing techniques that improve treatment results and make medical solutions available to all countries.
- The U.S. FDA’s expedited regulatory pathways, including Fast Track, Breakthrough Therapy Designation, Priority Review, and Accelerated Approval, play a significant role in supporting the Global Recombinant Therapeutic Antibodies and Proteins Market by accelerating the development and approval of biologics such as monoclonal antibodies and recombinant therapeutic proteins used in oncology and autoimmune diseases.
- Government biotechnology and regulatory initiatives worldwide play a crucial role in accelerating the development, approval, and manufacturing of biologics, including monoclonal antibodies and recombinant therapeutic proteins, thereby supporting the growth of the Global Recombinant Therapeutic Antibodies and Proteins Market.
Notable Insights: -
- North America is anticipated to hold the largest share of approximately 38% in the recombinant therapeutic antibodies and proteins market over the forecast period.
- Asia Pacific is expected to grow at a rapid CAGR of approximately 13.5 % in the recombinant therapeutic antibodies and proteins market during the forecast period.
- The monoclonal antibodies segment dominated the market in 2025, approximately 50 %, and is projected to grow at a substantial CAGR during the forecast period.
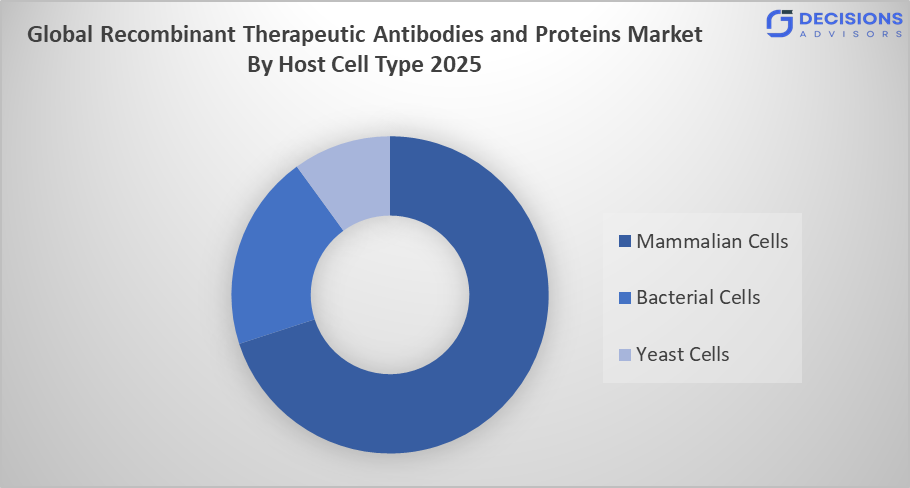
- The mammalian cells segment dominated the market in 2025, approximately 65%, and is projected to grow at a substantial CAGR during the forecast period.
- The biotechnology and pharmaceutical companies segment dominated the market in 2025, approximately 68%, and is projected to grow at a substantial CAGR during the forecast period.
- The compound annual growth rate of the Global Recombinant Therapeutic Antibodies and Proteins Market is 17%.
- The market is likely to achieve a valuation of USD 998.4Billion by 2035.
What is role of technology in grooming the market?
Technology plays a pivotal role in advancing the Global Recombinant Therapeutic Antibodies and Proteins Market by enhancing drug development, production efficiency, and treatment precision. Innovations in genetic engineering and recombinant DNA technology enable the creation of highly targeted biologics with improved efficacy and reduced side effects. Advanced expression systems, such as mammalian cell cultures and microbial platforms, increase the yield and scalability of antibody and protein production. Artificial intelligence and bioinformatics accelerate drug discovery, optimize molecular design, and predict clinical outcomes. Continuous bioprocessing and automation streamline manufacturing, reduce costs, and ensure consistent quality. Additionally, advancements in purification technologies and single-use systems improve safety and flexibility. Overall, technology drives faster innovation, regulatory compliance, and personalized medicine approaches, significantly expanding the market’s growth and therapeutic potential.
Market Drivers
The Global Recombinant Therapeutic Antibodies and Proteins Market experiences market growth because of increasing cancer, autoimmune disease, and infectious disease rates, create a greater demand for biologic treatments that include monoclonal antibodies. The production process and medical treatment results improve through ongoing progress in recombinant DNA technology, cell culture systems, and protein engineering, involves bispecific antibodies and antibody-drug conjugates. The global healthcare expenditure increases, which leads to pharmaceutical companies making larger investments that result in quicker drug development. The introduction of biosimilars results in greater treatment options, which become more affordable and accessible for patients. The development of protein expression technologies through phage display libraries and mammalian cell systems enables organizations to produce products efficiently at large production scales
.
Restrain
The Global Recombinant Therapeutic Antibodies and Proteins Market faces restraints due to high development and manufacturing costs, complex regulatory approval processes, and stringent quality requirements. Limited accessibility in low-income regions, risk of immunogenicity, and challenges in large-scale production further hinder market growth and widespread adoption of these advanced biologics.
Competitive Analysis:
The report offers the appropriate analysis of the key organizations/companies involved within the global recombinant therapeutic antibodies and proteins market, along with a comparative evaluation primarily based on their product offerings, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborative analysis focusing on the current news and developments of the companies, which includes product development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
Top Companies in Global Recombinant Therapeutic Antibodies and Proteins Market
- F. Hoffmann-La Roche Ltd.
- Pfizer Inc.
- AbbVie Inc.
- Johnson & Johnson
- Amgen Inc.
- Novartis AG
- Sanofi S.A.
- Bristol Myers Squibb
- Eli Lilly and Company
- Merck & Co., Inc.
- Biogen Inc.
- AstraZeneca PLC
Government Initiatives
|
Country |
Key Government Initiatives |
|
UK |
The MHRA (Medicines and Healthcare products Regulatory Agency) promotes biologics innovation via the Innovative Licensing and Access Pathway (ILAP), which accelerates access to monoclonal antibodies and protein-based therapies. The UK also encourages biosimilar uptake through NHS policies. |
|
China |
The National Medical Products Administration (NMPA) has introduced regulatory reforms such as priority review and conditional approval pathways for biologics. The 14th Five-Year Plan for Biopharmaceuticals prioritizes monoclonal antibody drugs and recombinant protein innovation. |
|
Japan |
The PMDA (Pharmaceuticals and Medical Devices Agency) promotes biologics via the Sakigake Designation System and Conditional Early Approval System, which accelerate approval of innovative recombinant therapies and antibody drugs. |
Study on the Supply, Demand, Distribution, and Market Environment of the Recombinant Therapeutic Antibodies and Proteins Market
The Global Recombinant Therapeutic Antibodies and Proteins Market is shaped by strong demand driven by rising prevalence of chronic diseases, increasing adoption of biologics, and advancements in personalized medicine. Supply is supported by robust biopharmaceutical manufacturing capabilities, though it faces challenges such as high production costs and capacity constraints. Distribution networks rely on cold chain logistics and specialized handling to maintain product stability and efficacy. The market environment is influenced by stringent regulatory frameworks and growing investments in R&D. Raw materials, including cell culture media, expression systems (mammalian or microbial), reagents, and purification resins, play a critical role in production efficiency and quality. Fluctuations in raw material availability and costs can impact manufacturing timelines, making supply chain optimization essential for sustainable market growth.
Price Analysis and Consumer Behaviour Analysis
Price analysis and consumer behavior in the Global Recombinant Therapeutic Antibodies and Proteins Market are shaped by high production complexity and value-based pricing strategies. Prices of biologics remain significantly high due to expensive R&D, clinical trials, and advanced manufacturing processes, even though actual production costs have declined over time. Companies often adopt premium pricing based on therapeutic effectiveness, unmet medical needs, and patent exclusivity. The introduction of biosimilars has started to reduce prices, increasing affordability and competition. Consumer behavior is largely influenced by physician recommendations, reimbursement policies, and treatment outcomes rather than price sensitivity alone. However, in developing regions, high costs limit accessibility, making patients and healthcare providers more inclined toward cost-effective alternatives, thereby driving demand for biosimilars and value-based healthcare solutions.
Market Segmentation
The Recombinant Therapeutic Antibodies and Proteins Market share is classified into drug class, host cell type, and end-user.
The monoclonal antibodies segment dominated the market in 2025, approximately 50%, and is projected to grow at a substantial CAGR during the forecast period.
Based on the drug class, the recombinant therapeutic antibodies and proteins market is divided into plasma proteins, growth factors, fusion proteins, monoclonal antibodies, hormones, enzymes, and coagulation factors. Among these, the monoclonal antibodies segment dominated the market in 2025, approximately 50%, and is projected to grow at a substantial CAGR during the forecast period. The growth of the monoclonal antibodies segment is growing due to the treatment demonstrates particular effectiveness because its high specificity enables it to effectively treat cancer and autoimmune diseases while receiving more clinical approvals. The company achieves its substantial market presence through three main factors, which include its ongoing product developments, its expanding range of medical treatments, and its increasing use of targeted biologic medications.
- The mammalian cells segment dominated the market in 2025, approximately 65%, and is projected to grow at a substantial CAGR during the forecast period.
Based on the host cell type, the recombinant therapeutic antibodies and proteins market is divided into mammalian cells, bacterial cells, and yeast cells. Among these, the mammalian cells segment dominated the market in 2025, approximately 65%, and is projected to grow at a substantial CAGR during the forecast period. The mammalian cells segment dominated due to the system producing intricate, high-grade proteins that achieve precise protein folding and complete post-translational modification processes. The system provides essential support for monoclonal antibodies and advanced biologics because it ensures their safety and effectiveness while meeting regulatory standards. This drives the company's main market share and positive growth forecast.
- The biotechnology and pharmaceutical companies drugs segment dominated the market in 2025, approximately 68%, and is projected to grow at a substantial CAGR during the forecast period.
Based on the end-user, the recombinant therapeutic antibodies and proteins market is divided into biotechnology and pharmaceutical companies, research organizations and laboratories, and diagnostic centers. Among these, the biotechnology and pharmaceutical companies segment dominated the market in 2025, approximately 68%, and is projected to grow at a substantial CAGR during the forecast period. The growth of the biotechnology and pharmaceutical companies segment is growing due to the companies operate through three main resources, which include their strong research and development capabilities their extensive manufacturing facilities, and their wide network of market distribution partners. The companies maintain their market position and continuous business expansion by developing new products through their research efforts and their partnership agreements, which enable them to create and introduce new medical products.
Strategies to Implement for Growth of the Market in Non-Leading Regions
To drive growth of the Global Recombinant Therapeutic Antibodies and Proteins Market in non-leading regions, companies should focus on improving affordability and accessibility through localized manufacturing and the adoption of cost-effective biosimilars. Strategic partnerships with regional healthcare providers and governments can strengthen distribution and market penetration. Enhancing regulatory alignment and simplifying approval pathways can accelerate product availability. Investment in healthcare infrastructure, including cold chain logistics, is essential to support biologics distribution. Companies should also increase awareness through physician education and patient outreach programs to boost acceptance of advanced therapies. Leveraging digital health technologies and telemedicine can expand reach in underserved areas. Additionally, flexible pricing models and reimbursement support can improve patient access, ultimately fostering sustainable market growth in emerging regions.
Regional Segment Analysis of the Recombinant Therapeutic Antibodies and Proteins Market
- North America (U.S., Canada, Mexico)
- Europe (Germany, France, U.K., Italy, Spain, Rest of Europe)
- Asia-Pacific (China, Japan, India, Rest of APAC)
- South America (Brazil and the Rest of South America)
- The Middle East and Africa (UAE, South Africa, Rest of MEA)
North America is anticipated to hold the largest share of approximately 38% the recombinant therapeutic antibodies and proteins market over the forecast period.
North America is anticipated to hold the largest share of approximately 38% the recombinant therapeutic antibodies and proteins market over the forecast period. The dominance of this region is primarily driven by its research and development expenditures, its commitment to developing new biologic treatments, its existing regulatory environment, and its rising chronic disease rates. The region maintains its substantial market share because of its research and development expenditures, its commitment to developing new biologic treatments, its existing regulatory environment, and its rising chronic disease rates.
Asia Pacific is expected to grow at a rapid CAGR of approximately 13.5% in the recombinant therapeutic antibodies and proteins market during the forecast period.
Asia Pacific is expected to grow at a rapid CAGR of approximately 13.5% in the recombinant therapeutic antibodies and proteins market during the forecast period. This growth is driven by the healthcare system needs to expand because there are more patients and more people with chronic diseases. The region experiences a strong compound annual growth rate because of rising investments in biotechnology, its ability to produce affordable products, government support, and rising biosimilar adoption.
Europe is the 3rd largest region to grow in the recombinant therapeutic antibodies and proteins market during the period.
Europe is the third-largest region in the recombinant therapeutic antibodies and proteins market due to its well-established healthcare systems, strong regulatory framework, and growing adoption of advanced biologics. Increasing investments in research and development, the presence of leading pharmaceutical companies, and rising demand for biosimilars are supporting steady market growth across the region.
Future Market Trends in Global Recombinant Therapeutic Antibodies and Proteins Market: -
1. Growth of Personalized and Targeted Therapies
The market is increasingly shifting toward personalized medicine, where recombinant antibodies and proteins are designed to target specific disease pathways with high precision. This trend is driven by rising chronic diseases and demand for effective treatments with fewer side effects. Advanced technologies like protein engineering and genomics enable customized biologics, improving patient outcomes and expanding therapeutic applications globally.
2. Rising Adoption of Biosimilars and Cost-Effective Biologics
Biosimilars are gaining strong momentum as patents of blockbuster biologics expire, offering more affordable alternatives without compromising efficacy. This trend is improving accessibility in emerging markets and intensifying competition among pharmaceutical companies. Increasing healthcare cost pressures and regulatory support for biosimilars are further accelerating their adoption across global healthcare systems.
3. Integration of AI and Advanced Biomanufacturing Technologies
Artificial intelligence, machine learning, and next-generation expression systems are transforming drug discovery and production processes. These technologies enhance protein design, increase manufacturing efficiency, and reduce development timelines. Additionally, innovations like continuous bioprocessing and single-use systems are improving scalability and cost efficiency, making biologics more accessible and commercially viable.
Recent Development
- In January 2026, IL-17 inhibitors targeting plaque psoriasis and ankylosing spondylitis, such as gumokimab and roconkibart (IL-17A monoclonal antibodies), have had their New Drug Applications (NDAs) accepted by China’s National Medical Products Administration (NMPA) regulatory review cycle, reflecting continued expansion of biologic therapies in autoimmune diseases.
- In December 2025, Veligrotug, an anti-IGF-1R monoclonal antibody for Thyroid Eye Disease (TED), received FDA Breakthrough Therapy Designation, and its Biologics License Application (BLA) was accepted by the U.S. FDA.
- In September 2025, Mim8 (denecimig), a recombinant bispecific monoclonal antibody (FVIIIa-mimetic) developed for hemophilia A, had its Biologics License Application (BLA) submitted to the U.S. FDA.
- In March 2025, Bentracimab, a recombinant human monoclonal antibody Fab fragment developed to reverse the antiplatelet effects of ticagrelor, received FDA Biologics License Application (BLA) acceptance with Priority Review, and pivotal Phase 3 REVERSE-IT trial data were presented.
How is Recent Developments Helping the Market?
Recent developments are significantly accelerating the growth of the global recombinant therapeutic antibodies and proteins market by enhancing innovation, efficiency, and accessibility. Advances in biomanufacturing technologies, such as continuous processing and improved expression systems, have increased production efficiency and reduced costs. The integration of artificial intelligence and protein engineering enables faster drug discovery and optimized molecular design, leading to more effective therapies. Additionally, the growing focus on personalized medicine and precision therapeutics is expanding the application scope of recombinant antibodies and proteins. Strategic collaborations between biotechnology firms, research institutions, and pharmaceutical companies are further driving innovation and market expansion. Moreover, increasing investments in r&d and supportive government initiatives are strengthening the market environment, resulting in sustained growth and a projected strong cagr in the coming years.
Market Segment
This study forecasts revenue at global, regional, and country levels from 2020 to 2035. Decision Advisors has segmented the Recombinant therapeutic antibodies and proteins market based on the below-mentioned segments:
Global Recombinant Therapeutic Antibodies and Proteins Market, Drug Class
- Plasma Proteins
- Growth Factors
- Fusion Proteins
- Monoclonal Antibodies
- Hormones
- Enzymes
- Coagulation Factors
Global Recombinant Therapeutic Antibodies and Proteins Market, By Host Cell Type
- Mammalian Cells
- Bacterial Cells
- Yeast Cells
Global Recombinant Therapeutic Antibodies and Proteins Market, By End-User
- Biotechnology and Pharmaceutical Companies
- Research Organizations and Laboratories
- Diagnostic Centers
Global Recombinant Therapeutic Antibodies and Proteins Market, By Regional Analysis
- North America
- US
- Canada
- Mexico
- Europe
- Germany
- UK
- France
- Italy
- Spain
- Russia
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- Australia
- Rest of Asia Pacific
- South America
- Brazil
- Argentina
- Rest of South America
- Middle East & Africa
- UAE
- Saudi Arabia
- Qatar
- South Africa
- Rest of the Middle East & Africa
Frequently Asked Questions (FAQ)
Q What role do biosimilars play in shaping market competition?
A. Biosimilars are increasing competition by offering cost-effective alternatives to patented biologics. As patents expire, more companies enter the market, reducing treatment costs and expanding patient access, especially in emerging economies, thereby reshaping pricing strategies and market dynamics.
Q. How does regulatory approval impact market growth?
A. Strict regulatory frameworks ensure safety and efficacy but can delay product launches and increase development costs. However, streamlined approval pathways for biologics and biosimilars in some regions are helping accelerate market entry and encouraging innovation.
Q. What challenges are associated with large-scale biologics manufacturing?
A. Large-scale production requires advanced infrastructure, high capital investment, and strict quality control. Maintaining consistency, avoiding contamination, and managing complex supply chains are key challenges that can impact production timelines and costs.
Q. How is the market contributing to advancements in precision medicine?
A. The market supports precision medicine by enabling the development of targeted therapies that act on specific disease pathways. Recombinant antibodies and proteins improve treatment effectiveness, reduce side effects, and allow personalized treatment approaches based on patient-specific biological markers.
Check Licence
Choose the plan that fits you best: Single User, Multi-User, or Enterprise solutions tailored for your needs.
We Have You Covered
- 24/7 Analyst Support
- Clients Across the Globe
- Tailored Insights
- Technology Tracking
- Competitive Intelligence
- Custom Research
- Syndicated Market Studies
- Market Overview
- Market Segmentation
- Growth Drivers
- Market Opportunities
- Regulatory Insights
- Innovation & Sustainability
Report Details
| Scope | Global |
| Pages | 240 |
| Delivery | PDF & Excel via Email |
| Language | English |
| Release | Apr 2026 |
| Access | Download from this page |
