Global Regulatory Affairs Outsourcing Market
Global Regulatory Affairs Outsourcing Market Size, Share, By Service (Regulatory Writing and Publishing, Regulatory Submissions, Clinical Trial Applications, Services Registrations, Regulatory Consulting and Legal Representation), By Indication (Oncology, Neurology, Cardiology, Immunology)By End Users (Mid-Size Pharmaceutical Companies, Large Pharmaceutical Companies, Biotechnology Companies, Medical Device Companies, Food and Beverage Companies), and By Region (North America, Europe, Asia-Pacific, Latin America, Middle East & Africa), Analysis and Forecast 2026-2035
Report Overview
Table of Contents
Market Snapshot
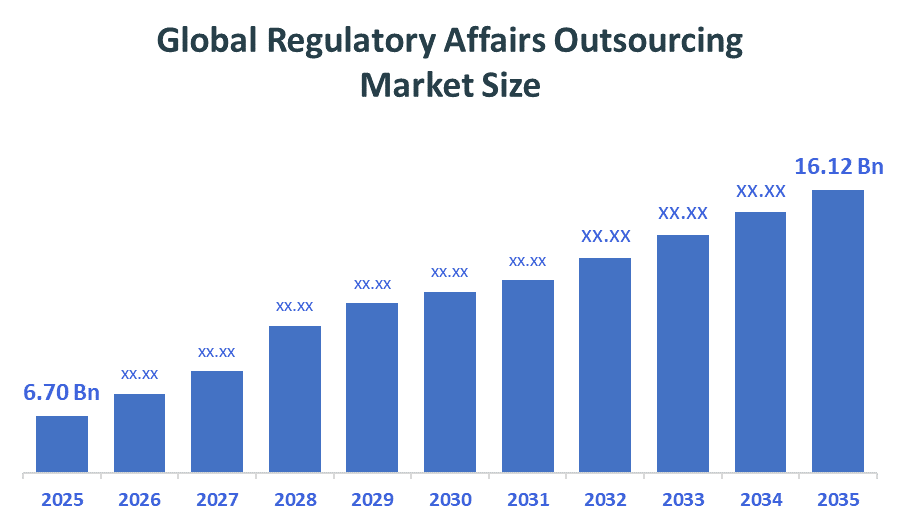
- Global Regulatory Affairs Outsourcing Market Size (2025): USD 6.70Billion
- Projected Global Regulatory Affairs Outsourcing Market Size (2035): USD 16.12Billion
- Global Regulatory Affairs Outsourcing Market Compound Annual Growth Rate (CAGR): 9.18%
- Largest Regional Market: North America
- Fastest Growing Region: Asia-Pacific
- Market 3rd Largest Region: Europe
- Base Year: 2025
- Historical Period: 2021–2024
- Forecast Period: 2026–2035
Market Overview/ Introduction
The Global Regulatory Affairs Outsourcing Market refers to the industry where pharmaceutical, biotechnology, and medical device companies delegate their regulatory tasks to external service providers who ensure their compliance with international standards. Regulatory affairs outsourcing handles all aspects of product approval, together with the necessary documentation and submission processes, and post-market compliance through dedicated third-party services. The market experiences strong growth because of major factors, which include increasing regulatory complexity, rising research and development activities, and the requirement to decrease operational expenses. Future opportunities will emerge through the implementation of AI-based regulatory solutions and market entry into developing regions and increasing demand for biologics and personalized medicine products. The globalization of clinical trials, combined with changing compliance standards, has led companies to depend on expert outsourcing partners, which results in faster approval processes and better operational performance.
- The International Council for Harmonisation (ICH) develops global guidelines that standardize pharmaceutical regulatory requirements across the US, EU, and Japan. It harmonizes submission formats like CTD/eCTD, reducing duplication but increasing compliance complexity, thereby driving demand for regulatory writing, publishing, and outsourcing services.
- The European Medicines Agency (EMA) supports work-sharing and reliance procedures enabling coordinated assessment of medicines across EU member states. Through centralized evaluation and mutual recognition mechanisms, it streamlines approvals while increasing complexity in multi-country submissions, driving demand for regulatory affairs outsourcing services.
Notable Insights: -
- North America is anticipated to hold the largest share of approximately 42% in the regulatory affairs outsourcing market over the forecast period.
- Asia Pacific is expected to grow at a rapid CAGR of approximately 10 % in the regulatory affairs outsourcing market during the forecast period.
- The regulatory writing and publishing segment dominated the market in 2025, approximately 27.4 %, and is projected to grow at a substantial CAGR during the forecast period.
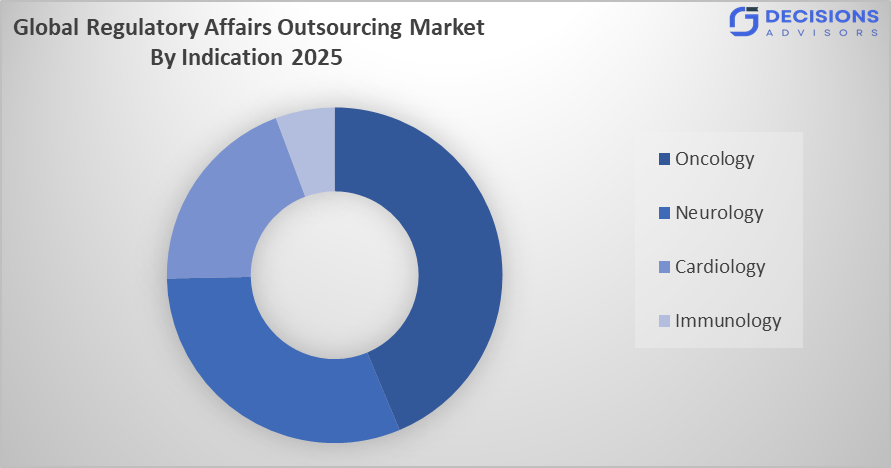
- The oncology segment dominated the market in 2025, approximately 26.9%, and is projected to grow at a substantial CAGR during the forecast period.
- The large pharmaceutical companies segment dominated the market in 2025, approximately 40%, and is projected to grow at a substantial CAGR during the forecast period.
- The compound annual growth rate of the Global Regulatory Affairs Outsourcing Market is 9.18%.
- The market is likely to achieve a valuation of USD 16.12Billion by 2035.
What is role of technology in grooming the market?
Technology plays a pivotal role in grooming the Global Regulatory Affairs Outsourcing Market by enhancing efficiency, accuracy, and compliance management. Advanced digital tools such as artificial intelligence, machine learning, and regulatory information management systems (RIMS) streamline the preparation, submission, and tracking of regulatory documents across multiple regions. Automation reduces manual errors and accelerates approval timelines, enabling faster product launches. Cloud-based platforms facilitate real-time collaboration between pharmaceutical companies and outsourcing partners, improving transparency and data accessibility. Additionally, data analytics helps organizations stay updated with evolving global regulations and predict compliance risks. Technologies like natural language processing support faster review of complex regulatory texts. Overall, technology empowers outsourcing providers to deliver cost-effective, scalable, and high-quality regulatory services, thereby driving market growth and increasing reliance on specialized external partners.
Market Drivers
The Global Regulatory Affairs Outsourcing Market is driven by increasing R&D activities and the rising costs of maintaining in-house regulatory teams, which lead companies to seek external expertise for their drug development and approval processes. The need for specialized regulatory assistance grows because of the strict government regulations that keep changing and apply to both pharmaceuticals and medical devices. The rising demand for personalized medicines and biologics requires companies to develop their specialized knowledge through outsourcing relationships. The need for local regulatory knowledge arises because companies expand their operations into different international markets. Digital technologies, which include artificial intelligence and machine learning together with big data analytics, have become essential for companies that want to achieve cost savings and compliance through outsourcing partnerships with digital service providers.
Restrain
The Global Regulatory Affairs Outsourcing Market faces restraints such as data security concerns, strict regulatory variations across regions, and high dependency on third-party providers. Limited in-house control, communication gaps, and compliance risks may delay approvals. Additionally, the high cost of advanced regulatory technologies and the lack of skilled professionals can hinder market growth.
Competitive Analysis:
The report offers the appropriate analysis of the key organizations/companies involved within the global regulatory affairs outsourcing market, along with a comparative evaluation primarily based on their product offerings, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborative analysis focusing on the current news and developments of the companies, which includes product development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
Top Companies in Global Regulatory Affairs Outsourcing Market
- IQVIA
- ICON plc
- Thermo Fisher Scientific (PPD)
- Labcorp Drug Development
- Syneos Health
- Parexel International
- Charles River Laboratories
- Freyr Solutions
- ProPharma Group
- Celegence
- Medpace
- WuXi AppTec
Government Initiatives
|
Country |
Key Government Initiatives |
|
US |
The U.S. Food and Drug Administration has introduced initiatives such as Project Orbis and Real-Time Oncology Review to accelerate drug approvals and enhance global regulatory collaboration. Additionally, the FDA is promoting AI-based regulatory decision-making frameworks and structured data submissions (e.g., SPL, IDMP), improving efficiency and increasing demand for regulatory outsourcing services. |
|
Europe |
The European Medicines Agency is advancing regulatory harmonization through the European Medicines Regulatory Network (EMRN) and digital transformation initiatives. Programs such as AI integration strategies, regulatory optimization groups, and international collaboration (e.g., WHO-listed authority status) aim to standardize processes, boosting outsourcing demand for compliance and digital expertise. |
|
India |
The Central Drugs Standard Control Organization, under the Ministry of Health, is strengthening regulatory frameworks through digitalization and faster approval pathways. India is also aligning with global standards (ICH guidelines) and encouraging clinical trials, which supports outsourcing growth by increasing the need for regulatory expertise and compliance management in global submissions. |
Study on the Supply, Demand, Distribution, and Market Environment of the Regulatory Affairs Outsourcing Market
The Global Regulatory Affairs Outsourcing Market is shaped by a dynamic balance of supply, demand, distribution, and market environment. Supply is driven by specialized service providers offering regulatory consulting, submission management, and compliance solutions, supported by skilled professionals and advanced digital tools. Demand is increasing due to rising pharmaceutical R&D activities, complex global regulations, and the need for faster product approvals. Distribution is primarily service-based, leveraging digital platforms and global delivery models to serve clients efficiently across regions. The market environment is influenced by stringent regulatory frameworks and increasing outsourcing trends among biotech and pharma companies. Additionally, key raw materials in this market include regulatory data, documentation systems, software platforms, and skilled human expertise, which are essential inputs for delivering accurate, timely, and compliant regulatory services globally.
Price Analysis and Consumer Behaviour Analysis
Price analysis in the Global Regulatory Affairs Outsourcing Market is influenced by service complexity, geographic scope, and regulatory requirements. Costs vary depending on services such as consulting, submission management, and clinical trial support, with higher pricing for specialized expertise and multi-country compliance. Competitive pressure and the need to reduce operational costs encourage flexible pricing models, including project-based and long-term contracts. Consumer behaviour is driven by pharmaceutical and biotech companies seeking cost efficiency, faster approvals, and access to skilled professionals. Organizations increasingly prefer outsourcing to reduce in-house burden and ensure regulatory accuracy. Demand is also shaped by globalization and evolving regulations, pushing companies to select providers offering advanced technologies, reliability, and end-to-end regulatory solutions, thereby strengthening long-term partnerships and market growth.
Market Segmentation
The Regulatory Affairs Outsourcing Market share is classified into service, indication, and end-user.
- The regulatory writing and publishing segment dominated the market in 2025, approximately 27.4%, and is projected to grow at a substantial CAGR during the forecast period.
Based on the service, the regulatory affairs outsourcing market is divided into regulatory writing and publishing, regulatory submissions, clinical trial applications, services registrations, regulatory consulting, and legal representation. Among these, the regulatory writing and publishing segment dominated the market in 2025, approximately 27.4%, and is projected to grow at a substantial CAGR during the forecast period. The growth of the regulatory writing and publishing segment is growing due to the critical need for accurate, standardized, and compliant documentation in regulatory submissions. Increasing clinical trials and complex global regulations require expert preparation of dossiers, reports, and filings, driving demand for specialized outsourcing services that ensure faster approvals and reduced risk of regulatory errors.
- The oncology segment dominated the market in 2025, approximately 26.9%, and is projected to grow at a substantial CAGR during the forecast period.
Based on the indication, the regulatory affairs outsourcing market is divided into oncology, neurology, cardiology, and immunology. Among these, the oncology segment dominated the market in 2025, approximately 26.9%, and is projected to grow at a substantial CAGR during the forecast period. The oncology segment dominated due to the high number of cancer-related clinical trials and continuous development of innovative therapies such as biologics and personalized medicines. Stringent regulatory requirements for oncology drugs increase the need for specialized expertise, driving outsourcing demand to ensure accurate submissions, faster approvals, and compliance across global markets.
- The large pharmaceutical companies drug segment dominated the market in 2025, approximately 40%, and is projected to grow at a substantial CAGR during the forecast period.
Based on the end-user, the regulatory affairs outsourcing market is divided into mid-size pharmaceutical companies, large pharmaceutical companies, biotechnology companies, medical device companies, and food and beverage companies. Among these, the large pharmaceutical companies segment dominated the market in 2025, approximately 40%, and is projected to grow at a substantial CAGR during the forecast period. The growth of the large pharmaceutical companies segment is growing due to their extensive R&D pipelines, global product portfolios, and high volume of regulatory submissions. These firms increasingly rely on outsourcing to manage complex compliance requirements, reduce operational costs, and accelerate approvals, driving strong demand for specialized regulatory services and long-term outsourcing partnerships.
Strategies to Implement for Growth of the Market in Non-Leading Regions
Strategies to drive growth of the Global Regulatory Affairs Outsourcing Market in non-leading regions (such as emerging markets in Asia, Latin America, and Africa) focus on localization, cost efficiency, and capability building. Establishing regional hubs with skilled, cost-effective talent enhances service delivery and attracts global clients. Partnerships with local regulatory bodies and firms help navigate complex and evolving compliance frameworks more effectively. Additionally, adopting advanced digital tools like AI and automation improves efficiency and reduces approval timelines. Companies should also invest in training programs to bridge skill gaps and ensure regulatory expertise. Expanding multilingual and region-specific services supports better market penetration. Furthermore, leveraging flexible outsourcing models and strategic collaborations enables faster entry into untapped markets, driving sustainable growth.
Regional Segment Analysis of the Regulatory Affairs Outsourcing Market
- North America (U.S., Canada, Mexico)
- Europe (Germany, France, U.K., Italy, Spain, Rest of Europe)
- Asia-Pacific (China, Japan, India, Rest of APAC)
- South America (Brazil and the Rest of South America)
- The Middle East and Africa (UAE, South Africa, Rest of MEA)
North America is anticipated to hold the largest share of approximately 42% the regulatory affairs outsourcing market over the forecast period.
North America is anticipated to hold the largest share of approximately 42% the regulatory affairs outsourcing market over the forecast period. The dominance of this region is primarily driven by its strong presence of leading pharmaceutical and biotechnology companies, high R&D investments, and advanced healthcare infrastructure. Additionally, stringent regulatory requirements from agencies like the FDA increase the need for specialized outsourcing services, driving sustained demand and market leadership in the region.
Asia Pacific is expected to grow at a rapid CAGR of approximately 10% in the regulatory affairs outsourcing market during the forecast period.
Asia Pacific is expected to grow at a rapid CAGR of approximately 10% in the regulatory affairs outsourcing market during the forecast period. This growth is driven by its cost-effective outsourcing environment, expanding pharmaceutical and biotechnology industries, and the increasing number of clinical trials. Additionally, the availability of skilled professionals, supportive government initiatives, and growing adoption of advanced technologies are attracting global companies to outsource regulatory services in the region.
Europe is the 3rd largest region to grow in the regulatory affairs outsourcing market during the period.
Europe is the third-largest region in the regulatory affairs outsourcing market due to its well-established pharmaceutical industry, strong regulatory framework, and increasing focus on compliance. The presence of key market players, rising clinical trial activities, and evolving regulations encourages companies to outsource regulatory functions for efficiency and cost optimization.
Future Market Trends in Global Regulatory Affairs Outsourcing Market: -
1. Increasing Adoption of AI and Automation
The integration of artificial intelligence and automation will transform regulatory processes by reducing manual workload and improving accuracy. These technologies enable faster document preparation, real-time compliance tracking, and predictive insights, helping companies accelerate approvals and reduce costs, thereby increasing reliance on outsourcing partners with advanced digital capabilities.
2. Growth of Emerging Markets Outsourcing
Pharmaceutical companies are increasingly outsourcing regulatory activities to emerging regions due to cost advantages and expanding talent pools. Countries in Asia-Pacific and Latin America offer skilled professionals at lower costs, encouraging global firms to establish partnerships and regional hubs, thus driving market expansion beyond traditional regions.
3. Rising Demand for End-to-End Regulatory Services
Clients are shifting toward vendors offering comprehensive, end-to-end regulatory solutions, from strategy development to post-market surveillance. This trend is driven by the need for streamlined processes, reduced complexity, and better coordination, leading companies to prefer long-term partnerships with full-service outsourcing providers.
Recent Development
- In September 2025, Rimsys launched its AI-powered platform, “Rimsys AI,” which includes embedded AI agents designed to streamline regulatory submissions, clinical data review, regulatory change monitoring, and compliance workflows. The system enhances efficiency in regulatory operations and accelerates decision-making, supporting faster clinical trial and market approval processes.
- In June 2025, the U.S. Food and Drug Administration (FDA) launched its internal generative AI system “Elsa”, designed to enhance clinical protocol review, summarize safety and adverse event data, and support regulatory scientific evaluations. The system improves efficiency in clinical data review and regulatory decision-making, accelerating internal workflows and indirectly increasing demand for specialized regulatory compliance and outsourcing services.
- Ongoing, 2024–2026, Regulatory consulting firms and life sciences technology providers have integrated AI-driven compliance tools to streamline pharmaceutical regulatory submissions, automate documentation review, and improve accuracy. These advancements enhance regulatory efficiency and increase demand for outsourced regulatory affairs, submission management, and compliance services.
- In October 2024, a strategic outsourcing partnership was formed between NAMSA and Terumo Corporation to accelerate the regulatory approval and commercialization of Terumo’s global medical device portfolio. The collaboration leverages clinical research, regulatory consulting, and testing services to reduce development timelines and improve market access efficiency.
How is Recent Developments Helping the Market?
Recent developments are significantly supporting the growth of the Global Regulatory Affairs Outsourcing Market by improving efficiency, compliance, and global reach. The adoption of advanced technologies such as artificial intelligence, automation, and cloud-based regulatory platforms has streamlined submission processes and reduced approval timelines. Increasing harmonization of international regulations and digital transformation initiatives by regulatory authorities are making cross-border compliance more manageable. Additionally, rising strategic collaborations, mergers, and acquisitions among key players are expanding service portfolios and geographic presence. The growing complexity of clinical trials and rapid expansion of biologics and personalized medicines have further increased the demand for specialized regulatory expertise. These advancements enable outsourcing providers to deliver faster, more accurate, and cost-effective solutions, thereby strengthening client confidence and accelerating overall market growth.
Market Segment
This study forecasts revenue at global, regional, and country levels from 2020 to 2035. Decision Advisors has segmented the Regulatory affairs outsourcing market based on the below-mentioned segments:
Global Regulatory Affairs Outsourcing Market, Service
- Regulatory Writing and Publishing
- Regulatory Submissions
- Clinical Trial Applications
- Services Registrations
- Regulatory Consulting and Legal Representation
Global Regulatory Affairs Outsourcing Market, By Indication
- Oncology
- Neurology
- Cardiology
- Immunology
Global Regulatory Affairs Outsourcing Market, By End-User
- Mid-Size Pharmaceutical Companies
- Large Pharmaceutical Companies
- Biotechnology Companies
- Medical Device Companies
- Food and Beverage Companies
Global Regulatory Affairs Outsourcing Market, By Regional Analysis
- North America
- US
- Canada
- Mexico
- Europe
- Germany
- UK
- France
- Italy
- Spain
- Russia
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- Australia
- Rest of Asia Pacific
- South America
- Brazil
- Argentina
- Rest of South America
- Middle East & Africa
- UAE
- Saudi Arabia
- Qatar
- South Africa
- Rest of the Middle East & Africa
Frequently Asked Questions (FAQ)
Q What challenges do companies face while outsourcing regulatory affairs?
A. Companies often face challenges such as data confidentiality risks, lack of direct control over processes, communication gaps across regions, and dependency on third-party vendors. Additionally, differences in regulatory standards across countries can complicate coordination and increase the risk of compliance delays.
Q. How does globalization impact the regulatory affairs outsourcing market?
A. Globalization increases the need for regulatory outsourcing as companies expand into multiple countries with diverse regulations. It drives demand for local expertise, multilingual documentation, and region-specific compliance strategies, encouraging firms to partner with outsourcing providers for efficient global market entry.
Q. Why are small and mid-size pharmaceutical companies increasingly adopting outsourcing?
A. Small and mid-size companies adopt outsourcing to reduce operational costs, access specialized regulatory expertise, and avoid building expensive in-house teams. This helps them focus on core research activities while ensuring efficient regulatory approvals and faster time-to-market.
Q. What role do partnerships and collaborations play in this market?
A. Strategic partnerships and collaborations enable companies to expand service capabilities, enhance geographic reach, and access advanced technologies. These alliances help outsourcing firms offer integrated solutions, improve service efficiency, and strengthen their competitive position in the global market.
Check Licence
Choose the plan that fits you best: Single User, Multi-User, or Enterprise solutions tailored for your needs.
We Have You Covered
- 24/7 Analyst Support
- Clients Across the Globe
- Tailored Insights
- Technology Tracking
- Competitive Intelligence
- Custom Research
- Syndicated Market Studies
- Market Overview
- Market Segmentation
- Growth Drivers
- Market Opportunities
- Regulatory Insights
- Innovation & Sustainability
Report Details
| Scope | Global |
| Pages | 240 |
| Delivery | PDF & Excel via Email |
| Language | English |
| Release | Apr 2026 |
| Access | Download from this page |
