Global Spinal Muscular Atrophy Market
Global Spinal Muscular Atrophy Market Size, Share, and COVID-19 Impact Analysis, By Type (Type 1, Type 2, Type 3, and Type 4), By Treatment (Gene Therapy, Drug, Spinraza, Zolgensma (AVXS-101), Evrysdi, and Others), By Route of Administration (Oral, and Injection), and By Region (North America, Europe, Asia-Pacific, Latin America, Middle East, and Africa), Analysis and Forecast 2025-2035
Report Overview
Table of Contents
Global Spinal Muscular Atrophy Market Size Insights Forecasts to 2035
- The Global Spinal Muscular Atrophy Market Size Was Estimated at USD 4.35 Billion in 2024
- The Market Size is Expected to Grow at a CAGR of around 17.74 % from 2025 to 2035
- The Worldwide Spinal Muscular Atrophy Market Size is Expected to Reach USD 26.21 Billion by 2035
- North America is expected to grow the fastest during the forecast period.
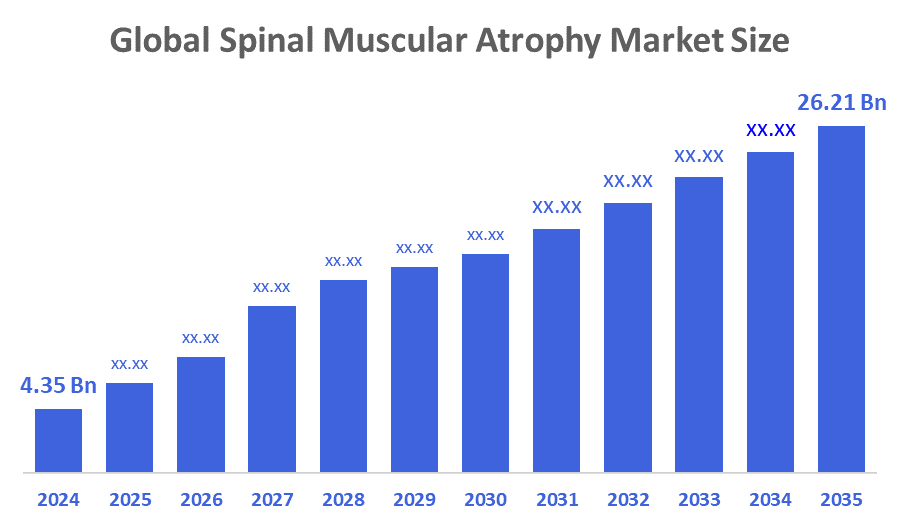
According to a research report published by Decisions Advisors and Consulting, The Global Spinal Muscular Atrophy Market Size Was Worth Around USD 4.35 Billion In 2024 And Is Predicted To Grow To Around USD 26.21 Billion By 2035 With A Compound Annual Growth Rate (CAGR) Of 17.74 % From 2025 To 2035. The increasing diagnosis and prevalence of spinal muscular atrophy mostly drive the need for treatments. A greater number of patients require therapy as a result of improved screening programs, such as newborn screening for spinal muscular atrophy, which have increased diagnosis rates. It is now possible to diagnose spinal muscular atrophy earlier and treat it more effectively as awareness of the problem grows, especially in newborns and infants. Additionally, more medical professionals can now identify and treat spinal muscular atrophy at earlier stages, improving patient outcomes.
Market Overview
Spinal muscular atrophy (SMA) is a rare genetic disorder that affects the motor nerves of the spinal cord and results in muscle weakness and wasting. There are four different types of spinal muscular atrophy, and they are classified depending on the symptoms and age of onset. These include type 1, type 2, type 3, and type 4 SMA. Some of the common symptoms of spinal muscular atrophy include movement difficulties, weakness of limbs, poor muscle tone, delayed motor skills, contractures of joints, etc. Other symptoms of spinal muscular atrophy include tremors, scoliosis, breathing difficulties, and speaking or swallowing difficulties. The diagnosis of spinal muscular atrophy (SMA) can be made by a combination of tests such as clinical examination, genetic tests, and imaging studies. In addition to blood tests to detect genetic abnormalities related to spinal muscular atrophy, a physical examination of a patient can also be carried out to determine muscular strength and tone. Various imaging studies, such as MRI or electromyography, can also be used to diagnose spinal muscular atrophy.
The global spinal muscular atrophy (SMA) patient pool is estimated at 1-2 per 100,000 population (prevalence), with ~75,000-150,000 patients worldwide; incidence 1 in 6,000-10,000 live births (~10,000-16,000 new cases/year).
Collected studies with varying economic data from 32 nations based on the SMA subtype, study design, and region. The highest expenditures were seen in type 1 individuals, who had direct medical expenses of US $187,88 without disease-modifying therapies. The weighted average yearly cost for an SMA patient was US $109,906. Together with frequent hospital stays, substantial caregiver productivity losses, and the need for 2,947 hours of care each year, the non-medical expenses came to US $109,379 per patient.
Report Coverage
This research report categorizes the spinal muscular atrophy market based on various segments and regions, forecasts revenue growth, and analyzes trends in each submarket. The report analyses the key growth drivers, opportunities, and challenges influencing the spinal muscular atrophy market. Recent market developments and competitive strategies, such as expansion, product launch, development, partnership, merger, and acquisition, have been included to draw the competitive landscape in the market. The report strategically identifies and profiles the key market players and analyzes their core competencies in each sub-segment of the spinal muscular atrophy market.
Driving Factors
Moreover, the increasing rate of rare genetic disorders and the growing need to create new treatment regimens for these diseases are mainly fueling the spinal muscular atrophy market. Additionally, the high rate of adoption of medicines such as nusinersen and risdiplam, which are used to increase the levels of a protein called SMN and restore motor function and survival in individuals with spinal muscular atrophy, is also contributing to the growth of this market. Moreover, the implementation of newborn screening for spinal muscular atrophy, which includes tests for the presence of the SMN1 gene, is also boosting the market growth. This can eventually enable effective treatment and possibly a better outcome for children with spinal muscular atrophy. Apart from this, many key players are putting high investment into research and development to create therapies that can treat the underlying cause of spinal muscular atrophy. This is also acting as a major growth-inducing factor for this market. Additionally, the high rate of usage of respiratory assistance, such as non-invasive ventilation and cough assistance devices, to enable individuals with spinal muscular atrophy to breathe, is also creating a favourable market growth scenario.
An international clinical trial has demonstrated that newborns with spinal muscular atrophy (SMA) can safely begin treatment at birth. Researchers tested the oral drug risdiplam, previously approved only for infants two months and older, and found it effective when started as early as 16 days old. Early intervention prevented severe motor delays, improved survival, and offered hope for infants who otherwise face rapid disease progression.
Restraining Factors
High therapy costs, restricted accessibility in low- and middle-income areas, complicated reimbursement processes, and manufacturing difficulties for cutting-edge biologics and gene treatments are some of the reasons limiting the spinal muscular atrophy (SMA) market. Despite high clinical efficacy, these obstacles hinder acceptance and limit the growth of the market as a whole.
Market Segmentation
The spinal muscular atrophy market share is classified into type, treatment, and route of administration.
- The type 1 segment accounted for the largest market share in 2024 and is anticipated to grow at a substantial CAGR over the forecast period.
Based on the type, the spinal muscular atrophy market is divided into type 1, type 2, type 3, and type 4. Among these, the type 1 segment accounted for the largest market share in 2024 and is anticipated to grow at a substantial CAGR over the forecast period. This segment's revenue is influenced by the growing incidence of spinal muscular atrophy in young children and newborns. Type 1 spinal muscular atrophy can now be detected earlier and treated more quickly due to increased awareness and better diagnostic techniques. More families are seeking treatment as a result of the availability of efficient treatments, such as gene therapies and novel drugs, which have greatly improved patient outcomes.
For instance, A Phase II clinical trial (RAINBOWFISH) showed that presymptomatic infants with spinal muscular atrophy (SMA) 1 treated early with Evrysdi® (risdiplam) achieved key motor milestones such as sitting, standing, and walking within typical developmental windows.
- The drug segment accounted for the highest market share in 2024 and is anticipated to grow at a significant CAGR during the forecast period.
Based on the treatment, the spinal muscular atrophy market is divided into gene therapy, drug, Spinraza, Zolgensma (avxs-101), Evrysdi, and others. Among these, the drug segment accounted for the highest market share in 2024 and is anticipated to grow at a significant CAGR during the forecast period. This segment growth, addressing the genetic origins of spinal muscular atrophy, has revolutionised the treatment of the condition. The need for these treatments is being driven by the increasing number of drug approvals and clinical achievements, which have increased patient and healthcare professional knowledge. Additionally, access to these transformative treatments is being improved by pharmaceutical firms' patient assistance programs.
For instance, the UAE has become the second country worldwide to approve Novartis’ gene therapy Itvisma® for spinal muscular atrophy (SMA), expanding access beyond the U.S. and marking a major milestone in global SMA care.
- The injection segment accounted for the highest market revenue in 2024 and is anticipated to grow at a significant CAGR during the forecast period.
Based on the route of administration, the spinal muscular atrophy market is divided into oral and injection. Among these, the injection segment accounted for the highest market revenue in 2024 and is anticipated to grow at a significant CAGR during the forecast period. propelled by its proven effectiveness and quick bloodstream delivery of medicinal substances. Injectable treatments can provide results right away; they are frequently used, especially in severe cases of spinal muscular atrophy, where prompt intervention is crucial. Injection segment supremacy in the treatment of spinal muscular atrophy has been further cemented by improvements in patient compliance and comfort brought about by innovations in injection technologies.
For instance, A new wave of research and regulatory progress is bringing one-time gene therapy within reach for older children with spinal muscular atrophy (SMA). Unlike earlier approvals limited to infants, therapies such as Itvisma® (onasemnogene abeparvovec-brve) are now authorised for patients two years and older, including teens and adults. This expansion means that older children—who previously relied on lifelong treatments like Spinraza or Evrysdi—may soon benefit from a single-dose therapy that directly replaces the defective SMN1 gene.
Regional Segment Analysis of the Spinal Muscular Atrophy Market
- North America (U.S., Canada, Mexico)
- Europe (Germany, France, U.K., Italy, Spain, Rest of Europe)
- Asia-Pacific (China, Japan, India, Rest of APAC)
- South America (Brazil and the Rest of South America)
- The Middle East and Africa (UAE, South Africa, Rest of MEA)
Asia Pacific is anticipated to hold the largest share of the spinal muscular atrophy market over the predicted timeframe.
Asia Pacific is anticipated to hold the largest share of the spinal muscular atrophy market over the predicted timeframe. This can be linked to better healthcare facilities and increased knowledge of uncommon illnesses like spinal muscular atrophy. As more families look for solutions for impacted individuals, the region's expanding population base also adds to the growing demand for efficient treatment choices. Collaborations between international pharmaceutical companies and regional biotech firms provide access to cutting-edge treatments throughout the Asia Pacific. the Japan spinal muscular atrophy treatment market dominated the Asia Pacific region due to its sophisticated healthcare system and high rates of innovative therapy adoption. Patients with spinal muscular atrophy now have more access to effective medicines thanks to Japan's dedication to tackling uncommon diseases through research efforts.
Bangkok Hospital has launched a Precision Medicine Hub in Southeast Asia, positioning itself as a regional leader in advanced therapeutics. The initiative introduces gene therapy for children with spinal muscular atrophy (SMA) and anti-amyloid therapy for early-stage Alzheimer’s disease, aiming to transform patient care with cutting-edge, personalised treatments.
Biogen has introduced a higher-dose version of Spinraza (nusinersen) in Japan, aiming to reinvigorate its spinal muscular atrophy (SMA) franchise after declining sales in 2025. The new formulation, already approved in Europe, is showing stronger-than-expected adoption in Japan, with patients switching back to Spinraza from rival therapies.
North America is expected to grow at a rapid CAGR in the spinal muscular atrophy market during the forecast period. This is propelled by significant investments in R&D and a strong healthcare infrastructure. Leading pharmaceutical firms are present in this area, which promotes innovation and speeds up the release of novel medications. Additionally, early identification and successful treatment approaches are greatly aided by patients' and healthcare professionals' high levels of awareness about spinal muscular atrophy. Additionally, the United States leads clinical studies for novel treatments for spinal muscular atrophy, improving patients' access to state-of-the-art care. Because of these circumstances, the United States is positioned to play a major role in expanding the treatment landscape for spinal muscular atrophy in North America.
Novartis has received FDA approval for Itvisma (onasemnogene abeparvovec-brve), making it the first and only gene replacement therapy available for children two years and older, teens, and adults with spinal muscular atrophy (SMA). This one-time treatment works by replacing the defective SMN1 gene, aiming to restore motor function and reduce the need for lifelong therapies. Phase III studies showed improvements in motor skills and stabilisation across patients regardless of prior SMA treatment history.
Competitive Analysis:
The report offers the appropriate analysis of the key organizations/companies involved within the spinal muscular atrophy market, along with a comparative evaluation primarily based on their product offering, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborative analysis focusing on the current news and developments of the companies, which includes product development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
List of Key Companies
- Astellas Pharma
- Biogen
- Catalyst Pharmaceuticals
- Cytokinetics
- F. Hoffmann-La Roche Ltd
- Ionis Pharmaceuticals
- NMD PHARMA A/S
- Novartis AG
- Pfizer
- Pfizer Inc.
- PTC Therapeutics
- Sarepta Therapeutics
- Scholar Rock, Inc.
- Others
Key Target Audience
- Market Players
- Investors
- End-users
- Government Authorities
- Consulting and Research Firm
- Venture capitalists
- Value-Added Resellers (VARs)
Recent Development
- In November 2025, the European Medicines Agency’s Committee for Medicinal Products for Human Use (CHMP) gave a positive opinion on Biogen’s high-dose nusinersen (Spinraza) regimen for spinal muscular atrophy (SMA). Based on the DEVOTE trial, the higher-dose protocol showed improved motor outcomes and reduced risks compared to the standard 12 mg dose, paving the way for EU approval in early 2026.
- In September 2025, the U.S. Food and Drug Administration (FDA) issued a Complete Response Letter (CRL) to Scholar Rock for its spinal muscular atrophy (SMA) therapy candidate apitegromab. The setback was due to manufacturing observations at Catalent Indiana LLC, a third-party facility, and not related to the drug’s safety or efficacy. Scholar Rock plans to resubmit its application once the issues are resolved.
Market Segment
This study forecasts revenue at global, regional, and country levels from 2020 to 2035. Decisions Advisors has segmented the spinal muscular atrophy market based on the below-mentioned segments:
Global Spinal Muscular Atrophy Market, By Type
- Type 1
- Type 2
- Type 3
- Type 4
Global Spinal Muscular Atrophy Market, By Treatment
- Gene Therapy
- Drug
- Spinraza
- Zolgensma (AVXS-101)
- Evrysdi
- Others
Global Spinal Muscular Atrophy Market, By Route of Administration
- Oral
- Injection
Global Spinal Muscular Atrophy Market, By Regional Analysis
- North America
- US
- Canada
- Mexico
- Europe
- Germany
- UK
- France
- Italy
- Spain
- Russia
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- Australia
- Rest of Asia Pacific
- South America
- Brazil
- Argentina
- Rest of South America
- Middle East & Africa
- UAE
- Saudi Arabia
- Qatar
- South Africa
- Rest of the Middle East & Africa
Frequently Asked Questions (FAQ)
- How does newborn screening impact the long-term treatment outcomes for spinal muscular atrophy patients?
Newborn screening allows SMA to be detected before symptoms appear, enabling physicians to start disease-modifying treatments at a very early stage. Early intervention significantly improves motor function, survival rates, and quality of life for affected infants. It also reduces long-term complications such as respiratory failure and severe muscle degeneration, making early screening programs a crucial component of SMA disease management.
- What role do patient advocacy organisations play in the spinal muscular atrophy market?
Patient advocacy organisations help increase disease awareness, support early diagnosis programs, and advocate for reimbursement and access to innovative therapies. These organisations often collaborate with pharmaceutical companies and regulatory agencies to promote clinical trials, fund research initiatives, and improve healthcare policies related to rare diseases such as SMA.
- How are biosimilars expected to influence the spinal muscular atrophy treatment market in the future?
As patents for leading biologic therapies eventually expire, biosimilar versions may enter the market. Biosimilars can potentially lower treatment costs and increase access to SMA therapies, particularly in developing countries where affordability is a major concern. However, regulatory approval and clinical equivalence requirements may slow the immediate introduction of such alternatives.
- What technological advancements are supporting better diagnosis of spinal muscular atrophy?
Advances in genetic testing technologies, including next-generation sequencing (NGS) and polymerase chain reaction (PCR)-based screening methods, have significantly improved the accuracy and speed of SMA diagnosis. These technologies allow clinicians to detect SMN1 gene deletions earlier and more reliably, enabling timely treatment decisions.
- How could combination therapies shape the future treatment landscape for spinal muscular atrophy?
Combination therapies involving gene therapy, SMN-enhancing drugs, and muscle-strengthening agents may improve therapeutic outcomes for SMA patients. Researchers are investigating whether combining treatments can further enhance motor neuron survival, slow disease progression, and provide long-term functional improvements.
- What challenges exist in conducting clinical trials for spinal muscular atrophy therapies?
Clinical trials for SMA treatments face challenges such as limited patient populations due to the rarity of the disease, ethical considerations when testing new treatments in infants, and difficulties in measuring long-term neurological outcomes. Additionally, recruiting patients across different SMA subtypes can complicate study design and data interpretation.
- How will digital health technologies support spinal muscular atrophy patient management?
Digital health tools such as remote patient monitoring, wearable devices, and telemedicine platforms can help physicians track motor function, respiratory health, and treatment response in SMA patients. These technologies improve long-term disease monitoring, facilitate personalised care, and reduce the need for frequent hospital visits.
Check Licence
Choose the plan that fits you best: Single User, Multi-User, or Enterprise solutions tailored for your needs.
We Have You Covered
- 24/7 Analyst Support
- Clients Across the Globe
- Tailored Insights
- Technology Tracking
- Competitive Intelligence
- Custom Research
- Syndicated Market Studies
- Market Overview
- Market Segmentation
- Growth Drivers
- Market Opportunities
- Regulatory Insights
- Innovation & Sustainability
Report Details
| Scope | Global |
| Pages | 240 |
| Delivery | PDF & Excel via Email |
| Language | English |
| Release | Mar 2026 |
| Access | Download from this page |
