Global Stills Disease Treatment Market
Global Stills disease treatment market, Share, By Drugs (Anakinra, Canakinumab, Tocilizumab), By Therapy Type (Corticosteroids Therapy, Pain Management), By End User (Hospitals, Homecare, Specialty Clinics), and By Region (North America, Europe, Asia-Pacific, Latin America, Middle East & Africa), Analysis and Forecast 2026-2035
Report Overview
Table of Contents
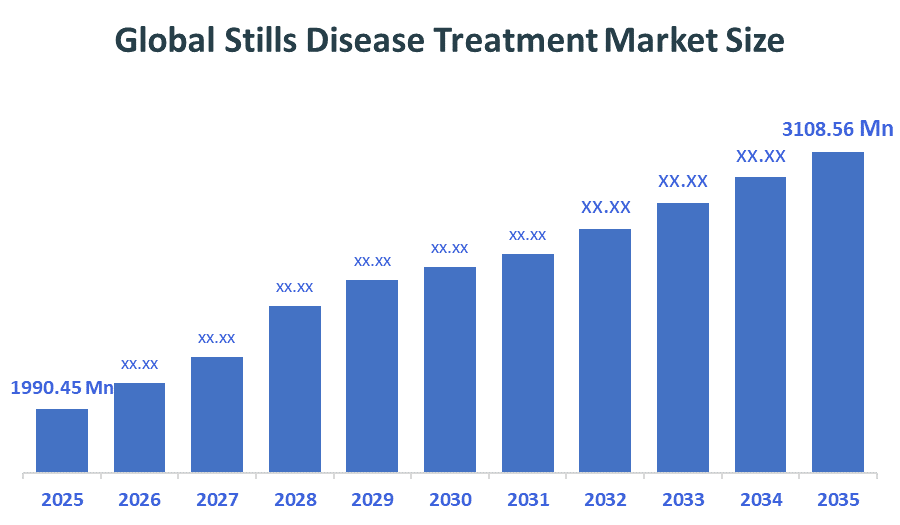
The Global Stills disease treatment market is forecast to grow from USD 1990.45 Million in 2025 to approximately USD 3108.56 Million by 2035. According to Decision Advisors, a detailed research report on the Stills Disease Treatment Market indicates that Targeted Biologics dominates the market, accounting for approximately 65% of the total share globally. Novartis AG the market with an estimated 18-22% global market share. The company reported a 2025 revenue of approximately USD 50 billion, making it one of the most influential forces shaping industry trends and overall market growth.
Market Snapshot
- Global Stills Disease Treatment Market (2025): USD 1990.45 Million
- Projected Global Stills Disease Treatment Market (2035): USD 3108.56 Million
- Global Stills Disease Treatment Market Compound Annual Growth Rate (CAGR): 4.56%
- Largest Regional Market: North America
- Fastest Growing Region: Asia-Pacific
- 2nd Largest Region: Europe
- Base Year: 2025
- Historical Period: 2021–2024
- Forecast Period: 2026–2035
Market Overview/ Introduction
The Global Still’s Disease Treatment Market refers to the worldwide industry focused on developing, manufacturing, and distributing therapies for Still’s disease, a rare inflammatory disorder. Still’s disease treatment includes the use of corticosteroids, immunosuppressants, and advanced biologic drugs such as IL-1 and IL-6 inhibitors that help control systemic inflammation and prevent complications. The market is expected to expand significantly due to rising disease awareness, improved diagnostic capabilities, and increasing adoption of targeted biologic therapies. Future opportunities lie in biosimilars development, personalized medicine, and expanding clinical trials in emerging regions. Market growth is further driven by strong orphan drug incentives, increasing healthcare expenditure, and advancements in biotechnology, which collectively enhance treatment accessibility, effectiveness, and long-term patient outcomes globally.
- The U.S. Orphan Drug Act provides tax credits, fast-track approvals, and 7-year market exclusivity to encourage the development of treatments for rare diseases like Still’s disease. The program reduces R&D risk for pharmaceutical companies and improves patient access to biologics such as IL-1 and IL-6 inhibitors, supporting innovation and treatment availability in the country.
- The European Union offers incentives such as fee reductions, protocol assistance, and 10-year market exclusivity for orphan drugs. This scheme supports research into rare inflammatory diseases like Still’s disease and ensures early access to advanced biologic therapies across member countries under harmonized regulatory pathways.
- Japan’s Ministry of Health provides subsidies, tax incentives, and priority review for orphan drugs affecting fewer than 50,000 patients. The program accelerates approval of treatments for rare diseases, improving access to advanced biologics and strengthening clinical research for conditions like Still’s disease.
Notable Insights: -
- North America is anticipated to hold the largest share of approximately 37% in the Stills Disease Treatment Market over the forecast period.
- Asia Pacific is expected to grow at a rapid CAGR of approximately 11.5% in the Stills Disease Treatment Market during the forecast period.
- The anakinra segment dominated the market in 2025, approximately 43.1%, and is projected to grow at a substantial CAGR during the forecast period.
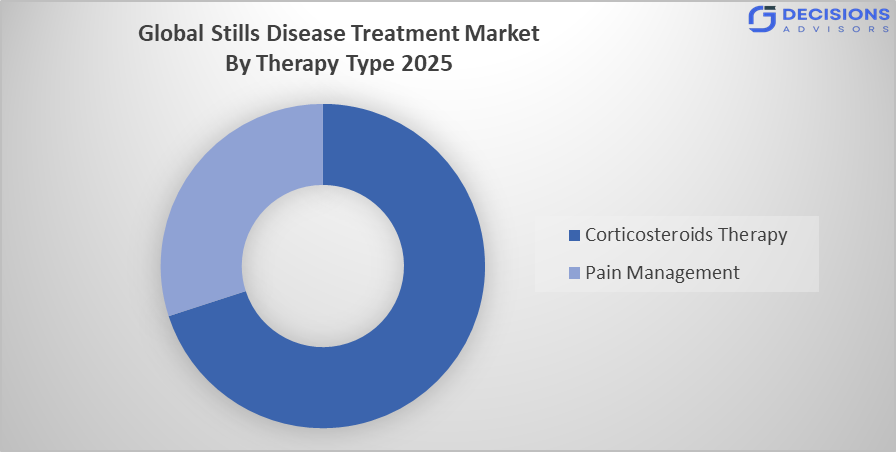
- The corticosteroids therapy segment dominated the market in 2025, approximately 45%, and is projected to grow at a substantial CAGR during the forecast period.
- The hospitals segment dominated the market in 2025, approximately 53.5%, and is projected to grow at a substantial CAGR during the forecast period.
- The compound annual growth rate of the Global Stills Disease Treatment Market is 4.56%.
- The market is likely to achieve a valuation of USD 3108.56 Million by 2035.
What is role of technology in grooming the market?
Technology plays a crucial role in grooming the global Still’s disease treatment market by improving diagnosis, treatment precision, and drug development. Advanced diagnostic tools, including biomarker testing and AI-based imaging analysis, enable earlier and more accurate detection of the disease, reducing delays in treatment initiation. In therapeutics, biotechnology advancements have led to the development of targeted biologics such as IL-1 and IL-6 inhibitors, which provide more effective and personalized treatment outcomes. Artificial intelligence and machine learning are increasingly used in drug discovery, accelerating the identification of new therapeutic targets. Additionally, digital health platforms and telemedicine enhance patient monitoring and disease management, especially in remote areas. Real-world data analytics also support clinical decision-making and post-treatment evaluation. Collectively, these technological innovations are improving treatment efficiency, reducing disease burden, and driving sustained growth in the global Still’s disease treatment market.
Market Drivers
The global Still’s disease treatment market is driven by increasing prevalence of rare autoinflammatory disorders and rising awareness among healthcare professionals leading to improved diagnosis rates. Advancements in biologic therapies, particularly IL-1 and IL-6 inhibitors, have significantly improved patient outcomes, boosting treatment adoption. Growing investment in orphan drug development and supportive regulatory frameworks, such as fast-track approvals and incentives, are encouraging pharmaceutical innovation. Additionally, expanding healthcare infrastructure and better access to rheumatology care in emerging economies are supporting market growth. Rising healthcare expenditure and increasing availability of targeted therapies further enhance treatment accessibility, collectively driving steady expansion of the global market.
Restrain
The global Stills Disease Treatment Market faces restraints due the high cost of biologic therapies, which limits patient access, especially in developing regions. Limited awareness and delayed diagnosis further restrict treatment uptake. Additionally, strict regulatory approvals and potential side effects of long-term immunosuppressive therapy also hinder market growth.
Competitive Analysis:
The report offers the appropriate analysis of the key organizations/companies involved within the global Stills Disease Treatment Market, along with a comparative evaluation primarily based on their product offerings, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborative analysis focusing on the current news and developments of the companies, which includes product development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
Top Companies in Global Stills Disease Treatment Market
- Novartis AG
- F. Hoffmann-La Roche Ltd
- Swedish Orphan Biovitrum AB (Sobi)
- Sanofi S.A.
- Regeneron Pharmaceuticals Inc.
- AbbVie Inc.
- Johnson & Johnson
- Pfizer Inc.
- Amgen Inc.
- Eli Lilly and Company
Government Initiatives
|
Country |
Key Government Initiatives |
|
US |
The U.S. Food and Drug Administration (FDA) supports rare disease treatment through the Orphan Drug Act (1983), offering fast-track approvals, tax incentives, and market exclusivity. These policies encourage development of biologics such as IL-1 and IL-6 inhibitors used in Still’s disease, improving innovation and patient access. |
|
India |
The Ministry of Health and Family Welfare implemented the National Policy for Rare Diseases (2021), providing financial support, treatment centres of excellence, and crowdfunding platforms for high-cost therapies. It improves access to biologics for rare diseases including Still’s disease. |
|
China |
The National Medical Products Administration (NMPA) has introduced rare disease catalogues, fast-track approvals, and reimbursement inclusion policies, enabling faster access to orphan drugs and expanding treatment availability for rare inflammatory conditions. |
Study on the Supply, Demand, Distribution, and Market Environment of the Stills Disease Treatment Market
The global Still’s disease treatment market shows a specialized supply–demand structure driven by rare disease prevalence and high-value biologic therapies. On the supply side, pharmaceutical companies focus on biologics such as IL-1 and IL-6 inhibitors, with limited but high-quality manufacturers due to complex production and regulatory requirements. Key suppliers operate mainly in developed regions with strong R&D capabilities. On the demand side, rising diagnosis rates, improved awareness of autoinflammatory diseases, and better access to rheumatology care are increasing patient identification globally. Demand is strongest in developed healthcare systems but is gradually expanding in emerging markets. The distribution system relies heavily on hospital pharmacies, specialty clinics, and controlled supply chains due to cold-chain requirements of biologics. The market environment is shaped by orphan drug incentives, regulatory approvals, and high treatment costs, creating both opportunities for innovation and challenges in affordability and access across different regions.
Price Analysis and Consumer Behaviour Analysis
The global Still’s disease treatment market is characterized by high-cost biologic therapies, making pricing a major constraint for patients and healthcare systems. Advanced drugs such as IL-1 and IL-6 inhibitors (e.g., anakinra, canakinumab, tocilizumab) often have annual costs reaching thousands of dollars, limiting accessibility, especially in low- and middle-income regions. This price sensitivity influences consumer behavior, where patients and providers often prefer corticosteroids or older immunosuppressants initially due to affordability. However, increasing awareness of better outcomes with biologics is gradually shifting demand toward premium therapies despite higher costs. Consumer behavior is also driven by insurance coverage, reimbursement policies, and hospital procurement decisions, rather than direct patient purchase. In developed regions like North America and Europe, stronger reimbursement systems support biologic adoption, while in Asia-Pacific, cost remains a key barrier, encouraging gradual uptake through biosimilars and government support programs.
Market Segmentation
The Stills Disease Treatment Market share is classified into drugs, therapy type, and end user.
- The anakinra segment dominated the market in 2025, approximately 43.1%, and is projected to grow at a substantial CAGR during the forecast period.
Based on the drugs, the Stills Disease Treatment Market is divided into anakinra, canakinumab, and tocilizumab. Among these, the anakinra segment dominated the market in 2025, with approximately 43.1%, and is projected to grow at a substantial CAGR during the forecast period. The growth of the anakinra segment is driven by its strong clinical effectiveness as an IL-1 receptor antagonist. It is widely used for both adult and pediatric Still’s disease because of its rapid action in reducing inflammation and fever. Its first-line positioning in many treatment guidelines and increasing physician preference in severe cases further strengthen its dominance in the biologics-driven market.
- The corticosteroids therapy segment dominated the market in 2025, approximately 45%, and is projected to grow at a substantial CAGR during the forecast period.
Based on the therapy type, the Stills Disease Treatment Market is divided into corticosteroids therapy and pain management. Among these, the corticosteroids therapy segment dominated the market in 2025, approximately 45%, and is projected to grow at a substantial CAGR during the forecast period. The corticosteroids therapy segment dominated due to they are commonly used as first-line treatment to quickly control inflammation and acute disease flares. Their affordability, wide availability, and immediate symptom relief make them the most frequently prescribed option, especially before transitioning patients to biologic therapies in long-term care.
- The hospitals segment dominated the market in 2025, approximately 53.5%, and is projected to grow at a substantial CAGR during the forecast period.
Based on the end user, the Stills Disease Treatment Market is divided into hospitals, homecare, and specialty clinics. Among these, the hospitals segment dominated the market in 2025, approximately 53.5%, and is projected to grow at a substantial CAGR during the forecast period. The growth of the hospitals segment is due to they provide specialized care, infusion facilities, and close monitoring required for biologic therapies. Severe cases of Still’s disease are typically managed in hospitals due to the need for multidisciplinary care, advanced diagnostics, and management of complications like systemic inflammation.
Strategies to Implement for Growth of the Market in Non-Leading Regions
To accelerate growth of the global Still’s disease treatment market in non-leading regions, several strategic initiatives are essential. Expanding early diagnosis programs and improving physician awareness can help reduce underdiagnosis of this rare inflammatory disorder. Strengthening healthcare infrastructure in emerging economies such as parts of Asia, Latin America, and the Middle East will improve access to advanced biologic therapies like IL-1 and IL-6 inhibitors. Partnerships between global pharmaceutical companies and local healthcare providers can enhance drug availability and distribution networks. Increasing government support through orphan drug incentives, subsidies, and reimbursement policies will improve treatment affordability. Additionally, clinical trial expansion in underrepresented regions will support data generation for diverse populations. Digital health tools and telemedicine can further improve disease monitoring and specialist access, collectively driving market penetration beyond leading regions.
Regional Segment Analysis of the Stills Disease Treatment Market
- North America (U.S., Canada, Mexico)
- Europe (Germany, France, U.K., Italy, Spain, Rest of Europe)
- Asia-Pacific (China, Japan, India, Rest of APAC)
- South America (Brazil and the Rest of South America)
- The Middle East and Africa (UAE, South Africa, Rest of MEA)
North America is anticipated to hold the largest share of approximately 37% the Stills Disease Treatment Market over the forecast period.
North America is anticipated to hold the largest share of approximately 37% the Stills Disease Treatment Market over the forecast period. The dominance of this region is driven by high adoption of advanced biologic therapies such as IL-1 and IL-6 inhibitors, strong healthcare infrastructure, and significant R&D investments. The United States leads the region due to early diagnosis rates, availability of approved treatments, and favorable reimbursement policies for rare diseases.
Asia Pacific is expected to grow at a rapid CAGR of approximately 11.5% in the Stills Disease Treatment Market during the forecast period.
Asia Pacific is expected to grow at a rapid CAGR of approximately 11.5% in the Stills Disease Treatment Market during the forecast period. This growth is fueled by increasing awareness of rare diseases, improving healthcare infrastructure, and rising access to biologic therapies. Expanding patient population and government support for orphan drugs further contribute to regional market expansion.
Europe is the 2nd largest region to grow in the Stills Disease Treatment Market during the period.
Europe is the second-largest region in the Still’s Disease Treatment Market during the forecast period. Countries such as Germany, United Kingdom, and France drive growth due to strong healthcare systems, increasing use of targeted biologics, and supportive reimbursement frameworks for rare disease treatments.
Future Market Trends in Global Stills Disease Treatment Market: -
1. Rising Adoption of Targeted Biologic Therapies
A key future trend is the growing use of advanced biologics such as IL-1, IL-6, and emerging IL-18 inhibitors. These therapies directly target inflammatory pathways, offering faster and more effective disease control. Their increasing clinical success, especially in refractory cases, is driving demand and reshaping treatment standards in the Still’s disease market.
2. Shift Toward Personalized and Biomarker-Driven Treatment
The market is moving toward precision medicine, where therapies are tailored using biomarkers like ferritin and cytokine levels. This approach improves treatment outcomes, reduces adverse effects, and supports early intervention strategies. Advances in genetic testing and AI-based diagnostics are further accelerating personalized care adoption globally.
3. Expansion of Biosimilars and Global Healthcare Access
Increasing development of biosimilars and improved healthcare infrastructure in emerging regions are enhancing treatment affordability and accessibility. This trend is expected to expand patient reach and boost market penetration. Additionally, supportive reimbursement policies and orphan drug incentives are encouraging innovation and wider adoption of advanced therapies.
Recent Development
- In September 2024, updated EULAR/PReS recommendations highlighted expanded clinical research on anakinra for Still’s disease, including pediatric populations. The collaboration emphasized early IL-1 inhibition to improve outcomes in children. These developments strengthened treatment approaches and supported growth in the global Still’s disease treatment market.
- In June 2024, Regeneron and Sanofi received FDA approval for sarilumab in polyarticular juvenile idiopathic arthritis and continued pivotal studies in systemic JIA, strengthening pipeline activity. Meanwhile, AB2 Bio had progressed tadekinig alfa in clinical development for inflammatory diseases, supporting ongoing innovation in the Still’s disease treatment market.
How is Recent Developments Helping the Market?
Recent developments are significantly accelerating growth in the global Still’s disease treatment market by improving treatment effectiveness, accessibility, and diagnosis rates. Advances in biologic therapies, particularly IL-1 inhibitors such as anakinra and canakinumab, have enhanced disease control and are widely adopted due to rapid action and strong clinical outcomes. Additionally, ongoing clinical research and pediatric studies are expanding indications and strengthening evidence for early intervention strategies, improving long-term patient outcomes. Regulatory progress, including new drug approvals and orphan designations, is encouraging innovation and investment in rare disease therapies. At the same time, favorable safety data and post-marketing studies are increasing physician confidence in biologics. Furthermore, rising healthcare spending, improved diagnostic capabilities, and expanding access to specialty care are enabling broader adoption of advanced therapies, collectively driving sustained market growth.
Market Segment
This study forecasts revenue at global, regional, and country levels from 2020 to 2035. Decision Advisors has segmented the Stills Disease Treatment Market based on the below-mentioned segments
Global Stills Disease Treatment Market, By Drugs
- Anakinra
- Canakinumab
- Tocilizumab
Global Stills Disease Treatment Market, By Therapy Type
- Corticosteroids Therapy
- Pain Management
Global Stills Disease Treatment Market, By End User
- Hospitals
- Homecare
- Specialty Clinics
Global Stills Disease Treatment Market, By Regional Analysis
- North America
- US
- Canada
- Mexico
- Europe
- Germany
- UK
- France
- Italy
- Spain
- Russia
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- Australia
- Rest of Asia Pacific
- South America
- Brazil
- Argentina
- Rest of South America
- Middle East & Africa
- UAE
- Saudi Arabia
- Qatar
- South Africa
- Rest of the Middle East & Africa
Frequently Asked Questions (FAQ)
Q. Why is Still’s disease considered a difficult condition to treat compared to other autoimmune diseases?
A. Still’s disease is challenging because it has unpredictable flare-ups, overlaps symptoms with infections and other inflammatory disorders, and lacks a single definitive diagnostic test. This often leads to delayed diagnosis, requiring a combination of clinical evaluation, biomarkers, and exclusion methods, which complicates early treatment initiation.
Q. How are biosimilars expected to influence the future of Still’s disease treatment?
A. Biosimilars are expected to significantly reduce treatment costs of biologic therapies like IL-1 and IL-6 inhibitors. This will improve affordability and expand access in cost-sensitive regions such as Asia-Pacific and Latin America, potentially increasing overall treatment adoption and reducing dependency on expensive originator biologics.
Q. What role do patient registries play in improving Still’s disease management?
A. Patient registries help collect long-term real-world data on disease progression, treatment response, and safety outcomes. This information supports clinical research, improves treatment guidelines, and helps pharmaceutical companies design more effective and targeted therapies for rare inflammatory conditions.
Q. How is pediatric Still’s disease influencing drug development strategies?
A. Pediatric cases, especially systemic juvenile idiopathic arthritis (sJIA), are pushing companies to develop safer, lower-dose biologics with improved safety profiles. This has increased clinical trials focused on early intervention therapies and has encouraged regulatory agencies to provide faster approvals for pediatric indications.
Check Licence
Choose the plan that fits you best: Single User, Multi-User, or Enterprise solutions tailored for your needs.
We Have You Covered
- 24/7 Analyst Support
- Clients Across the Globe
- Tailored Insights
- Technology Tracking
- Competitive Intelligence
- Custom Research
- Syndicated Market Studies
- Market Overview
- Market Segmentation
- Growth Drivers
- Market Opportunities
- Regulatory Insights
- Innovation & Sustainability
Report Details
| Scope | Global |
| Pages | 240 |
| Delivery | PDF & Excel via Email |
| Language | English |
| Release | May 2026 |
| Access | Download from this page |
