Global Thrombocytopenia Therapeutics Market
Global Thrombocytopenia Therapeutics Market Size, Share, By Drug Class (Glucocorticoids, Lysosomal Enzyme, Platelet Stimulating Agent), By Indication (Immune Thrombocytopenia (ITP), Chemotherapy-Induced Thrombocytopenia, Chronic Liver Disease-Related Thrombocytopenia), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), and By Region (North America, Europe, Asia-Pacific, Latin America, Middle East & Africa), Analysis and Forecast 2026?2035
Report Overview
Table of Contents
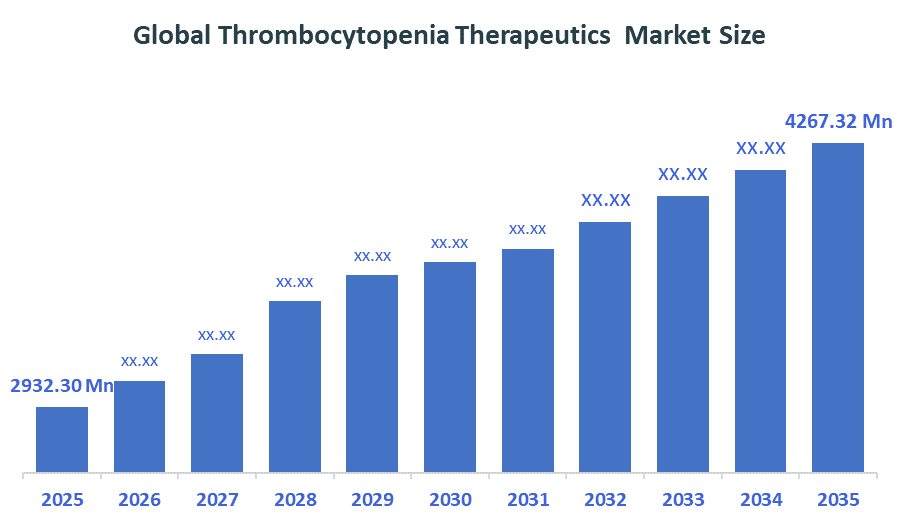
The Global Thrombocytopenia Therapeutics Market size is forecast to grow from USD 2932.30 Million in 2025 to approximately USD 4267.32 Million by 2035. According to Decision Advisors, a detailed research report on the thrombocytopenia therapeutics market indicates that the shift toward TPO-RAs & biologics trend dominates the market, accounting for approximately 30-35% of the total share globally. Novartis AG leads the market with an estimated 20-25% global market share. The company reported a 2025 revenue of approximately USD 45-47 Billion, making it one of the most influential forces shaping industry trends and overall market growth.
Market Snapshot
- Global Thrombocytopenia Therapeutics Market Size (2025): USD 2932.30Million
- Projected Global Thrombocytopenia Therapeutics Market Size (2035): USD 4267.32Million
- Global Thrombocytopenia Therapeutics Market Compound Annual Growth Rate (CAGR): 3.82%
- Largest Regional Market: North America
- Fastest Growing Region: Asia-Pacific
- Market 3rd Largest Region: Europe
- Base Year: 2025
- Historical Period: 2021–2024
- Forecast Period: 2026–2035
Market Overview/ Introduction
The global thrombocytopenia therapeutics market refers to the industry focused on the development, production, and commercialization of drugs and biologics used to treat low platelet count disorders. The thrombocytopenia therapeutics include drugs that use corticosteroids, immunoglobulins, thrombopoietin receptor agonists, and monoclonal antibodies to enhance platelet production, reducing platelet loss. The market will experience growth because of the increasing occurrence of immune thrombocytopenia, together with cancer-related complications and drug-induced platelet disorders. The upcoming opportunities in the medical field will emerge through the development of gene therapies, personalized medicine, and next-generation oral biologics that provide safer long-term management solutions. Market expansion occurs because research and development investments increase while developing countries enhance their healthcare systems through better targeted treatment options.
- The European Medicines Agency (EMA) holds stakeholder workshops to address challenges in thrombocytopenia drug development. In Canada, the government allocated $1.5 billion for a National Strategy for Drugs for Rare Diseases to secure capital for innovative hematology treatments.
- The Pharmacoeconomic (PE) Waiver system allows essential orphan drugs to be listed at premium prices based on their costs in other "A7" reference countries (e.g., USA, Japan). This helps expedite the entry of innovative rare disease treatments, such as those introduced through the Handok-Sobi joint venture.
Notable Insights: -
- North America is anticipated to hold the largest share of approximately 45% in the thrombocytopenia therapeutics market over the forecast period.
- Asia Pacific is expected to grow at a rapid CAGR of approximately 7.7 % in the thrombocytopenia therapeutics market during the forecast period.
- The platelet stimulating agent segment dominated the market in 2025, approximately 48%, and is projected to grow at a substantial CAGR during the forecast period.
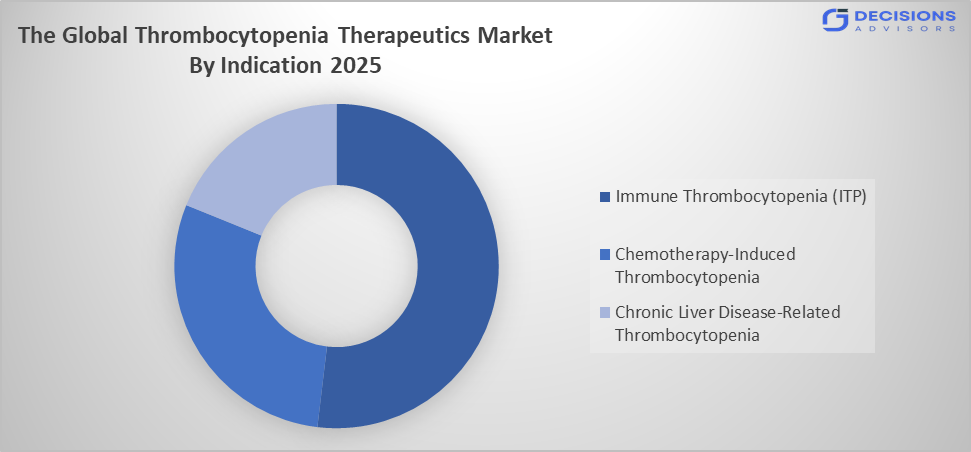
- The immune thrombocytopenia (ITP) segment dominated the market in 2025, approximately 55%, and is projected to grow at a substantial CAGR during the forecast period.
- The hospital pharmacies segment dominated the market in 2025, approximately 52%, and is projected to grow at a substantial CAGR during the forecast period.
- The compound annual growth rate of the Global Thrombocytopenia Therapeutics Market is 3.82%.
- The market is likely to achieve a valuation of USD 4267.32Million by 2035.
What is role of technology in grooming the market?
Technology plays a crucial role in shaping and advancing the global thrombocytopenia therapeutics market by improving diagnosis, treatment precision, and drug development. Advanced diagnostic technologies, including AI-based hematology analyzers and digital pathology, enable earlier and more accurate detection of platelet disorders. In drug development, bioinformatics, genomics, and high-throughput screening accelerate the discovery of targeted therapies and thrombopoietin receptor agonists. Artificial intelligence and machine learning support personalized medicine by predicting patient response and optimizing treatment plans. Additionally, digital health platforms and telemedicine improve patient monitoring and adherence to long-term therapies. Biotechnological advancements such as monoclonal antibody engineering and gene therapy research are driving next-generation treatment solutions. Overall, technology enhances efficiency, reduces development timelines, improves clinical outcomes, and expands access to innovative therapies, thereby significantly fueling market growth and transformation globally.
Market Drivers
The global market for thrombocytopenia treatments grows because of increasing cases of immune thrombocytopenia, cancer-related thrombocytopenia, and drug-induced platelet disorders. The usage of advanced biologics together with thrombopoietin receptor agonists has increased among physicians, resulting in improved patient results and higher treatment demand. The present medical knowledge of doctors and patients enables them to detect health problems earlier and determine suitable treatments. Market expansion receives support from both the healthcare system development in developing nations and the increase in healthcare spending. The process of continuous research and development, financial support, together with new drug approvals and the creation of oral and targeted therapies, drives innovative progress that extends worldwide treatment options.
Restrain
The global thrombocytopenia therapeutics market faces restraints due to the high cost of advanced biologics and thrombopoietin receptor agonists, limiting accessibility in low-income regions. Additionally, adverse effects such as thrombosis risk, strict regulatory approvals, and limited awareness in developing countries hinder widespread adoption and slow overall market expansion.
Competitive Analysis:
The report offers the appropriate analysis of the key organizations/companies involved within the global Thrombocytopenia therapeutics market, along with a comparative evaluation primarily based on their product offerings, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborative analysis focusing on the current news and developments of the companies, which includes product development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
Top Companies in Global Thrombocytopenia Therapeutics Market
- Amgen Inc.
- Novartis AG
- Pfizer Inc.
- Bristol Myers Squibb
- Roche Holding AG (F. Hoffmann-La Roche)
- Sanofi
- Takeda Pharmaceutical Company Limited
- Grifols S.A.
- Merck & Co., Inc.
- AbbVie Inc.
- GlaxoSmithKline (GSK plc)
- Rigel Pharmaceuticals, Inc.
Government Initiatives
|
Country |
Key Government Initiatives |
|
USA |
The FDA facilitates the thrombocytopenia market through expedited programs like Priority Review and Orphan Drug Designation, which were recently granted to treatments like rilzabrutinib (Wayrilz) for chronic ITP. Additionally, the FDA's MedWatch program maintains rigorous safety monitoring for approved TPO-R agonists to manage risks like thrombotic complications. |
|
India |
The CDSCO actively regulates the market by requiring local Phase III clinical trials for new thrombocytopenia drugs to ensure efficacy in the Indian population. Government initiatives like the PRIP Scheme (Promotion of Research and Innovation in Pharma MedTech) provide up to ?5000 crores in funding to transform India into a global R&D powerhouse, supporting the development of affordable treatments like romiplostim. |
|
China |
The NMPA utilizes Priority Review and Breakthrough Therapy Designation to accelerate the entry of innovative therapies such as sovleplenib and avatrombopag. China's regulatory system also emphasizes the approval of rare disease drugs, approving over 35 such treatments in 2025 to address critical unmet medical needs in the domestic market. |
Study on the Supply, Demand, Distribution, and Market Environment of the Thrombocytopenia Therapeutics Market
The global thrombocytopenia therapeutics market is characterized by a moderately consolidated supply structure, dominated by major pharmaceutical companies such as Amgen, Novartis, Pfizer, and Bristol Myers Squibb, which collectively drive innovation in biologics and thrombopoietin receptor agonists. Supply is strengthened by continuous R&D investments and expanding pipelines of targeted therapies, while biosimilars are gradually diversifying manufacturer participation and improving production capacity. On the demand side, rising prevalence of immune thrombocytopenia, cancer-related thrombocytopenia, and drug-induced cases is increasing patient intake globally. Demand is particularly strong in North America and Europe due to higher diagnosis rates, while Asia-Pacific shows the fastest growth because of improving healthcare access and awareness. The distribution system is primarily hospital-driven, as acute cases require immediate intervention with biologics and injectable therapies. Hospital pharmacies hold the largest share, followed by retail pharmacies for maintenance therapies and a growing online pharmacy segment supporting chronic care patients. The market environment is shaped by strong regulatory oversight, high treatment costs, rapid innovation in biologics, and increasing adoption of personalized medicine. Overall, the ecosystem reflects a high-innovation, value-based healthcare environment with expanding global access and competitive pharmaceutical positioning.
Price Analysis and Consumer Behaviour Analysis
The price structure in the global thrombocytopenia therapeutics market is relatively high due to the dominance of biologics, thrombopoietin receptor agonists, and branded specialty drugs. Advanced therapies such as monoclonal antibodies and novel oral agents contribute significantly to treatment costs, limiting affordability in low- and middle-income regions. However, increasing entry of biosimilars and generic versions is gradually putting downward pressure on prices, improving accessibility and widening market penetration. Pharmaceutical companies are also adopting tiered pricing strategies across developed and emerging economies to balance revenue and access. From a consumer behaviour perspective, treatment decisions are highly influenced by physician recommendations, insurance coverage, and hospital formularies rather than direct patient choice. Patients increasingly prefer oral and long-term therapies that reduce hospital visits and improve quality of life. Growing awareness, earlier diagnosis, and rising demand for personalized medicine are also shaping purchasing patterns toward safer, more convenient, and targeted treatment options.
Market Segmentation
The Thrombocytopenia Therapeutics Market share is classified into drug class, indication, and distribution channel.
- The platelet stimulating agent segment dominated the market in 2025, approximately 48%, and is projected to grow at a substantial CAGR during the forecast period.
Based on the drug class, the thrombocytopenia therapeutics market is divided into glucocorticoids, lysosomal enzyme, and platelet stimulating agent. Among these, the platelet stimulating agent segment dominated the market in 2025, approximately 48%, and is projected to grow at a substantial CAGR during the forecast period. The growth of the platelet stimulating agent segment is growing due to their high efficacy in increasing platelet production and suitability for long-term management of chronic thrombocytopenia. Their targeted mechanism, better safety profile compared to steroids, and growing physician preference significantly drive their dominance in the market.
- The immune thrombocytopenia (ITP) segment dominated the market in 2025, approximately 55%, and is projected to grow at a substantial CAGR during the forecast period.
Based on the indication, the thrombocytopenia therapeutics market is divided into immune thrombocytopenia (ITP), chemotherapy-induced thrombocytopenia, and chronic liver disease-related thrombocytopenia. Among these, the immune thrombocytopenia (ITP) segment dominated the market in 2025, approximately 55%, and is projected to grow at a substantial CAGR during the forecast period. The immune thrombocytopenia (ITP) segment dominated due to its being the most common form of thrombocytopenia requiring therapeutic intervention. Rising prevalence, increased diagnosis rates, and availability of multiple treatment options, including biologics and TPO-RAs, contribute to its leading position and sustained demand globally.
- The hospital pharmacies drugs segment dominated the market in 2025, approximately 52%, and is projected to grow at a substantial CAGR during the forecast period.
Based on the distribution channel, the thrombocytopenia therapeutics market is divided into hospital pharmacies, retail pharmacies, and online pharmacies. Among these, the hospital pharmacies segment dominated the market in 2025, approximately 52%, and is projected to grow at a substantial CAGR during the forecast period. The growth of the hospital pharmacies segment is due to the need for supervised administration of advanced therapies such as biologics and injectables. Severe and acute cases are primarily treated in hospital settings, ensuring consistent demand. Additionally, strong hospital procurement systems and insurance coverage further reinforce their leading role.
Strategies to Implement for Growth of the Market in Non-Leading Regions
To drive growth of the global thrombocytopenia therapeutics market in non-leading regions, companies should focus on improving healthcare accessibility and affordability. Expanding local manufacturing and distribution networks can reduce treatment costs and strengthen supply chain efficiency. Strategic partnerships with regional hospitals, government health programs, and NGOs can enhance patient awareness and early diagnosis rates. Increasing participation in public healthcare tenders and offering patient assistance programs will also improve drug adoption in price-sensitive markets. Additionally, investing in physician education and training initiatives can support better treatment adherence and correct diagnosis practices. Leveraging telemedicine platforms and digital health tools will help reach underserved rural populations. Regulatory alignment with local authorities and faster approval pathways can further accelerate market entry. Overall, a combined strategy of affordability, awareness, and accessibility is essential for sustainable growth in emerging and non-leading regions.
Regional Segment Analysis of the Thrombocytopenia Therapeutics Market
- North America (U.S., Canada, Mexico)
- Europe (Germany, France, U.K., Italy, Spain, Rest of Europe)
- Asia-Pacific (China, Japan, India, Rest of APAC)
- South America (Brazil and the Rest of South America)
- The Middle East and Africa (UAE, South Africa, Rest of MEA)
North America is anticipated to hold the largest share of approximately 45% the thrombocytopenia therapeutics market over the forecast period.
North America is anticipated to hold the largest share of approximately 45% the thrombocytopenia therapeutics market over the forecast period. The dominance of this region is primarily driven by advanced healthcare systems, high awareness, and strong presence of key pharmaceutical companies. Favorable reimbursement policies, early diagnosis, and rapid adoption of innovative biologics and targeted therapies further contribute to the region’s market dominance.
Asia Pacific is expected to grow at a rapid CAGR of approximately 7.7% in the thrombocytopenia therapeutics market during the forecast period.
Asia Pacific is expected to grow at a rapid CAGR of approximately 7.7% in the thrombocytopenia therapeutics market during the forecast period. This growth is driven by improving healthcare infrastructure, rising patient population, increasing awareness, and growing investments in pharmaceutical research, along with expanding access to advanced thrombocytopenia treatments across emerging economies.
Europe is the 3rd largest region to grow in the thrombocytopenia therapeutics market during the period.
Europe is the third-largest region in the global thrombocytopenia therapeutics market, supported by well-established healthcare systems, increasing prevalence of immune-related platelet disorders, and strong regulatory frameworks. Growth in the region is driven by rising adoption of advanced biologics, expanding research initiatives, and improved access to innovative treatment options across major European countries.
Future Market Trends in Global Thrombocytopenia Therapeutics Market: -
1. Expansion of Targeted Biologics
Targeted biologics, especially monoclonal antibodies, are expected to dominate future thrombocytopenia treatment due to their high specificity and reduced systemic toxicity. These therapies improve platelet counts by modulating immune pathways more precisely than conventional steroids, leading to better long-term outcomes and fewer adverse effects, driving strong clinical adoption globally.
2. Growth of Oral Thrombopoietin Receptor Agonists (TPO-RAs)
Oral TPO-RAs are gaining momentum because they offer convenient administration compared to injectable therapies. Their ability to stimulate platelet production effectively in chronic immune thrombocytopenia improves patient compliance. Increasing preference for home-based treatment and long-term disease management is accelerating demand, especially in outpatient and ambulatory care settings worldwide.
3. Rise of Personalized and Gene-Based Therapies
Personalized medicine and gene-based approaches are emerging as transformative trends, focusing on patient-specific immune and genetic profiles. These therapies aim to provide long-term or potentially curative solutions rather than symptom control. Advancements in genomics and precision medicine technologies are fueling research investments, making individualized thrombocytopenia treatment increasingly viable.
Recent Development
- In March 2026, Gilead Sciences announced plans to acquire Ouro Medicines and advance its immunology pipeline, including the BCMA×CD3 bispecific T-cell engager Gamgertamig (OM336). The therapy, originally developed with links to Keymed Biosciences, had previously received U.S. FDA Orphan Drug Designation for immune thrombocytopenia (ITP) in December 2025. The collaboration discussions with Galapagos NV aimed to accelerate development of this novel immune-reset therapy.
- In December 2025, Novartis AG presented positive Phase 3 results from the VAYHIT2 trial for Ianalumab, demonstrating that the combination of ianalumab with eltrombopag significantly prolonged the time to treatment failure compared to eltrombopag alone in patients with immune thrombocytopenia (ITP). The findings, presented at the American Society of Hematology Annual Meeting 2025, highlighted improved disease control and longer maintenance of safe platelet levels.
- In July 2025, the U.S. Food and Drug Administration approved Avatrombopag (Doptelet) for the treatment of pediatric patients aged one year and older with persistent or chronic immune thrombocytopenia (ITP) who had an inadequate response to prior therapies. The approval was supported by results from the Phase 3b AVA-PED-301 trial, which demonstrated significant efficacy in improving platelet counts in children.
- In August 2025, the U.S. Food and Drug Administration approved Rilzabrutinib (Wayrilz), developed by Sanofi, for the treatment of adults with persistent or chronic immune thrombocytopenia (ITP) who had an insufficient response to prior therapies. The therapy became the first Bruton’s tyrosine kinase (BTK) inhibitor approved for ITP, offering a novel mechanism targeting immune-mediated platelet destruction.
How is Recent Developments Helping the Market?
Recent developments in the global thrombocytopenia therapeutics market are significantly accelerating growth by improving treatment efficacy, safety, and accessibility. Advancements in thrombopoietin receptor agonists, such as next-generation oral agents, have expanded long-term management options for immune thrombocytopenia. Increased clinical trial activity for biologics and monoclonal antibodies is enhancing targeted treatment approaches with fewer side effects. Regulatory agencies granting orphan drug designations and fast-track approvals are encouraging innovation and reducing time-to-market. Additionally, the integration of AI-driven diagnostics is enabling earlier detection and personalized therapy selection. Growing adoption of combination therapies and biosimilars is also improving affordability in emerging markets. Pharmaceutical companies are investing heavily in gene therapy research, aiming for potential curative solutions. Overall, these developments are expanding the therapeutic landscape and driving strong market expansion globally in the coming years.
Market Segment
This study forecasts revenue at global, regional, and country levels from 2020 to 2035. Decision Advisors has segmented the Global Thrombocytopenia Therapeutics Market based on the below-mentioned segments:
Global Thrombocytopenia Therapeutics Market, By Drug Class
- Glucocorticoids
- Lysosomal Enzyme
- Platelet Stimulating Agent
Global Thrombocytopenia Therapeutics Market, By Indication
- Immune Thrombocytopenia (ITP)
- Chemotherapy-Induced Thrombocytopenia
- Chronic Liver Disease-Related Thrombocytopenia
Global Thrombocytopenia Therapeutics Market, By Distribution Channel
- Hospital Pharmacies
- Retail Pharmacies
- Online Pharmacies
Global Thrombocytopenia Therapeutics Market, By Regional Analysis
- North America
- US
- Canada
- Mexico
- Europe
- Germany
- UK
- France
- Italy
- Spain
- Russia
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- Australia
- Rest of Asia Pacific
- South America
- Brazil
- Argentina
- Rest of South America
- Middle East & Africa
- UAE
- Saudi Arabia
- Qatar
- South Africa
- Rest of the Middle East & Africa
Frequently Asked Questions (FAQ)
Q What factors are influencing the adoption of biosimilars in the thrombocytopenia therapeutics market?
A. The adoption of biosimilars is increasing due to their cost-effectiveness compared to branded biologics, improving access in price-sensitive regions. Additionally, favorable regulatory pathways and growing physician confidence in biosimilar efficacy and safety are accelerating their market penetration globally.
Q. How does reimbursement policy impact the thrombocytopenia therapeutics market?
A. Reimbursement policies play a critical role by determining patient access to expensive treatments such as biologics and TPO-RAs. Favorable insurance coverage in developed regions boosts treatment adoption, while limited reimbursement in emerging markets restricts access and slows market growth.
Q. What role do clinical trials play in shaping the thrombocytopenia therapeutics market?
A. Clinical trials are essential for introducing innovative therapies and expanding treatment options. Ongoing studies in biologics, gene therapies, and combination treatments help improve efficacy, safety, and long-term outcomes, thereby driving market expansion and competitive differentiation.
Q. How are emerging markets contributing to the global thrombocytopenia therapeutics market growth?
A. Emerging markets are contributing through improving healthcare infrastructure, rising awareness, and increasing government healthcare spending. Expanding access to diagnostics and treatments, along with growing pharmaceutical investments, is creating new growth opportunities and expanding the patient treatment base.
Check Licence
Choose the plan that fits you best: Single User, Multi-User, or Enterprise solutions tailored for your needs.
We Have You Covered
- 24/7 Analyst Support
- Clients Across the Globe
- Tailored Insights
- Technology Tracking
- Competitive Intelligence
- Custom Research
- Syndicated Market Studies
- Market Overview
- Market Segmentation
- Growth Drivers
- Market Opportunities
- Regulatory Insights
- Innovation & Sustainability
Report Details
| Scope | Global |
| Pages | 210 |
| Delivery | PDF & Excel via Email |
| Language | English |
| Release | Apr 2026 |
| Access | Download from this page |
