United Kingdom Autoimmune Disease Therapeutics Market
United Kingdom Autoimmune Disease Therapeutics Market Size, Share, By Drug Class (Anti-inflammatory Drugs, Immunosuppressants, Nonsteroidal Anti-Inflammatory Drugs, Biologics), By Indication (Rheumatoid Arthritis, Multiple Sclerosis, Type 1 Diabetes, Inflammatory Bowel Disease), By End User (Hospitals, Specialty Clinics, Homecare Settings), Analysis and Forecast 2026 ? 2035.
Report Overview
Table of Contents
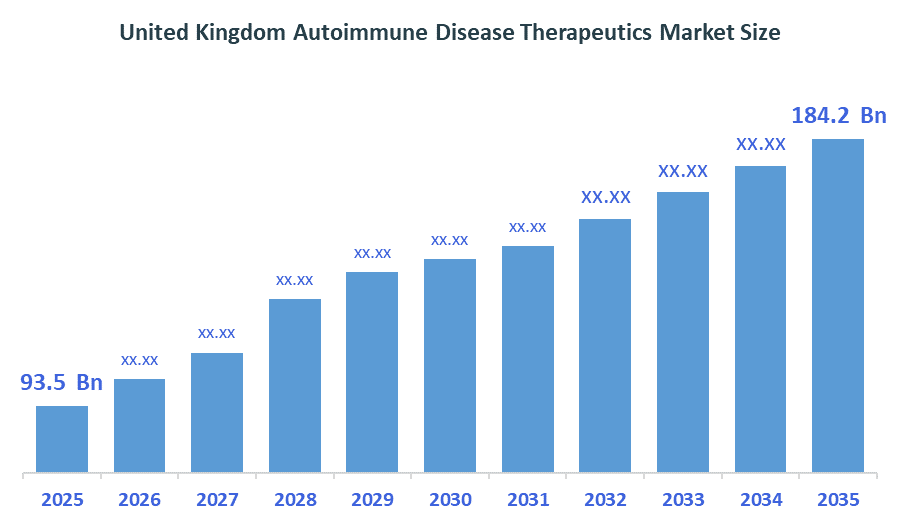
The United Kingdom Autoimmune Disease Therapeutics Market size is forecast to grow from approximately USD 93.5 Billion in 2025 to around USD 184.2Billion by 2035. According to Decision Advisors, a detailed research report on the autoimmune disease therapeutics market indicates that the rapid adoption of biologics trend dominates the market, accounting for approximately 35-40% of the total share in the United Kingdom. AbbVie Inc. leads the market with an estimated 21% United Kingdom market share. The company's annual revenue of approximately USD 54-56 billion is making it one of the most influential forces shaping industry trends and overall market growth.
Market Snapshot
- United Kingdom Autoimmune Disease Therapeutics Market size (2025): USD 93.5 Billion
- Projected United Kingdom Autoimmune Disease Therapeutics Market Size (2035): USD 184.2Billion
- United Kingdom Autoimmune Disease Therapeutics Market Compound Annual Growth Rate (CAGR): 7.02%
- Base Year: 2025
- Historical Period: 2021–2024
- Forecast Period: 2026–2035
Market Overview/Introduction
The market for autoimmune disease treatments in the United Kingdom includes drugs and biologics that help people with rheumatoid arthritis, multiple sclerosis, lupus, psoriasis, and inflammatory bowel disease. Immunosuppressants, corticosteroids, biologics, and targeted small molecules that control abnormal immune responses are some of the treatments that fall under this category. The market is growing because more people are getting sick, more people are becoming aware of the problems, and precision medicine is getting better. The introduction of biosimilars and next-generation biologics is making treatments easier to get and less expensive. Also, more research in immunology and personalized medicine is likely to lead to new treatment options. The United Kingdom's strong healthcare system and government support continue to make it easier for people to get treatment and manage their diseases over the long term.
- The United Kingdom government’s long-term healthcare strategy, led by National Health Service (NHS), emphasizes preventive care, digital transformation, and decentralized treatment models. It promotes early diagnosis, remote monitoring, and community-based services to manage chronic conditions like autoimmune diseases. The plan enhances patient access to advanced therapies, integrates data-driven decision-making, and encourages innovation in biologics and personalized treatment approaches.
- The NHS England Clinical Commissioning Policy for immunoglobulin therapies provides structured guidance for prescribing and prioritizing treatment in autoimmune and immune-related disorders. It classifies indications by clinical urgency, ensuring limited immunoglobulin supplies are allocated efficiently. The policy supports evidence-based decision-making, standardizes care pathways, and improves patient outcomes by prioritizing high-need cases.
Notable Insights: -
- The biologics segment dominated the market in 2025 and holds the largest market share, accounting for approximately 52% during the forecast period.
- The rheumatoid arthritis segment dominated the market in 2025 and holds the largest market share, accounting for approximately 38% during the forecast period.
- The hospitals segment dominated the market in 2025 and holds the largest market share, accounting for approximately 49% during the forecast period.
- The compound annual growth rate of the United Kingdom Autoimmune Disease Therapeutics Market is 7.02%.
- The market is likely to achieve a valuation of USD 184.2 Billion by 2035.
What is the role of technology in grooming the market?
Technology plays a crucial role in advancing the United Kingdom autoimmune disease therapeutics market by enhancing diagnosis, treatment precision, and patient outcomes. Advanced biologic drug development by companies like Pfizer Inc. and Eli Lilly and Company has improved treatment effectiveness and accessibility. Digital health platforms, including telemedicine and remote monitoring systems, enable continuous disease management and early intervention through NHS integration. Artificial intelligence and big data analytics are increasingly used to predict disease progression and personalize treatment strategies. Innovations in drug delivery, such as self-administered injectables and oral biologics, enhance patient convenience and adherence. Overall, technology is accelerating the shift toward precision medicine, improving cost efficiency, and strengthening patient-centric healthcare delivery.
Market Drivers
The United Kingdom autoimmune disease therapeutics market is growing due to the rising prevalence of autoimmune disorders, increasing demand for long-term disease management, and strong healthcare support from the NHS. Pharmaceutical companies like Johnson & Johnson and Bristol-Myers Squibb Company are advancing biologics and targeted therapies, improving treatment outcomes and accessibility. Growing awareness about early diagnosis, favorable reimbursement policies, and expanding use of biosimilars are further driving market growth. Additionally, ongoing research and innovation in immunotherapy and precision medicine are significantly contributing to the expansion of therapeutic options and overall market development.
Restrain
The United Kingdom autoimmune therapeutics market faces challenges due to high costs of biologics from companies like AbbVie Inc., strict pricing regulations under the NHS, and complex approval processes. Supply chain disruptions, biosimilar competition, and limited access to advanced therapies also restrict overall market growth.
Study on the Supply, Demand, Distribution, and Market Environment of the United Kingdom Autoimmune Disease Therapeutics Market
The United Kingdom autoimmune disease therapeutics market demonstrates a well-structured supply–demand ecosystem driven by rising prevalence of conditions such as Rheumatoid Arthritis and Multiple Sclerosis. Demand is increasing due to aging populations, improved diagnosis rates, and growing awareness, while supply is supported by strong participation from global pharmaceutical companies and robust R&D pipelines. Distribution channels are dominated by hospital pharmacies, specialty clinics, and retail pharmacies, ensuring widespread drug accessibility across urban and rural regions. The market environment is highly regulated, with oversight from bodies like the National Health Service and Medicines and Healthcare products Regulatory Agency, promoting safety, pricing control, and reimbursement frameworks, which collectively shape market stability and competitive dynamics.
Price Analysis and Consumer Behaviour Analysis
The United Kingdom autoimmune therapeutics market features a tiered pricing structure influenced by high-cost biologics, biosimilars, and conventional therapies. Premium biologics from companies like Johnson & Johnson dominate overall costs, while biosimilars are increasing price competition and affordability under NHS frameworks. Consumer behavior is largely guided by physician recommendations, clinical effectiveness, and reimbursement availability. Patients increasingly prefer advanced biologics for long-term disease control despite higher costs. However, NHS coverage reduces price sensitivity. Rising awareness of early diagnosis and treatment adherence is encouraging timely intervention, while demand for personalized therapies reflects a growing shift toward value-based healthcare.
Market Segmentation
The United Kingdom Autoimmune Disease Therapeutics Market Share is classified into drug class, indication, and end user
- The biologics segment dominated the market in 2025 and holds the largest market share, accounting for approximately 52% during the forecast period.
Based on the drug class, the autoimmune disease therapeutics market is divided into anti-inflammatory drugs, immunosuppressants, nonsteroidal anti-inflammatory drugs, and biologics. Among these, the biologics segment dominated the market in 2025 and holds the largest market share, accounting for approximately 52% during the forecast period. The biologics segment is driven by superior efficacy, targeted mechanisms, and growing adoption in chronic conditions like rheumatoid arthritis and inflammatory bowel disease. Increased physician preference, strong clinical outcomes, and expanding biosimilar availability further support the sustained growth of this segment.
- The rheumatoid arthritis segment dominated the market in 2025 and holds the largest market share, accounting for approximately 38% during the forecast period.
Based on the indication, the autoimmune disease therapeutics market is divided into rheumatoid arthritis, multiple sclerosis, type 1 diabetes, and inflammatory bowel disease. Among these, the rheumatoid arthritis segment dominated the market in 2025 and holds the largest market share, accounting for approximately 38% during the forecast period. The rheumatoid arthritis segment in the U.K. is attributed to its high prevalence in the United Kingdom and continuous demand for long-term treatment. Early diagnosis initiatives, availability of advanced biologics, and government-supported treatment frameworks have significantly improved patient access, making this the leading therapeutic indication segment.
- The hospitals segment dominated the market in 2025 and holds the largest market share, accounting for approximately 49% during the forecast period.
Based on the end user, the autoimmune disease therapeutics market is divided into Hospitals, specialty clinics, and homecare settings. Among these, the hospitals segment dominated the market in 2025 and holds the largest market share, accounting for approximately 49% during the forecast period. The hospitals segment in the U.K. is driven by serving as primary centers for diagnosis, biologic administration, and specialist care. Availability of advanced infrastructure, skilled healthcare professionals, and reimbursement support under public healthcare systems like the NHS drives higher patient reliance on hospital-based treatment.
Recent Development
- In February 2026, Boehringer Ingelheim and United Kingdom-based Sitryx Therapeutics entered into a strategic licensing agreement valued at over $500 million, focused on developing oral small-molecule inhibitors targeting immune cell metabolism for the treatment of autoimmune and inflammatory diseases. Under the agreement, Boehringer Ingelheim obtained exclusive global rights to Sitryx’s preclinical immunometabolic program, while Sitryx became eligible for upfront, milestone, and royalty payments. The collaboration strengthened Boehringer’s immunology pipeline and supported the development of next-generation oral precision therapies aimed at modulating disease-driving immune pathways in autoimmune disorders.
- In February 2026, ImmunityBio announced that ANKTIVA (nogapendekin alfa inbakicept) in collaboration with Accord Healthcare, was being commercialized across the United Kingdom and Europe for patients with non-muscle invasive bladder cancer (NMIBC). The partnership expanded access to immunotherapy in the United Kingdom through Accord Healthcare’s commercial network and supported broader European distribution strategies.
- In January 2026, the National Institute for Health and Care Excellence (NICE) approved obinutuzumab in combination with mycophenolate mofetil as a new treatment option for adults with active lupus nephritis in the United Kingdom. The decision was based on strong clinical evidence showing improved kidney outcomes and higher complete renal response rates compared with standard therapy.
Competitive Analysis
The report offers the appropriate analysis of the key organisations/companies involved within the United Kingdom autoimmune disease therapeutics market, along with a comparative evaluation primarily based on their product offering, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborative analysis focusing on the current news and developments of the companies, which includes product development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
Top Key Companies in the United Kingdom Autoimmune Disease Therapeutics Market
- Pfizer Inc.
- AbbVie Inc.
- Johnson & Johnson
- Novartis AG
- F. Hoffmann-La Roche Ltd
- Others
Key Target Audience
- Market Players
- Investors
- End-users
- Government Authorities
- Consulting and Research Firm
- Venture capitalists
- Value-Added Resellers (VARs)
Market Segment
This study forecasts revenue at the United Kingdom, regional, and country levels from 2020 to 2035. Spherical Insights has segmented the United Kingdom Autoimmune disease therapeutics market based on the below-mentioned segments
United Kingdom Autoimmune Disease Therapeutics Market, By Drug Class
- Anti-inflammatory Drugs
- Immunosuppressants
- Nonsteroidal Anti-Inflammatory Drugs
- Biologics
United Kingdom Autoimmune Disease Therapeutics Market, By Indication
- Rheumatoid Arthritis
- Multiple Sclerosis
- Type 1 Diabetes
- Inflammatory Bowel Disease
United Kingdom Autoimmune Disease Therapeutics Market, By End User
- Hospitals
- Specialty Clinics
- Homecare Settings
Frequently Asked Questions (FAQ)
Q. What is driving the shift toward oral therapies in the United Kingdom autoimmune disease therapeutics market?
A. The shift toward oral therapies is driven by patient preference for convenience, improved adherence, and reduced hospital dependency. Pharmaceutical companies are increasingly developing oral small-molecule inhibitors that target immune pathways, reducing reliance on injectable biologics. NHS cost-containment policies and biosimilar competition are also encouraging innovation in orally administered treatments for long-term autoimmune disease management.
Q. How does the United Kingdom regulatory environment impact autoimmune drug approvals?
A. The United Kingdom regulatory system, led by the MHRA and NICE, ensures that autoimmune therapies are approved based on strong clinical and cost-effectiveness evidence. NICE health technology assessments determine NHS reimbursement eligibility, often influencing market adoption speed. This system encourages manufacturers to demonstrate both therapeutic benefit and economic value before widespread patient access.
Q. Why are biosimilars gaining importance in autoimmune disease treatment in the United Kingdom?
A. Biosimilars are gaining importance due to their ability to significantly reduce treatment costs while maintaining similar efficacy to originator biologics. The United Kingdom healthcare system actively promotes biosimilar switching to improve budget efficiency. This has increased competition in the biologics space and improved patient access to advanced therapies for chronic autoimmune conditions.
Q. What role do research collaborations play in advancing autoimmune therapeutics in the United Kingdom?
A. Research collaborations between pharmaceutical companies, biotech firms, and academic institutions accelerate innovation in autoimmune disease treatments. These partnerships help develop next-generation biologics, gene-targeted therapies, and immunomodulators. The United Kingdom’s strong clinical research infrastructure and funding ecosystem support early-stage discovery and faster clinical translation of novel therapies.
Check Licence
Choose the plan that fits you best: Single User, Multi-User, or Enterprise solutions tailored for your needs.
We Have You Covered
- 24/7 Analyst Support
- Clients Across the Globe
- Tailored Insights
- Technology Tracking
- Competitive Intelligence
- Custom Research
- Syndicated Market Studies
- Market Overview
- Market Segmentation
- Growth Drivers
- Market Opportunities
- Regulatory Insights
- Innovation & Sustainability
Report Details
| Scope | Country |
| Pages | 210 |
| Delivery | PDF & Excel via Email |
| Language | English |
| Release | Apr 2026 |
| Access | Download from this page |
