United Kingdom High Potency Active Pharmaceutical Ingredients Market
United Kingdom High Potency Active Pharmaceutical Ingredients Market Size, Share, By Type (Synthetic HPAPI, Biotech HPAPI), By Therapeutic Application (Oncology, Capsules, Hormonal Disorders, Glaucoma, Cardiovascular Diseases, Neurological Disorders) and By End-User (Pharmaceutical Companies, Biotechnology Companies, Research & Academic Institutes), Analysis and Forecast 2025-2035
Report Overview
Table of Contents
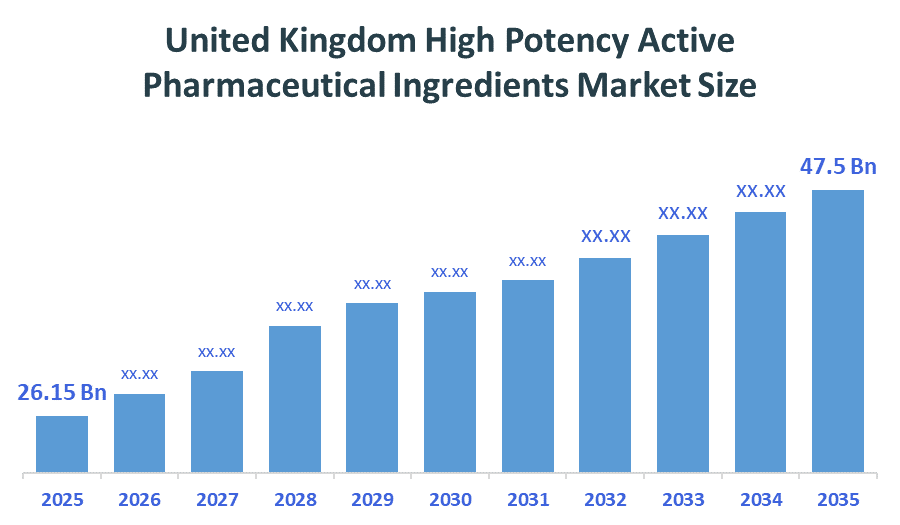
The United Kingdom anti-rheumatic market size is forecast to grow from approximately USD 26.15 Billion in 2025 to around USD 47.5 Billion by 2035. According to Decision Advisors, a detailed research report on the anti-rheumatics market indicates that targeted therapies trend dominates the market, accounting for approximately 55% of the total share in the United Kingdom. Pfizer Inc. leads the market with an estimated 16% United Kingdom market share. The company's annual revenue of approximately USD 58-60 billion is making it one of the most influential forces shaping industry trends and overall market growth.
Market Snapshot
- United Kingdom High Potency Active Pharmaceutical Ingredients Market Size (2025): USD 26.15Billion
- Projected United Kingdom High Potency Active Pharmaceutical Ingredients Market Size (2035): USD 47.5Billion
- United Kingdom High Potency Active Pharmaceutical Ingredients Market Compound Annual Growth Rate (CAGR): 6.15%
- Base Year: 2025
- Historical Period: 2021–2025
- Forecast Period: 2026–2035
Market Overview/Introduction
The United Kingdom High Potency Active Pharmaceutical Ingredients (HPAPI) market refers to the industry involved in the development, manufacturing, and commercialization of highly potent drug compounds used in targeted therapies. The handling of these substances requires special procedures because their deadly properties make them essential for use in oncology and chronic disease treatments. The handling of these substances requires special procedures because their deadly properties make them essential for use in oncology and chronic disease treatments. The United Kingdom High Potency Active Pharmaceutical Ingredients (HPAPI) market expands because of growing cancer and chronic disease rates, which create a demand for targeted therapies that deliver maximum treatment results. The market expansion receives support from two main factors, which are the increasing demand for precision medicine and the ongoing development of antibody-drug conjugates ADCs together with manufacturing containment solutions. The market keeps expanding because of two factors, which are the rising elderly population and the increasing investments in pharmaceutical research and development.
- The Life Sciences Innovative Manufacturing Fund provides up to £520 million to support advanced pharmaceutical manufacturing infrastructure. It promotes domestic production of complex APIs, including HPAPIs, by funding large-scale facilities, enhancing supply chain resilience, and strengthening the UK’s competitiveness in high-containment and oncology-focused drug manufacturing.
- The Medicines Manufacturing Labs of the Future Programme supports automation, AI, and robotics in pharmaceutical production with funding up to £7.5 million. It enhances efficiency, precision, and scalability in HPAPI manufacturing, particularly important for highly potent, low-dose drugs requiring advanced containment and digitalized process systems.
Notable Insights: -
- The synthetic HPAPI drugs segment dominated the market in 2025 and holds the largest market share, accounting for approximately 72.12% during the forecast period.
- The oncology segment dominated the market in 2025 and holds the largest market share, accounting for approximately 76.53% during the forecast period.
- The pharmaceutical companies segment dominated the market in 2025 and holds the largest market share, accounting for approximately 72.06% during the forecast period.
- The compound annual growth rate of the United Kingdom High Potency Active Pharmaceutical Ingredients is 6.15%.
- The market is likely to achieve a valuation of USD 47.5 billion by 2035.
What is the role of technology in grooming the market?
Technology plays a crucial role in shaping the growth of the United Kingdom high potency active pharmaceutical ingredients (HPAPI) market by enhancing safety, efficiency, and innovation. Advanced containment systems and automation technologies enable the safe handling of highly potent compounds, reducing occupational hazards and improving production accuracy. Continuous manufacturing and process analytical technologies (PAT) streamline production, ensuring consistent quality and regulatory compliance. Innovations in biotechnology, including antibody-drug conjugates (ADCs) and targeted therapies, are driving demand for HPAPIs. Additionally, digital tools such as artificial intelligence and data analytics support drug discovery, process optimization, and predictive maintenance. Overall, technological advancements accelerate product development timelines, reduce costs, and support the growing demand for precision medicines in oncology and other therapeutic areas.
Market Drivers
The United Kingdom high potency active pharmaceutical ingredients (HPAPI) market is driven by the growing incidence of cancer, which increases the need for highly effective and targeted therapies. Rising cases of autoimmune diseases, cardiovascular disorders, and other chronic conditions further boost demand for potent medications. The advancement of antibody-drug conjugates (ADCs) and other targeted treatments significantly relies on HPAPIs for precise action on diseased cells. Additionally, continuous innovation in containment systems and manufacturing technologies enhances the safe handling of highly toxic compounds. Moreover, the expanding aging population in the United Kingdom contributes to higher demand for advanced and long-term treatment options, supporting overall market growth.
Restrain
The United Kingdom high potency active pharmaceutical ingredients (HPAPI) market faces restraints due to high manufacturing and containment costs, stringent regulatory requirements, and complex handling procedures. Limited specialized facilities and skilled workforce shortages further hinder growth. Additionally, safety concerns related to potent compound exposure and lengthy approval timelines may slow market expansion.
Study on the Supply, Demand, Distribution, and Market Environment of the United Kingdom High Potency Active Pharmaceutical Ingredients
The United Kingdom High Potency Active Pharmaceutical Ingredients (HPAPI) market reflects a tightly regulated and innovation-driven ecosystem shaped by strong supply capabilities, rising demand, and evolving distribution networks. Supply is supported by advanced manufacturing infrastructure and government-backed initiatives such as the Life Sciences Innovative Manufacturing Fund, encouraging domestic production and reducing reliance on imports. Demand is primarily driven by the growing prevalence of cancer and the increasing adoption of targeted therapies, where HPAPIs play a critical role. Distribution channels are becoming more specialized, requiring high-containment logistics and compliance with stringent regulatory standards set by the Medicines and Healthcare products Regulatory Agency. The market environment is highly competitive, with pharmaceutical and biotechnology firms investing in R&D, sustainability, and advanced manufacturing technologies to maintain global competitiveness and ensure long-term supply chain resilience.
Price Analysis and Consumer Behaviour Analysis
Price dynamics in the United Kingdom High Potency Active Pharmaceutical Ingredients (HPAPI) market are strongly influenced by high production complexity, regulatory compliance costs, and limited manufacturing capacity. HPAPIs require specialized containment systems, skilled labor, and strict safety standards, significantly increasing production costs and resulting in premium pricing structures. However, pricing pressure exists due to growing generic competition and cost-sensitive healthcare systems, encouraging value-based pricing and outsourcing strategies. From a consumer behavior perspective, pharmaceutical companies prioritize quality, reliability, and regulatory compliance over cost, especially for oncology and targeted therapies. Demand is driven by innovation, clinical efficacy, and long-term supplier partnerships, with increasing preference for specialized contract manufacturers offering scalability, advanced technologies, and consistent high-potency product quality.
Market Segmentation
The United Kingdom High Potency Active Pharmaceutical Ingredients share is classified into type, therapeutic application, and end-user
- The synthetic HPAPI drugs segment dominated the market in 2025 and holds the largest market share, accounting for approximately 72.12% during the forecast period.
Based on the type, the excipients market is divided into synthetic HPAPI and biotech HPAPI. Among these, the synthetic HPAPI drugs segment dominated the market in 2025 and holds the largest market share, accounting for approximately 72.12% during the forecast period. The synthetic HPAPI segment is driven by its widespread use in oncology and chronic disease treatments. It offers cost-effective production, scalability, and well-established manufacturing processes. Additionally, increasing demand for targeted therapies and growing pharmaceutical R&D investments further support the segment’s strong growth in the United Kingdom market.
- The oncology segment dominated the market in 2025 and holds the largest market share, accounting for approximately 76.53% during the forecast period.
Based on the therapeutic application, the excipients market is divided into oncology, hormonal disorders, glaucoma, cardiovascular diseases, and neurological disorders. Among these, the oncology segment dominated the market in 2025 and holds the largest market share, accounting for approximately 76.53% during the forecast period. The oncology segment leads the market due to the rising prevalence of cancer and increasing demand for targeted therapies. High potency APIs are essential for oncology drugs, particularly in advanced treatments like antibody-drug conjugates, driving strong growth and continued investment in this segment.
- The pharmaceutical companies segment dominated the market in 2025 and holds the largest market share, accounting for approximately 72.06% during the forecast period.
Based on the end-user, the excipients market is divided into pharmaceutical companies, biotechnology companies, and research & academic institutes. Among these, the pharmaceutical companies segment dominated the market in 2025 and holds the largest market share, accounting for approximately 72.06% during the forecast period. The pharmaceutical companies segment dominates due to strong in-house manufacturing capabilities, high investment in drug development, and growing demand for targeted therapies. These companies focus on innovation, regulatory compliance, and large-scale production, driving consistent demand for HPAPIs and supporting significant market growth in the United Kingdom.
Recent Development
- In November 2025, the United Kingdom’s pharmaceutical manufacturing sector reported strong economic growth, with gross value added (GVA) reaching approximately £19 billion in 2023, according to data compiled by the Institute of Chartered Accountants in England and Wales based on Office for National Statistics figures.
- In September 2022, Lonza Group expanded its highly potent active pharmaceutical ingredients (HPAPI) multipurpose manufacturing suite at its Visp, Switzerland facility. The expansion added advanced capacity for antibody-drug conjugate (ADC) payload production, strengthening supply capabilities across Europe, including the United Kingdom, amid rising demand for oncology-focused high-potency therapies.
Competitive Analysis
The report offers the appropriate analysis of the key organisations/companies involved within the United Kingdom High Potency Active Pharmaceutical Ingredients Market, along with a comparative evaluation primarily based on their product offering, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborative analysis focusing on the current news and developments of the companies, which includes product development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
Top Key Companies in the United Kingdom High Potency Active Pharmaceutical Ingredients Market
- Pfizer Inc.
- Bristol-Myers Squibb Company
- Boehringer Ingelheim International GmbH
- CordenPharma
- CARBOGEN AMCIS AG
- Others
Key Target Audience
- Market Players
- Investors
- End-users
- Government Authorities
- Consulting and Research Firm
- Venture capitalists
- Value-Added Resellers (VARs)
Market Segment
This study forecasts revenue at the United Kingdom, regional, and country levels from 2020 to 2035. Decision Advisor has segmented the United Kingdom High Potency Active Pharmaceutical Ingredients Market based on the below-mentioned segments
United Kingdom High Potency Active Pharmaceutical Ingredients Market, By Type
- Synthetic HPAPI
- Biotech HPAPIs
United Kingdom High Potency Active Pharmaceutical Ingredients Market, By Therapeutic Application
- Oncology
- Capsules
- Hormonal Disorders
- Glaucoma
- Cardiovascular Diseases
- Neurological Disorders
United Kingdom High Potency Active Pharmaceutical Ingredients Market, By End-User
- Pharmaceutical Companies
- Biotechnology Companies
- Research & Academic Institutes
Frequently Asked Questions (FAQ)
Q. How does outsourcing impact the HPAPI market in the United Kingdom?
A. Outsourcing plays a significant role in the HPAPI market by allowing pharmaceutical companies to reduce capital expenditure and focus on core competencies such as drug discovery and marketing. Contract Development and Manufacturing Organizations (CDMOs) provide specialized containment facilities and expertise, enabling efficient and safe production of highly potent compounds. This trend is expected to grow as demand for complex and small-batch drugs increases.
Q. What role do regulatory frameworks play in shaping the HPAPI market?
A. Regulatory frameworks in the United Kingdom ensure strict compliance with safety, quality, and environmental standards when handling highly potent compounds. Agencies such as the FDA impose rigorous guidelines on manufacturing practices, containment, and worker safety. While these regulations ensure high-quality drug production, they also increase operational costs and create barriers for new entrants.
Q. How is the shift toward personalized medicine influencing HPAPI demand?
A. The growing adoption of personalized medicine is significantly boosting demand for HPAPIs, as these therapies often require highly targeted and potent compounds. Treatments tailored to individual patient profiles, especially in oncology and rare diseases, rely on precise drug formulations. This trend is encouraging innovation and increasing investment in specialized HPAPI production technologies.
Q. What challenges do supply chains face in the HPAPI market?
A. HPAPI supply chains face challenges such as dependency on specialized raw materials, limited manufacturing facilities, and geopolitical uncertainties. Disruptions can impact the timely availability of critical drug components. Additionally, stringent transportation and storage requirements for potent compounds add complexity, making supply chain resilience a key focus area for market players.
Check Licence
Choose the plan that fits you best: Single User, Multi-User, or Enterprise solutions tailored for your needs.
We Have You Covered
- 24/7 Analyst Support
- Clients Across the Globe
- Tailored Insights
- Technology Tracking
- Competitive Intelligence
- Custom Research
- Syndicated Market Studies
- Market Overview
- Market Segmentation
- Growth Drivers
- Market Opportunities
- Regulatory Insights
- Innovation & Sustainability
Report Details
| Scope | country |
| Pages | 240 |
| Delivery | PDF & Excel via Email |
| Language | English |
| Release | Apr 2026 |
| Access | Download from this page |
