United Kingdom Intravenous Ibuprofen Market
United Kingdom Intravenous Ibuprofen Market Size, Share, By Indication (Inflammatory Management, Fever Management), By Age Group (Pediatrics, Adults), By End User (Hospitals, Specialty Clinics, Ambulatory Surgical Centers), Analysis and Forecast 2026-2035
Report Overview
Table of Contents
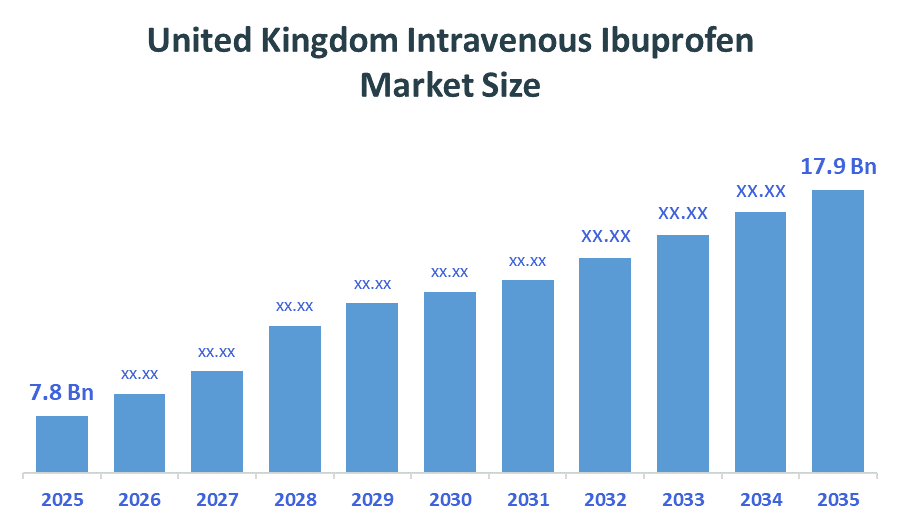
The United Kingdom Intravenous Ibuprofen Market size is forecast to grow from approximately USD 7.8 Billion in 2025 to around USD 17.9Billion by 2035. According to Decision Advisors, a detailed research report on the intravenous ibuprofen market indicates that the strong clinical efficacy, affordability of generics, and widespread physician preference trend dominate the market, accounting for approximately 85% of the total share in the United Kingdom. Pfizer Inc. leads the market with an estimated 54-57% United Kingdom market share. The company's annual revenue of approximately USD 3.3 billion is making it one of the most influential forces shaping industry trends and overall market growth.
Market Snapshot
- United Kingdom Intravenous Ibuprofen Market size (2025): USD 7.8 Billion
- Projected United Kingdom Intravenous Ibuprofen Market Size (2035): USD 17.9 Billion
- United Kingdom Intravenous Ibuprofen Market Compound Annual Growth Rate (CAGR): 8.66%
- Base Year: 2025
- Historical Period: 2021–2024
- Forecast Period: 2026–2035
Market Overview/Introduction
The United Kingdom Intravenous Ibuprofen Market refers to pharmaceutical industries dedicated to manufacturing, supply chain, and clinical practice of IV formulated ibuprofen for pain and fever control in hospital settings. Intravenous ibuprofen is an NSAID that is introduced directly into the bloodstream, therefore acting rapidly in a treatment setting. The market is reinforced through the National Health Service and is assisted by the organized system. The future opportunities for this market are more opioid sparing alternatives, ambulatory care services, and formulation development. The United Kingdom Intravenous Ibuprofen Market grows with increasing surgical procedures performed in hospitals, greater demands for fast-acting analgesics, and advances in hospital care management, favoring better, more effective, and safer patient treatment and better outcomes.
- The NHS England Medicines Procurement and Supply Chain framework ensures bulk purchasing and cost-effective sourcing of hospital medicines, including IV analgesics like ibuprofen. It standardises contracts across NHS trusts, improves supply continuity, and supports price negotiations with pharmaceutical suppliers to maintain affordability and availability in secondary care settings.
- The NHS Payment Scheme defines how hospitals are funded for treatments delivered, including inpatient pain management therapies where intravenous ibuprofen may be used. It reimburses healthcare providers based on activity and treatment complexity, encouraging efficient use of injectable analgesics in post-operative and acute pain care pathways.
Notable Insights: -
- The inflammatory management segment dominated the market in 2025 and holds the largest market share, accounting for approximately 65% during the forecast period.
- The adult segment dominated the market in 2025 and holds the largest market share, accounting for approximately 70% during the forecast period.
- The hospitals segment dominated the market in 2025 and holds the largest market share, accounting for approximately 75% during the forecast period.
- The compound annual growth rate of the United Kingdom Intravenous Ibuprofen Market is 8.66%.
- The market is likely to achieve a valuation of USD 17.9 Billion by 2035.
What is the role of technology in grooming the market?
Technology plays a significant role in shaping the United Kingdom intravenous ibuprofen market by enhancing drug delivery, safety, and overall treatment efficiency. Advanced sterile manufacturing technologies ensure high-quality, contamination-free formulations, improving reliability and regulatory compliance. Smart infusion systems and automated dosing devices in hospitals help deliver precise drug administration, reducing medication errors and improving patient outcomes. Digital healthcare integration within the National Health Service supports better inventory management, demand forecasting, and cost control. Additionally, electronic health records enable clinicians to optimize pain management protocols using real-time patient data. Innovations in packaging and storage technologies also extend product shelf life and ease of use. Overall, technological advancements are driving efficiency, safety, and broader adoption of intravenous ibuprofen in clinical settings.
Market Drivers
The United Kingdom Intravenous Ibuprofen Market is driven by the rising number of surgical procedures and increasing demand for effective post-operative pain management. The shift toward opioid-sparing therapies is significantly boosting adoption, as clinicians seek safer alternatives with fewer side effects. The growing incidence of acute conditions, trauma cases, and hospital admissions further supports demand. Additionally, standardized treatment protocols within the National Health Service encourage the use of clinically effective intravenous formulations. Faster onset of action, improved patient outcomes, and increasing awareness among healthcare professionals are also contributing to market growth across hospital and critical care settings.
Restrain
The United Kingdom intravenous ibuprofen market faces restraints due to the high cost compared to oral alternatives, limiting widespread adoption. Strict regulatory requirements and dependence on hospital-based administration further restrict usage. Additionally, the availability of alternative analgesics and budget constraints within the National Health Service create pricing pressure and slow market expansion.
Study on the Supply, Demand, Distribution, and Market Environment of the United Kingdom Intravenous Ibuprofen Market
The United Kingdom intravenous ibuprofen market operates within a structured healthcare ecosystem shaped by centralized procurement and clinical demand. Supply is largely dependent on a limited number of global manufacturers, with strict regulatory oversight ensuring product quality and continuity. Demand is driven by rising surgical procedures, trauma cases, and the need for effective non-opioid pain and fever management in acute care settings. Distribution is primarily controlled through hospital pharmacies under the National Health Service, which streamlines access and pricing through bulk purchasing agreements. The market environment is highly regulated, emphasizing cost-efficiency and clinical effectiveness. Additionally, increasing focus on opioid reduction strategies and enhanced recovery protocols is encouraging wider adoption of intravenous ibuprofen, supporting steady growth despite pricing pressures and supply chain constraints.
Price Analysis and Consumer Behaviour Analysis
The price analysis of the United Kingdom intravenous ibuprofen market reflects relatively high treatment costs compared to oral alternatives, driven by sterile manufacturing requirements, hospital-grade formulations, and regulatory compliance. Pricing is largely influenced by procurement contracts within the National Health Service, where bulk purchasing helps moderate costs. Consumer behaviour is primarily institutional rather than individual, with hospitals prioritizing cost-effectiveness, clinical efficacy, and opioid-sparing benefits. Demand is strongly linked to surgical volumes and acute care needs, while prescribers favor IV ibuprofen for faster onset of action and reduced gastrointestinal side effects. Additionally, there is an increasing preference for non-opioid analgesics, shaping purchasing decisions toward safer pain management solutions despite premium pricing.
Market Segmentation
The United Kingdom Intravenous Ibuprofen Market Share is classified into indication, age group, and end user.
- The inflammatory management segment dominated the market in 2025 and holds the largest market share, accounting for approximately 65% during the forecast period.
Based on the indication, the intravenous ibuprofen market is divided into inflammatory management and fever management. Among these, the inflammatory management segment dominated the market in 2025 and holds the largest market share, accounting for approximately 65% during the forecast period. The inflammatory management segment is driven by its widespread use in post-operative care, trauma, and acute inflammation cases. Hospitals prefer IV ibuprofen as an opioid-sparing alternative, supporting faster recovery and reducing complications associated with narcotic analgesics.
- The adult segment dominated the market in 2025 and holds the largest market share, accounting for approximately 70% during the forecast period.
Based on the age group, the intravenous ibuprofen market is divided into pediatrics and adults. Among these, the adult segment dominated the market in 2025 and holds the largest market share, accounting for approximately 70% during the forecast period. The adult segment in the U.K. is attributed to higher hospitalization and surgical procedure rates. The prevalence of chronic conditions, orthopedic surgeries, and post-operative pain management needs is significantly higher in adults compared to pediatrics, leading to greater reliance on intravenous ibuprofen in adult patient care settings.
- The hospitals segment dominated the market in 2025 and holds the largest market share, accounting for approximately 75% during the forecast period.
Based on the end user, the intravenous ibuprofen market is divided into Hospitals, specialty clinics, and Ambulatory Surgical Centers. Among these, the hospitals segment dominated the market in 2025 and holds the largest market share, accounting for approximately 75% during the forecast period. The hospitals segment in the U.K. is driven by the fact that intravenous ibuprofen is primarily administered in controlled clinical environments. Its use is concentrated in surgical units, emergency care, and intensive care settings where rapid pain and fever management is essential, making hospitals the primary point of administration and demand.
Recent Development
- In October 2025, Cumberland Pharmaceuticals and PiSA Farmaceutica expanded their global collaboration to strengthen access to intravenous ibuprofen formulations, including 800 mg vial presentations designed for hospital-based postoperative pain management. The partnership focused on improving the availability of injectable ibuprofen in international markets through coordinated regulatory and distribution efforts. PiSA planned to commercialize the product in Mexico.
- In March 2025, IOL Chemicals and Pharmaceuticals Limited played a key role in strengthening the global supply of high-quality generic ibuprofen APIs, supported by its ISO and GMP-certified manufacturing facilities. The company maintained strict compliance with international quality standards, enabling consistent production of pharmaceutical-grade ibuprofen used in pain management formulations, including injectable and hospital-based therapies.
- In January 2025, pharmaceutical research in the United Kingdom and Europe advanced the development of parenteral ready-to-use fixed-dose combinations (FDCs) incorporating NSAIDs such as ibuprofen for improved multimodal pain management. The research focused on optimizing injectable formulations to deliver faster and more effective postoperative and acute pain relief in hospital settings.
Competitive Analysis
The report offers the appropriate analysis of the key organisations/companies involved within the United Kingdom Intravenous Ibuprofen Market, along with a comparative evaluation primarily based on their product offering, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborative analysis focusing on the current news and developments of the companies, which includes product development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
Top Key Companies in the United Kingdom Intravenous Ibuprofen Market
- Cumberland Pharmaceuticals Inc.
- Pfizer Inc.
- Fresenius Kabi AG
- Hikma Pharmaceuticals PLC
- CSL Limited
- Others
Key Target Audience
- Market Players
- Investors
- End-users
- Government Authorities
- Consulting and Research Firm
- Venture capitalists
- Value-Added Resellers (VARs)
Market Segment
This study forecasts revenue at the United Kingdom, regional, and country levels from 2020 to 2035. Decision Advisor has segmented the United Kingdom Intravenous Ibuprofen Market based on the below-mentioned segments
United Kingdom Intravenous Ibuprofen Market, By Indication
- Inflammatory Management
- Fever Management
United Kingdom Intravenous Ibuprofen Market, By Age Group
- Pediatrics
- Injectable
United Kingdom Intravenous Ibuprofen Market, By End User
- Hospitals
- Specialty Clinics
- Ambulatory Surgical Centers
Frequently Asked Questions (FAQ)
Q Why is intravenous ibuprofen preferred over oral NSAIDs in UK hospitals?
A. Intravenous ibuprofen is preferred in UK hospitals because it provides rapid onset of action, predictable absorption, and effective pain control in patients who cannot take oral medications. It is especially useful in post-surgical and emergency settings where fast analgesia and better patient recovery outcomes are required.
Q. How does the UK regulatory environment influence IV ibuprofen usage?
A. The UK regulatory environment ensures strict approval, safety monitoring, and hospital-only administration of intravenous ibuprofen. Agencies like the MHRA regulate quality and pharmacovigilance, ensuring safe use in controlled clinical settings, which increases physician confidence but limits outpatient use.
Q. What are the key challenges in the supply chain of IV ibuprofen in the UK?
A. Key challenges include dependence on imported active pharmaceutical ingredients, limited number of manufacturers, and strict sterile production requirements. Any disruption in global supply chains or raw material shortages can impact hospital availability, making procurement planning within the NHS critical.
Q. How is the shift toward opioid reduction impacting the IV ibuprofen market in the UK?
A. The shift toward opioid-sparing pain management is significantly increasing the adoption of intravenous ibuprofen. Hospitals are integrating it into multimodal analgesia protocols to reduce opioid-related side effects, dependency risks, and improve recovery outcomes, thereby supporting market growth across surgical departments.
Check Licence
Choose the plan that fits you best: Single User, Multi-User, or Enterprise solutions tailored for your needs.
We Have You Covered
- 24/7 Analyst Support
- Clients Across the Globe
- Tailored Insights
- Technology Tracking
- Competitive Intelligence
- Custom Research
- Syndicated Market Studies
- Market Overview
- Market Segmentation
- Growth Drivers
- Market Opportunities
- Regulatory Insights
- Innovation & Sustainability
Report Details
| Scope | country |
| Pages | 240 |
| Delivery | PDF & Excel via Email |
| Language | English |
| Release | Apr 2026 |
| Access | Download from this page |
