United Kingdom Intravenous Solutions Market
United Kingdom Intravenous Solutions Market Size, Share, By Nutrition Type (Total Parenteral Nutrition, Peripheral Parenteral Nutrition), By Bag Type (Large Volume Bags (>250 ml), Small Volume Bags (250 ml)), and By End Users (Hospitals, Ambulatory Surgical Centers, Home Care), Analysis and Forecast 2026-2035
Report Overview
Table of Contents
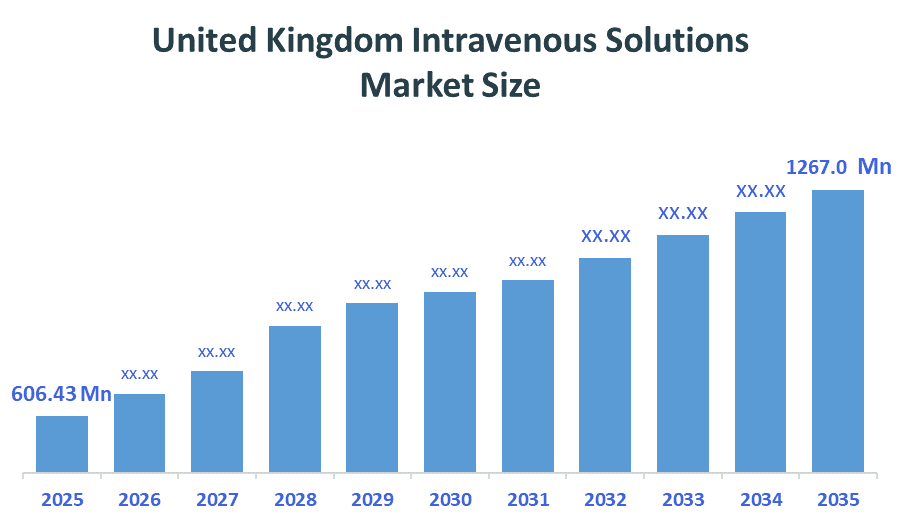
The United Kingdom Intravenous Solutions Market size is forecast to grow from approximately USD 606.43 Million in 2025 to around USD 1267.0Million by 2035. According to Decision Advisors, a detailed research report on the intravenous solutions market indicates that the Home Infusion trend dominates the market, accounting for approximately 55-60% of the total share in the United Kingdom. Baxter International leads the market with an estimated 18% United Kingdom market share. The company's annual revenue of approximately USD 11.3 billion is making it one of the most influential forces shaping industry trends and overall market growth.
Market Snapshot
- United Kingdom Intravenous Solutions Market Size (2025): USD 606.43 Million
- Projected United Kingdom Intravenous Solutions Market Size (2035): USD 1267.0 Million
- United Kingdom Intravenous Solutions Market Compound Annual Growth Rate (CAGR): 7.65%
- Base Year: 2025
- Historical Period: 2021–2024
- Forecast Period: 2026–2035
Market Overview/Introduction
The United Kingdom Intravenous Solutions Market refers to the production, distribution, and utilization of sterile fluids administered directly into veins across healthcare settings such as NHS hospitals, clinics, and home care. Intravenous solutions are liquid formulations containing electrolytes, nutrients, or medications used for hydration, nutrition, and drug delivery. Market growth is driven by rising chronic diseases, aging population, and increasing surgical procedures. Future opportunities lie in advanced parenteral nutrition, ready-to-use formulations, and expansion of home healthcare services. Additionally, technological innovations and efficient supply chain systems are expected to enhance product accessibility, improve patient outcomes, and support sustained market expansion in the United Kingdom.
- The Medicines Procurement and Supply Chain (MPSC) framework by NHS England ensures cost-effective procurement of medicines, including intravenous solutions and fluids. It standardizes pricing, secures supply continuity, and prevents monopolies through competitive tendering. This scheme directly supports the stable availability and affordability of IV products across NHS hospitals.
- The National Pricing Matrix initiative under NHS Supply Chain provides discounted pricing for healthcare products, including intravenous solutions, based on volume commitments. This scheme encourages bulk purchasing, reduces procurement costs, and improves affordability for NHS trusts, supporting wider adoption of IV therapies while maintaining supplier partnerships.
- The Pharmaceutical Price Regulation Scheme ensures that medicines supplied to the NHS are priced reasonably while allowing fair returns to manufacturers. It controls expenditure growth and supports access to essential treatments, including IV therapies, balancing affordability with innovation in the UK healthcare system.
Notable Insights: -
- The total parenteral nutrition segment dominated the market in 2025 and holds the largest market share, accounting for approximately 65% during the forecast period.
- The large volume bags (>250) ml segment dominated the market in 2025 and holds the largest market share, accounting for approximately 68% during the forecast period.
- The hospitals segment dominated the market in 2025 and holds the largest market share, accounting for approximately 70% during the forecast period.
- The compound annual growth rate of the United Kingdom Intravenous Solutions Market is 7.65%.
- The market is likely to achieve a valuation of USD 1267.0 Million by 2035.
What is the role of technology in grooming the market?
Technology significantly shapes the growth of the United Kingdom intravenous solutions market by enhancing clinical efficiency, safety, and supply chain management. Advanced infusion technologies, such as smart pumps with dose error reduction systems, minimize medication errors and improve patient outcomes. Automation in compounding and sterile manufacturing ensures consistent quality and compliance with stringent regulatory standards. Digital integration with electronic health records enables real-time monitoring of fluid administration and patient response, supporting data-driven clinical decisions. Additionally, innovations in packaging, including ready-to-use and prefilled IV bags, reduce preparation time and contamination risks. Supply chain technologies like predictive analytics and inventory tracking help prevent shortages and optimize distribution. These technological advancements collectively improve treatment reliability, operational efficiency, and accessibility across UK healthcare settings.
Market Drivers
The United Kingdom intravenous solutions market is primarily driven by the rising prevalence of chronic diseases such as cancer, gastrointestinal disorders, and kidney conditions, which require continuous fluid and nutritional support. Increasing surgical procedures and hospital admissions across NHS facilities further boost demand for intravenous therapies. The growing aging population also contributes significantly, as elderly patients often need long-term care and hydration management. Additionally, advancements in parenteral nutrition and ready-to-use formulations improve treatment efficiency and safety. Expansion of home healthcare services and outpatient care is another key driver, encouraging the adoption of convenient intravenous solutions and supporting overall market growth.
Restrain
The United Kingdom intravenous solutions market faces restraints from strict regulatory compliance and pricing pressures imposed by NHS procurement systems, which limit profit margins for manufacturers. Additionally, supply chain disruptions, high production costs, and dependency on imports for raw materials can affect product availability and slow market growth.
Study on the Supply, Demand, Distribution, and Market Environment of the United Kingdom Intravenous Solutions Market
The United Kingdom intravenous solutions market is characterized by a well-structured supply chain supported by domestic manufacturing and imports, ensuring consistent product availability despite occasional disruptions such as raw material shortages or logistics constraints. Demand is driven by increasing hospital admissions, rising prevalence of chronic diseases, and expanding surgical procedures, particularly within NHS facilities. Distribution is highly centralized, with procurement largely managed through national frameworks and group purchasing organizations, ensuring cost efficiency and standardized supply across healthcare institutions. The market environment is regulated by strict quality and safety standards, encouraging compliance and limiting entry of low-quality products. Additionally, growing emphasis on home healthcare and outpatient care is gradually reshaping demand patterns, promoting the adoption of user-friendly intravenous solutions and flexible distribution channels across the UK healthcare system.
Price Analysis and Consumer Behaviour Analysis
In the United Kingdom intravenous solutions market, pricing is largely influenced by centralized procurement through the NHS, bulk purchasing contracts, and strict regulatory standards, which help control costs but pressure supplier margins. High manufacturing costs due to sterility requirements and quality compliance also impact pricing structures. Consumer behavior is primarily institution-driven, with hospitals prioritizing cost-effective, clinically efficient solutions over brand preference. Demand is strongly linked to rising chronic diseases, aging population, and increased hospital admissions, particularly for oncology and critical care treatments. Additionally, the growing shift toward home healthcare and outpatient services is influencing purchasing patterns, with increased preference for ready-to-use, easy-to-administer formulations. Overall, value-based purchasing and clinical outcomes significantly guide buying decisions in the UK market.
Market Segmentation
The United Kingdom Intravenous Solutions Market Share is classified Into nutrition type, bag type, and end user.
The total parenteral nutrition segment dominated the market in 2025 and holds the largest market share, accounting for approximately 65% during the forecast period.
Based on the nutrition type, the intravenous solutions market is divided into Total parenteral nutrition and peripheral parenteral nutrition. Among these, the total parenteral nutrition segment dominated the market in 2025 and holds the largest market share, accounting for approximately 65% during the forecast period. The total parenteral nutrition segment is driven by its critical role in managing patients unable to receive adequate nutrition orally or enterally. ?? Rising prevalence of gastrointestinal disorders, cancer, and preterm births, along with increased demand for specialized hospital nutrition support, significantly drives adoption of TPN therapy across healthcare settings.
- The large volume bags (>250 ml) segment dominated the market in 2025 and holds the largest market share, accounting for approximately 68% during the forecast period.
Based on the bag type, the intravenous solutions market is divided into large volume bags (>250 ml) and small volume bags (250 ml). Among these, the large volume bags (>250 ml) segment dominated the market in 2025 and holds the largest market share, accounting for approximately 68% during the forecast period. The large volume bags (>250 ml) segment in the U.K. is attributed to their extensive use in hospitals for fluid resuscitation, electrolyte balance, and parenteral nutrition. Rising chronic disease prevalence, increasing surgical procedures, and higher inpatient admissions drive demand, as these settings require continuous, high-volume fluid administration for effective patient management.
- The hospitals segment dominated the market in 2025 and holds the largest market share, accounting for approximately 70% during the forecast period.
Based on the end user, the intravenous solutions market is divided into hospitals, ambulatory surgical centers, and home care. Among these, the hospitals segment dominated the market in 2025 and holds the largest market share, accounting for approximately 70% during the forecast period. The hospitals segment in the U.K. is driven by high patient inflow, critical care requirements, surgical procedures, and continuous IV therapy usage. Advanced healthcare infrastructure and reliance on IV treatments for acute and chronic conditions further strengthen this segment.
Recent Development
- In March 2026, the Department for Energy Security and Net Zero announced a national strategy to lead the global fusion energy race, aiming to strengthen long-term energy security and innovation.
- In May 2025, ICU Medical and Otsuka Pharmaceutical Factory completed a joint venture to strengthen IV solutions supply chains and enhance innovation. The partnership formed a large-scale global manufacturing network aimed at improving resilience and introducing advanced IV technologies, indirectly supporting supply stability across markets, including the United Kingdom.
Competitive Analysis
The report offers the appropriate analysis of the key organisations/companies involved within the United Kingdom intravenous solutions market, along with a comparative evaluation primarily based on their product offering, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborative analysis focusing on the current news and developments of the companies, which includes product development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
Top Key Companies in the United Kingdom Intravenous Solutions Market
- Baxter International Inc.
- Fresenius Kabi AG
- B. Braun SE
- Pfizer Inc.
- Otsuka Pharmaceutical Co., Ltd.
- Others
Key Target Audience
- Market Players
- Investors
- End-users
- Government Authorities
- Consulting and Research Firm
- Venture capitalists
- Value-Added Resellers (VARs)
Market Segment
This study forecasts revenue at the United Kingdom, regional, and country levels from 2020 to 2035. Decision Advisor has segmented the United Kingdom Intravenous Solutions Market based on the below-mentioned segments
United Kingdom Intravenous Solutions Market, By Nutrition Type
- Total Parenteral Nutrition
- Peripheral Parenteral Nutrition
United Kingdom Intravenous Solutions Market, By Bag Type
- Large Volume Bags (>250 ml)
- Small Volume Bags (250 ml)
United Kingdom Intravenous Solutions Market, By End User
- Hospitals
- Ambulatory Surgical Centers
- Home Care
Frequently Asked Questions (FAQ)
Q. How does sustainability influence the intravenous solutions market in the UK?
A. Sustainability is increasingly important in the United Kingdom intravenous solutions market as healthcare providers aim to reduce medical waste and carbon emissions. Hospitals are adopting eco-friendly IV packaging, recyclable materials, and energy-efficient manufacturing processes. NHS sustainability goals are also encouraging suppliers to develop greener supply chains while maintaining sterility and safety standards.
Q. What impact do shortages of medical raw materials have on IV solutions supply?
A. Shortages of key raw materials such as saline components and plastic polymers can disrupt production of intravenous solutions. In the UK, this leads to delayed hospital supplies and increased reliance on imports. Manufacturers are focusing on diversifying sourcing strategies and building buffer inventories to reduce dependency risks.
Q. How is home healthcare changing IV treatment delivery in the UK?
A. Home healthcare is transforming intravenous therapy by enabling patients to receive fluids and nutrition at home instead of hospitals. This reduces hospital burden, lowers costs, and improves patient comfort. Portable infusion devices and ready-to-use IV solutions are supporting this shift significantly in the UK market.
Q. What role do clinical guidelines play in IV solution usage in UK hospitals?
A. Clinical guidelines issued by healthcare authorities ensure standardized use of intravenous solutions across UK hospitals. They help clinicians choose appropriate fluid types, prevent overuse, and reduce complications such as fluid imbalance. This improves patient safety and ensures evidence-based treatment across NHS facilities.
Check Licence
Choose the plan that fits you best: Single User, Multi-User, or Enterprise solutions tailored for your needs.
We Have You Covered
- 24/7 Analyst Support
- Clients Across the Globe
- Tailored Insights
- Technology Tracking
- Competitive Intelligence
- Custom Research
- Syndicated Market Studies
- Market Overview
- Market Segmentation
- Growth Drivers
- Market Opportunities
- Regulatory Insights
- Innovation & Sustainability
Report Details
| Scope | country |
| Pages | 240 |
| Delivery | PDF & Excel via Email |
| Language | English |
| Release | Apr 2026 |
| Access | Download from this page |
