United Kingdom Lyophilized Injectable Drugs Market
United Kingdom Lyophilized Injectable Drugs Market Size, Share, By Drug Type (Biologics, Small molecule injectables, Vaccines, Peptides & Hormones), By Applications (Oncology, Anti-Infectives, Autoimmune Diseases, Vaccines), By End User (Hospitals, Specialty Clinics, Pharmaceutical companies, Contract Manufacturing Organizations (CMOs)), Analysis and Forecast 2026-2035
Report Overview
Table of Contents
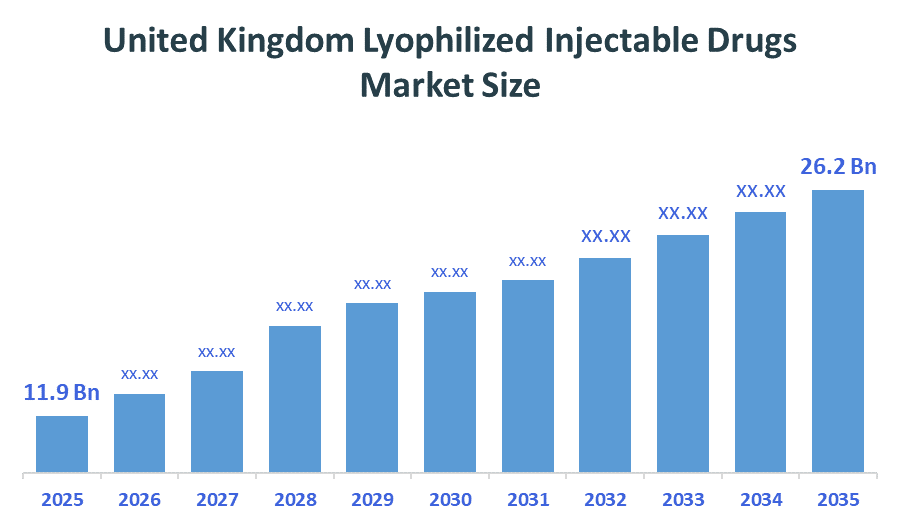
The United Kingdom Lyophilized Injectable Drugs Market size is forecast to grow from approximately USD 11.9 Billion in 2025 to around USD 26.2 Billion by 2035. According to Decision Advisors, a detailed research report on the lyophilized injectable drugs market indicates that the biologics and oncology-focused freeze-dried formulations trend dominates the market, accounting for approximately 30-35% of the total share in the United Kingdom. F. Hoffmann-La Roche leads the market with an estimated 7-8% United Kingdom market share. The company's annual revenue of approximately USD 70-75 billion is making it one of the most influential forces shaping industry trends and overall market growth.
Market Snapshot
- United Kingdom Lyophilized Injectable Drugs Market Size (2025): USD 11.9 Billion
- Projected United Kingdom Lyophilized Injectable Drugs Market Size (2035): USD 26.2 Billion
- United Kingdom Lyophilized Injectable Drugs Market Compound Annual Growth Rate (CAGR): 8.21%
- Base Year: 2025
- Historical Period: 2021–2024
- Forecast Period: 2026–2035
Market Overview/Introduction
The UK Lyophilized Injectables Drugs market can be defined as the pharmaceutical industry that is responsible for research, development, manufacture, distribution, and use of freeze-dried injectable medicines in the UK healthcare system to serve treatments for biologics, oncology, and intensive care indications. Lyophilized Injectable Drugs are sterile injectable medications that are freeze-dried under vacuum to extract water from the drug formulations, increasing the drugs' stability and shelf-life and simplifying shipment before they can be reconstituted to prepare for intravenous (IV) administration. Market opportunities are in the growth of biologics, personalized medications, and advanced oncology treatments, together with contract manufacturing and biosimilar markets. The factors driving market growth include increasing burden of chronic diseases, increasing need of stable injectable drugs, NHS-supported purchasing, and rapid technology innovation in freeze-drying and sterile manufacturing, which leads to improvement in efficiency and safety in addition to drug delivery.
- The UK government launched this fund to strengthen domestic manufacturing of medicines, including advanced injectables and biologics. It provides capital grants to expand production facilities, improve resilience, and accelerate access to new therapies. The scheme supports GMP-compliant manufacturing of human medicines, helping companies scale lyophilized injectable production and reduce dependency on imports.
- The government scheme supports upgrading manufacturing capabilities for medicines and diagnostics. It focuses on improving supply chain resilience and enabling faster production of critical drugs, including injectable and freeze-dried formulations. The scheme helps attract investment into UK-based production of complex pharmaceuticals such as lyophilized biologics used in hospitals and emergency care.
Notable Insights: -
- The biologics management segment dominated the market in 2025 and holds the largest market share, accounting for approximately 60% during the forecast period.
- The oncology segment dominated the market in 2025 and holds the largest market share, accounting for approximately 45% during the forecast period.
- The hospitals segment dominated the market in 2025 and holds the largest market share, accounting for approximately 65% during the forecast period.
- The compound annual growth rate of the United Kingdom Lyophilized Injectable Drugs Market is 8.21%.
- The market is likely to achieve a valuation of USD 26.2 Billion by 2035.
What is the role of technology in grooming the market?
Technology plays a crucial role in grooming the United Kingdom lyophilized injectable drugs market by enhancing drug stability, efficiency, and large-scale production capabilities. Advanced freeze-drying (lyophilization) systems enable precise control of temperature and pressure, ensuring improved preservation of biologics and oncology injectables. Automation and AI-driven monitoring systems optimize cycle times, reduce human error, and maintain consistent product quality. Innovations in aseptic processing and dual-chamber packaging improve reconstitution speed and patient safety. Cold-chain logistics technologies, including smart tracking and temperature sensors, ensure product integrity during storage and distribution. Additionally, continuous manufacturing and process analytical technology (PAT) help pharmaceutical companies reduce costs and accelerate production timelines. These technological advancements collectively support higher efficiency, reduced wastage, and expanded adoption of complex injectable therapies across the UK healthcare system.
Market Drivers
The United Kingdom Lyophilized injectable drugs market is driven by the rising prevalence of chronic diseases such as cancer, autoimmune disorders, and infectious diseases, which significantly increase demand for stable and effective injectable therapies. The growing adoption of biologics and oncology drugs, which often require lyophilization for stability, is a major growth factor. Expansion of hospital-based care and NHS procurement programs further supports market demand through bulk purchasing and standardized treatment protocols. Additionally, advancements in freeze-drying technology, improved drug shelf life, and increasing outsourcing to contract manufacturing organizations enhance production efficiency and accelerate market growth across the UK healthcare system.
Restrain
The United Kingdom lyophilized injectable drugs market faces restraints due to the high manufacturing and lyophilization costs, complex cold-chain logistics, and strict regulatory compliance requirements. Limited domestic production capacity and dependence on imports also constrain supply efficiency. Additionally, reimbursement pressures from the NHS restrict pricing flexibility and slow adoption of premium biologics.
Study on the Supply, Demand, Distribution, and Market Environment of the United Kingdom Lyophilized Injectable Drugs Market
The United Kingdom lyophilized injectable drugs market operates in a highly regulated, innovation-driven, and supply-chain sensitive environment where demand is mainly shaped by hospitals, NHS procurement systems, and specialty care providers. Supply is dominated by global pharmaceutical companies and expanding CMOs that provide sterile fill–finish and freeze-drying capacity, while imports of biologics and oncology injectables remain significant due to limited domestic manufacturing scale. Demand is steadily rising because of increasing use of biologics, oncology therapies, and temperature-sensitive injectables requiring long shelf life and stability. Distribution is largely centralized through hospital pharmacies and NHS tender-based procurement, ensuring controlled pricing and bulk purchasing efficiency. The market environment is characterized by strong regulatory oversight, high production costs, and growing outsourcing trends, while technological advancements in lyophilization and cold-chain optimization are improving reliability and reducing wastage across the supply system.
Price Analysis and Consumer Behaviour Analysis
The United Kingdom lyophilized injectable drugs market, pricing is generally high due to complex manufacturing, cold-chain requirements, and biologics dominance, with average treatment costs significantly higher than conventional injectables. Prices are strongly influenced by R&D intensity, sterility assurance, and freeze-drying technology costs, making oncology and biologic lyophilized products the most expensive segment. However, NHS bulk procurement and tender-based purchasing help stabilize final hospital acquisition costs and reduce variability across institutions. Consumer behaviour is primarily institution-driven rather than patient-driven, as hospitals, specialty clinics, and NHS procurement bodies control demand. Physicians prefer lyophilized injectables for their longer shelf life, improved stability, and reduced wastage, especially in oncology and emergency care. Increasing adoption of biologics and rising trust in advanced injectable therapies are driving consistent demand growth, while cost containment pressures push healthcare providers toward value-based purchasing and CMO-sourced generics where possible.
Market Segmentation
The United Kingdom Lyophilized Injectable Drugs Market Share is classified into drug type, applications, and end user.
- The biologics management segment dominated the market in 2025 and holds the largest market share, accounting for approximately 60% during the forecast period.
Based on the drug type, the lyophilized injectable drugs market is divided into biologics, small molecule injectables, vaccines, and peptides & hormones. Among these, the biologics management segment dominated the market in 2025 and holds the largest market share, accounting for approximately 60% during the forecast period. The biologics management segment is driven by strong demand for oncology biologics, monoclonal antibodies, and rare disease treatments. Their sensitivity to degradation makes lyophilization essential for stability, boosting adoption across hospitals and specialty care settings.
- The oncology segment dominated the market in 2025 and holds the largest market share, accounting for approximately 45% during the forecast period.
Based on the applications, the lyophilized injectable drugs market is divided into oncology, anti-infectives, autoimmune diseases, and vaccines. Among these, the oncology segment dominated the market in 2025 and holds the largest market share, accounting for approximately 45% during the forecast period. The oncology segment in the U.K. is attributed to rising cancer prevalence and increasing use of freeze-dried chemotherapy and targeted biologics are key drivers. Lyophilized injectables ensure long shelf life and precise dosing, making them critical in hospital-based cancer treatment protocols.
- The hospitals segment dominated the market in 2025 and holds the largest market share, accounting for approximately 65% during the forecast period.
Based on the end user, the lyophilized injectable drugs market is divided into hospitals, specialty clinics, pharmaceutical companies, and contract manufacturing organizations (CMOs). Among these, the hospitals segment dominated the market in 2025 and holds the largest market share, accounting for approximately 65% during the forecast period. The hospitals segment in the U.K. is driven by high inpatient usage, NHS-driven procurement, and widespread administration of oncology and emergency biologic therapies. Hospitals remain the primary hub for sterile injectable preparation and administration.
Recent Development
- In December 2025, Cipla and Pfizer entered into an exclusive distribution and marketing agreement for select injectable and critical care-related pharmaceutical products, strengthening global access to essential medicines. Under the partnership, Pfizer continued manufacturing while Cipla managed distribution through its strong supply chain network, improving availability of high-demand therapies often used in hospital and critical care settings. The collaboration highlighted a growing industry trend toward strategic alliances aimed at expanding access to injectable formulations.
- In June 2025, The UK lyophilized injectable drugs market observed continued product development activity as pharmaceutical manufacturers focused on stable formulations across key therapeutic areas, including antibiotics, vaccines, enzymes, and monoclonal antibodies. These launches were aligned with the growing demand for freeze-dried injectables that offer improved shelf life, enhanced stability, and reduced cold-chain dependency.
- In March 2025, The UK lyophilized injectable drugs market witnessed a growing focus on high-stability formulations as pharmaceutical companies advanced new product launches targeting critical care, oncology, and autoimmune diseases. These lyophilized powder-based injectable therapies were developed to improve drug stability, extend shelf life, and ensure better handling of temperature-sensitive biologics.
Competitive Analysis
The report offers the appropriate analysis of the key organisations/companies involved within the United Kingdom Lyophilized Injectable Drugs Market, along with a comparative evaluation primarily based on their product offering, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborative analysis focusing on the current news and developments of the companies, which includes product development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
Top Key Companies in the United Kingdom Lyophilized Injectable Drugs Market
- Pfizer Inc.
- F. Hoffmann-La Roche Ltd
- Sanofi S.A.
- Merck & Co., Inc.
- GlaxoSmithKline (GSK) plc
- Others
Key Target Audience
- Market Players
- Investors
- End-users
- Government Authorities
- Consulting and Research Firm
- Venture capitalists
- Value-Added Resellers (VARs)
Market Segment
This study forecasts revenue at the United Kingdom, regional, and country levels from 2020 to 2035. Decision Advisor has segmented the United Kingdom Lyophilized Injectable Drugs Market based on the below-mentioned segments
United Kingdom Lyophilized Injectable Drugs Market, By Drug Type
- Biologics Management
- Small molecule injectables
- Vaccines
- Peptides & hormones
United Kingdom Lyophilized Injectable Drugs Market, By Applications
- Oncology
- Anti-infectives
- Autoimmune diseases
- Vaccines
United Kingdom Lyophilized Injectable Drugs Market, By End User
- Hospitals
- Specialty Clinics
- Pharmaceutical companies
- Contract Manufacturing Organizations (CMOs)
Frequently Asked Questions (FAQ)
Q. How does NHS procurement influence the lyophilized injectable drugs market in the UK?
A. The NHS plays a central role by using centralized tender-based purchasing, which ensures bulk buying of lyophilized injectables at negotiated prices. This system stabilizes demand, improves accessibility across hospitals, and encourages manufacturers to comply with strict quality and cost-efficiency standards, shaping overall market competitiveness.
Q. Why are Contract Manufacturing Organizations (CMOs) becoming important in this market?
A. CMOs are increasingly important due to limited domestic sterile manufacturing capacity in the UK. They provide specialized freeze-drying, aseptic fill-finish, and scalable production services. Pharmaceutical companies rely on CMOs to reduce costs, accelerate drug launches, and meet growing demand for biologics and oncology injectables.
Q. What challenges does cold-chain logistics create for lyophilized injectable drugs in the UK?
A. Although lyophilized drugs are more stable than liquid injectables, they still require controlled storage and transport conditions after reconstitution. Maintaining temperature integrity, avoiding moisture exposure, and ensuring timely delivery across NHS facilities increases operational costs and complexity in distribution networks.
Q. How is the rise of personalized medicine affecting this market?
A. Personalized medicine is increasing demand for small-batch, high-value injectable formulations, especially in oncology and rare diseases. Lyophilization supports this trend by enabling long shelf life for customized biologics, making it easier to store and distribute patient-specific therapies safely and efficiently.
Check Licence
Choose the plan that fits you best: Single User, Multi-User, or Enterprise solutions tailored for your needs.
We Have You Covered
- 24/7 Analyst Support
- Clients Across the Globe
- Tailored Insights
- Technology Tracking
- Competitive Intelligence
- Custom Research
- Syndicated Market Studies
- Market Overview
- Market Segmentation
- Growth Drivers
- Market Opportunities
- Regulatory Insights
- Innovation & Sustainability
Report Details
| Scope | country |
| Pages | 240 |
| Delivery | PDF & Excel via Email |
| Language | English |
| Release | Apr 2026 |
| Access | Download from this page |
