United Kingdom Orphan Drugs Market
United Kingdom Orphan Drugs Market Size, Share, By Drug Type (Biologics, and Non-biologics), By Route of Administration (Oral, Injectable, and Others), and By Distribution Channel (Hospital Pharmacies, Specialty Pharmacies, and Online Pharmacies) Analysis and Forecast 2026-2035.
Report Overview
Table of Contents
Market Snapshot
- United Kingdom Orphan Drugs Market size (2025): USD 170.5 Billion
- Projected United Kingdom Orphan Drugs Market size (2035): USD 480.9 Billion
- United Kingdom Orphan Drugs Market Compound Annual Growth Rate (CAGR): 10.93%
- Base Year: 2025
- Historical Period: 2021–2024
- Forecast Period: 2026–2035
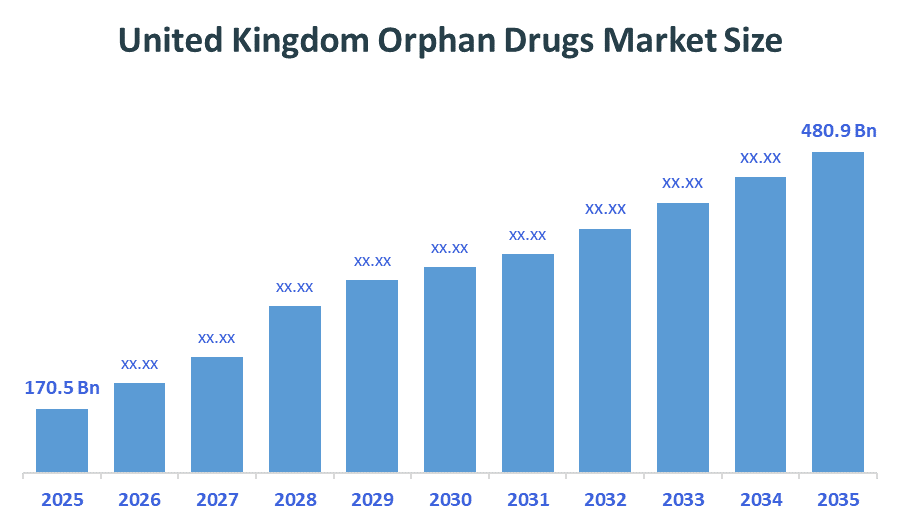
According to Decision Advisors, the United Kingdom Orphan Drugs Market size was grown from USD 170.5 Billion in 2025 to USD 480.9 Billion by 2035, at a CAGR of 10.93% during the forecast period 2026-2035. The United Kingdom orphan drug market is projected to grow significantly over the next decade, fueled by the innovative licensing and access pathway (ILAP), the innovative medicines fund, high demand for specialized therapies, accelerated regulatory pathways, rising prevalence and diagnosis rates, increased R&D and strategic focus, and market exclusivity and incentives.
Market Overview/Introduction
The United Kingdom Orphan Drugs Market describes the process, which includes developing rare disease medicines with specific production methods through their actual commercial distribution. Orphan drugs are specialized therapies that exist to treat rare diseases that cause life-threatening conditions and permanent disabilities. The market is experiencing consistent growth because people are becoming more aware of rare diseases, genomic research is advancing, and the National Health Service provides strong support to the industry. The upcoming market opportunities will arise from gene therapies, personalized medicine, and AI-based drug discovery solutions. The biotechnology sector experiences growth because of government policies that create faster approval processes and increased funding for research, which allows companies to develop new products while improving patient treatment options.
- The Medicines and Healthcare Products Regulatory Agency operated a UK-specific orphan medicinal product framework that granted incentives to companies developing rare disease treatments. This initiative supported innovation, reduced development barriers, encouraged investment, and strengthened the overall ecosystem for orphan drug research and commercialization.
- The Department of Health and Social Care and NHS England implemented the England Rare Diseases Action Plan 2026, which improved access to orphan and rare-disease medicines. It strengthened research support, enhanced diagnosis, and encouraged innovation, thereby boosting market growth and patient outcomes.
Notable Insights: -
- The non-biologics segment dominated the market in 2025 and holds the largest market share, accounting for approximately 90% during the forecast period.
- The oral segment dominated the market in 2025 and holds the largest market share, accounting for approximately 65% during the forecast period.
- The hospital pharmacies segment dominated the market in 2025 and holds the largest market share, accounting for approximately 50% during the forecast period.
- The compound annual growth rate of the United Kingdom Orphan Drugs Market is 10.93%.
- The market is likely to achieve a valuation of USD 480.9Billion by 2035.
What is the role of technology in grooming the market?
Technology plays a transformative role in advancing the orphan drugs market in the United Kingdom by accelerating drug discovery, improving diagnosis, and enhancing patient outcomes. Innovations such as genomics, artificial intelligence, and precision medicine enable early identification of rare diseases and targeted therapy development. Digital health tools and real-world data analytics support clinical trials and post-market surveillance, improving efficiency and reducing costs. Organizations like the National Health Service increasingly use electronic health records and data integration to streamline patient access to specialized treatments. Additionally, advancements in biologics manufacturing and gene therapy technologies are enabling scalable production of complex orphan drugs, fostering innovation and expanding treatment availability across the market.
Market Drivers
The UK orphan drugs market is growing because of things like the Innovative Licensing and Access Pathway. This program helps get treatments to patients faster. The Innovative Medicines Fund is also important. It helps patients get treatments for diseases that do not a lot of proof that they work. More patients are now aware of diseases, and doctors are diagnosing them more often. This means more people are getting the help they need. The pharmaceutical industry is really focused on making medicine and targeted treatments. They are working on things like monoclonal antibodies and gene therapies for the UK orphan drugs market. The rules that govern the UK orphan drugs market also help companies because they get to be the ones selling a treatment for a while. This makes it worth investing in the UK orphan drugs market.
Restrain
The United Kingdom orphan drugs market faces restraints due to high treatment costs, strict pricing assessments by the National Institute for Health and Care Excellence, and limited patient populations affecting commercial viability. Additionally, complex regulatory processes and supply chain dependencies can delay product availability and restrict broader patient access.
Study on the Supply, Demand, Distribution, and Market Environment of the United Kingdom Orphan Drugs Market
The United Kingdom Orphan Drugs Market is characterized by a specialized supply chain driven by advanced biologics manufacturing and limited production volumes. Supply depends on global pharmaceutical leaders and biotech firms, with critical raw materials including active pharmaceutical ingredients (APIs), biologic cell lines, and specialized excipients sourced internationally. Demand is steadily rising due to improved rare disease diagnosis, supported by institutions like the National Health Service, which ensures patient access through reimbursement frameworks. Distribution is highly regulated, involving hospital pharmacies, specialty distributors, and controlled logistics to maintain drug integrity. The market environment is shaped by strong regulatory support, incentives for orphan designation, and pricing scrutiny. Additionally, collaboration between research institutions and pharmaceutical companies enhances innovation, while supply chain disruptions and high manufacturing costs remain key challenges influencing market dynamics.
Price Analysis and Consumer Behaviour Analysis
The pricing of United Kingdom orphan drugs is typically high due to limited patient populations, complex R&D processes, and regulatory incentives such as market exclusivity. Prices are often negotiated and reimbursed through the National Health Service, which plays a critical role in cost control and patient access. Consumer behavior is largely influenced by physician recommendations and government reimbursement policies rather than direct purchasing decisions. Patients and caregivers prioritize treatment efficacy, availability, and long-term outcomes over cost. Additionally, increasing awareness of rare diseases and advocacy group support has improved diagnosis rates and demand for innovative therapies, while value-based pricing models are gradually shaping adoption patterns across the market.
Market Segmentation
The United Kingdom Orphan Drugs Market share is classified into drug type, route of administration, and distribution channel
- The non-biologics segment dominated the market in 2025 and holds the largest market share, accounting for approximately 90% during the forecast period.
Based on the drug type, the orphan drugs market is divided into biologics, and non-biologics. Among these, the non-biologics segment dominated the market in 2025 and holds the largest market share, accounting for approximately 90% during the forecast period. The non-biologics segment is driven by their lower production costs, easier manufacturing processes, and established regulatory pathways. These drugs offer greater affordability and wider accessibility through the National Health Service, making them preferable for treating rare conditions compared to expensive biologics.
- The oral segment dominated the market in 2025 and holds the largest market share, accounting for approximately 65% during the forecast period.
Based on the route of administration, the orphan drugs market is divided into oral, injectable, and others. Among these, the oral segment dominated the market in 2025 and holds the largest market share, accounting for approximately 65% during the forecast period. The oral segment in the U.K. is attributed to its ease of administration, improved patient compliance, and suitability for long-term treatment of rare chronic conditions. Oral drugs reduce hospitalization needs and are cost-effective for the National Health Service, enhancing accessibility and widespread adoption.
- The hospital pharmacies segment dominated the market in 2025 and holds the largest market share, accounting for approximately 50% during the forecast period.
Based on the distribution channel, the orphan drugs market is divided into hospital pharmacies, specialty pharmacies, and online pharmacies. Among these, the hospital pharmacies segment dominated the market in 2025 and holds the largest market share, accounting for approximately 50% during the forecast period. The hospital pharmacies segment in the U.K. is driven by the centralized treatment delivery for rare diseases requiring specialist supervision. The National Health Service primarily distributes high-cost orphan drugs through hospitals, ensuring controlled access, proper administration, and monitoring, which supports higher reliance on hospital-based dispensing channels.
Recent Development
- In February 2026, Brexit, the UK’s orphan-drug framework continued to offer up to 10 years of market exclusivity, extendable to 12 years upon completion of a Paediatric Investigation Plan (PIP). This policy strengthened incentives for pharmaceutical companies, encouraged rare disease innovation, and improved long-term market attractiveness.
- In February 2026, the Regulatory changes established that orphan designation was assessed at the time of Marketing Authorisation Application (MAA) rather than as a separate pre-approval step. This streamlined the approval process, reduced administrative burden, accelerated time-to-market, and enhanced efficiency for pharmaceutical companies developing rare disease therapies.
- In December 2024, the Legal and regulatory commentators highlighted that, orphan-drug protection became aligned across the entire UK rather than being limited to Great Britain. This harmonization improved regulatory clarity, ensured uniform patient access, and enhanced market attractiveness for pharmaceutical companies developing rare disease treatments.
Competitive Analysis
The report offers the appropriate analysis of the key organisations/companies involved within the United Kingdom orphan drugs market, along with a comparative evaluation primarily based on their product offering, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborative analysis focusing on the current news and developments of the companies, which includes product development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
Top Key Companies in the United Kingdom Orphan Drugs Market
- F. Hoffmann-La Roche
- Novartis AG
- AstraZeneca
- Sanofi
- Pfizer Inc.
- Others
Key Target Audience
- Market Players
- Investors
- End-users
- Government Authorities
- Consulting and Research Firm
- Venture capitalists
- Value-Added Resellers (VARs)
Market Segment
This study forecasts revenue at the United Kingdom, regional, and country levels from 2020 to 2035. Spherical Insights has segmented the United Kingdom orphan drugs market based on the below-mentioned segments
United Kingdom Orphan Drugs Market, By Drug Type
- Biologics
- Non-biologics
United Kingdom Orphan Drugs Market, By Route of Administration
- Oral
- Injectable
- Others
United Kingdom Orphan Drugs Market, By Distribution Channel
- Hospital Pharmacies
- Specialty Pharmacies
- Online Pharmacies
Frequently Asked Questions (FAQ)
Q. What are the key regulatory bodies influencing the UK orphan drugs market?
A. The Medicines and Healthcare products Regulatory Agency and the National Institute for Health and Care Excellence play crucial roles. MHRA oversees approvals and orphan designation, while NICE evaluates cost-effectiveness and reimbursement, directly impacting pricing, market access, and adoption of orphan therapies.
Q. How does reimbursement impact orphan drug adoption in the UK?
A. Reimbursement through the National Health Service significantly improves patient access to expensive orphan drugs. Government-backed funding programs reduce financial burden on patients, enabling wider adoption and ensuring that high-cost rare disease treatments reach eligible populations efficiently.
Q. What challenges do pharmaceutical companies face in the UK orphan drugs market?
A. Companies face challenges such as stringent cost-effectiveness evaluations by NICE, high R&D costs, and limited patient populations. Additionally, supply chain complexities and regulatory compliance requirements can delay product launches and impact profitability despite strong market incentives.
Q. How is research collaboration shaping the UK orphan drugs market?
A. Collaborations between academic institutions, biotech firms, and pharmaceutical companies are accelerating innovation. These partnerships enhance clinical research, improve understanding of rare diseases, and support faster development of targeted therapies, strengthening the UK’s position as a hub for rare disease research.
Check Licence
Choose the plan that fits you best: Single User, Multi-User, or Enterprise solutions tailored for your needs.
We Have You Covered
- 24/7 Analyst Support
- Clients Across the Globe
- Tailored Insights
- Technology Tracking
- Competitive Intelligence
- Custom Research
- Syndicated Market Studies
- Market Overview
- Market Segmentation
- Growth Drivers
- Market Opportunities
- Regulatory Insights
- Innovation & Sustainability
Report Details
| Scope | Country |
| Pages | 210 |
| Delivery | PDF & Excel via Email |
| Language | English |
| Release | Apr 2026 |
| Access | Download from this page |
