United Kingdom Peptide and Anticoagulant Drugs Market
United Kingdom Peptide and Anticoagulant Drugs Market Size, Share, By Drug Type (Peptide Drugs, and Anticoagulant Drugs), By Route of Administration (Oral, Injectable, and Others), and By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, and Online Pharmacies) Analysis and Forecast 2026-2035.
Report Overview
Table of Contents
Market Snapshot
- United Kingdom Peptide and Anticoagulant Drugs Market size (2025): USD 1.56 Billion
- Projected United Kingdom Peptide and Anticoagulant Drugs Market size (2035): USD 4.31 Billion
- United Kingdom Peptide and Anticoagulant Drugs Market Compound Annual Growth Rate (CAGR): 10.7%
- Base Year: 2025
- Historical Period: 2021–2024
- Forecast Period: 2026–2035
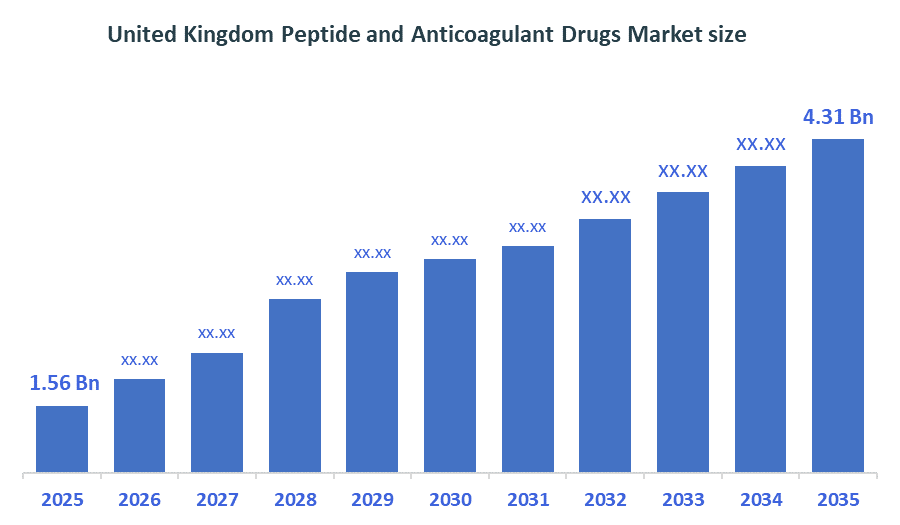
According to Decision Advisors, the United Kingdom Peptide and Anticoagulant Drugs Market size was grown from USD 1.56billion in 2025 to USD 4.31billion by 2035, at a CAGR of 10.7% during the forecast period 2026-2035. The United Kingdom peptide and anticoagulant drugs market is projected to grow significantly over the next decade, fueled by the growing incidence of thromboembolic conditions and the heightened inclination towards next-generation peptide therapies that provide greater effectiveness and reduced side effects. Enhanced diagnostic rates, expanding healthcare infrastructure, and government efforts supporting cardiovascular health management
Market Overview/Introduction
The United Kingdom Peptide and Anticoagulant Drugs Market refers to the key market players that develop, manufacture, and sell peptide-based therapeutics and anticoagulant medications, which doctors prescribe to treat blood clotting disorders. The market offers pharmaceutical products that include peptide therapies, that includes use of short amino acid chains for specific medical applications and anticoagulant medications, which serve to decrease blood coagulation for thrombus prevention. The market will experience substantial growth opportunities because of major factors, such as biotechnological breakthroughs, the rising demand for precise medical treatments, and the introduction of safer and more efficient oral anticoagulant medications. The market experiences continuous global expansion because of major main factors the increasing number of people who develop cardiovascular diseases, the growing elderly population, the rising public understanding of healthcare, and the better availability of new medical treatments.
- The UK Government, through Innovate UK, invested over £54 million in November 2025 under the Sustainable Medicines Manufacturing Innovation Programme (SMMIP) to enhance medicine manufacturing efficiency, sustainability, and advanced production technologies, supporting innovation in the United Kingdom peptide and anticoagulant drugs market.
- The Innovative Licensing and Access Pathway (ILAP), led by the Medicines and Healthcare products Regulatory Agency (MHRA), is a UK regulatory scheme that accelerates the development and access of innovative medicines through integrated collaboration with NHS England and health technology assessment bodies such as NICE, providing early guidance, priority regulatory support, and faster patient access, thereby supporting growth in the United Kingdom peptide and anticoagulant drugs market.
Notable Insights: -
- The anticoagulant drugs segment dominated the market in 2025 and holds the largest market share, accounting for approximately 60% during the forecast period.
- The injectable segment dominated the market in 2025 and holds the largest market share, accounting for approximately 45% during the forecast period.
- The hospital pharmacies segment dominated the market in 2025 and holds the largest market share, accounting for approximately 50% during the forecast period.
- The compound annual growth rate of the United Kingdom Peptide and Anticoagulant Drugs Market is 10.7%.
- The market is likely to achieve a valuation of USD 4.31Billion by 2035.
What is the role of technology in grooming the market?
The United Kingdom peptide and anticoagulant drugs market depends on technology because it improves drug discovery processes, manufacturing operations, and patient treatment systems. Advanced peptide synthesis technologies, together with biologics manufacturing methods, have created efficient manufacturing systems that produce higher precision results while decreasing production expenses. Digital health tools that combine AI-powered diagnostic systems with predictive analytics enable healthcare professionals to detect thrombotic risks during the early stage of patient assessment. The National Health Service implemented electronic prescribing systems, which enable doctors to monitor patient medication consumption and verify that patients adhere to their prescribed treatment. Patients achieve superior treatment outcomes because healthcare providers create drug delivery systems along with permanent medical solutions, which enable patients to access their treatments more effortlessly. The Medicines and Healthcare Products Regulatory Agency provides regulatory support that accelerates advanced therapy adoption to create a market system that improves patient care while promoting innovation.
Market Drivers
The United Kingdom Peptide and Anticoagulant Drugs Market is driven by consumers developing thrombosis and cardiovascular diseases, and metabolic disorders that need effective anticoagulant treatments. The ongoing progress in biotechnology and drug development results in the creation of next-generation anticoagulants together with improved peptide-based treatment options. The healthcare industry experiences a rising preference for novel oral anticoagulants (NOACs) and targeted peptide therapeutics because these medications offer improved safety profiles and reduced adverse effects compared to conventional treatment options. The UK market experiences high demand for its products because the country has an increasing elderly population that faces a greater risk of developing clotting disorders. The major factors of improved healthcare access and supportive reimbursement frameworks, and increased availability of advanced therapies contribute to the ongoing growth and spread of these medications throughout the nation.
Restrain
The United Kingdom peptide and anticoagulant drugs market faces restraints due to stringent pricing regulations imposed by the National Health Service and cost-effectiveness evaluations by the National Institute for Health and Care Excellence. High development costs, complex manufacturing of peptides, and potential side effects like bleeding risks further limit market growth.
Study on the Supply, Demand, Distribution, and Market Environment of the United Kingdom Peptide and Anticoagulant Drugs Market
The United Kingdom peptide and anticoagulant drugs market demonstrates a balanced supply–demand structure driven by rising cardiovascular and thrombotic disease prevalence. Demand is increasing due to an aging population and greater adoption of advanced biologics and oral anticoagulants. Supply is supported by global pharmaceutical manufacturers and domestic production, with strong regulatory oversight from the Medicines and Healthcare Products Regulatory Agency ensuring quality and safety. Distribution is highly streamlined through hospital pharmacies, retail chains, and centralized procurement via the National Health Service. The market environment is competitive yet regulated, encouraging innovation while controlling costs. Raw materials for peptide drugs include amino acids and specialized synthesis reagents, often sourced globally, while anticoagulants rely on chemical intermediates and biologically derived components, making supply chains sensitive to geopolitical and manufacturing disruptions.
Price Analysis and Consumer Behaviour Analysis
The United Kingdom peptide and anticoagulant drugs market exhibits controlled pricing dynamics due to strong regulation by the National Health Service (NHS) and cost-effectiveness assessments by the National Institute for Health and Care Excellence. Prices of anticoagulants, especially novel oral anticoagulants, remain relatively high but are balanced through reimbursement frameworks and generic competition. Peptide drugs, often complex to manufacture, command premium pricing due to innovation and limited alternatives. Consumer behavior is largely prescriber-driven, with physicians favoring clinically proven and guideline-recommended therapies. Patients in the UK show high adherence due to subsidized access and awareness of cardiovascular risks. Additionally, demand is rising with an aging population, increasing incidence of thrombotic disorders, and preference for safer, more convenient oral anticoagulant options over traditional therapies.
Market Segmentation
The United Kingdom Peptide and Anticoagulant Drugs Market share is classified into drug type, route of administration, and distribution channel
- The anticoagulant drugs segment dominated the market in 2025 and holds the largest market share, accounting for approximately 60% during the forecast period.
Based on the drug type, the peptide and anticoagulant drugs market is divided into peptide drugs and anticoagulant drugs. Among these, the anticoagulant drugs segment dominated the market in 2025 and holds the largest market share, accounting for approximately 60% during the forecast period. The anticoagulant drugs segment is driven by the high prevalence of cardiovascular diseases and thrombotic disorders. The market share for this product will increase because of its widespread clinical use, together with National Health Service recommendations and the availability of modern oral anticoagulants.
- The injectable segment dominated the market in 2025 and holds the largest market share, accounting for approximately 45% during the forecast period.
Based on the route of administration, the peptide and anticoagulant drugs market is divided into oral, injectable, and others. Among these, the injectable segment dominated the market in 2025 and holds the largest market share, accounting for approximately 45% during the forecast period. The injectable segment in the U.K. is attributed to the fact that the drug demonstrates a fast onset of effects and shows high effectiveness in emergency medical treatment. The National Health Service treatment protocols create a strong market demand for injectable anticoagulants, which hospitals use as their preferred method for providing immediate anticoagulation treatment.
- The hospital pharmacies segment dominated the market in 2025 and holds the largest market share, accounting for approximately 50% during the forecast period.
Based on the distribution channel, the peptide and anticoagulant drugs market is divided into hospital pharmacies, retail pharmacies, and online pharmacies. Among these, the hospital pharmacies segment dominated the market in 2025 and holds the largest market share, accounting for approximately 50% during the forecast period. The hospital pharmacies segment in the U.K. is driven by the hospital experiences high patient volume for cardiovascular and thrombotic treatment services. The National Health Service operates centralized procurement and distribution systems, which maintain uninterrupted supply chains while hospital facilities use special injectable medications to enhance their market position in this particular segment.
Recent Development
- In September 2026, Sirius Therapeutics is advancing its siRNA candidate (SRSD107) targeting Factor XI into Phase 2 clinical trials, aiming to develop a long-acting anticoagulant with infrequent dosing, potentially administered twice yearly to improve patient compliance and safety.
- In May 2024, Research from the University of Geneva and University of Sydney did introduce a reversible peptide-based anticoagulant with an on-demand antidote. The drug uses two peptide components linked by peptide nucleic acid (PNA), which can be rapidly deactivated using an antidote, improving safety during surgery and reducing bleeding risks.
- In February 2024, the launch of Orbis Medicines backed by €26 million in seed funding, highlights growing investment in the development of orally available macrocyclic peptide therapies, reflecting broader innovation trends influencing the European and indirectly the United Kingdom peptide and anticoagulant drugs market.
Competitive Analysis
The report offers the appropriate analysis of the key organisations/companies involved within the United Kingdom peptide and anticoagulant drugs market, along with a comparative evaluation primarily based on their product offering, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborative analysis focusing on the current news and developments of the companies, which includes product development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
Top Key Companies in the United Kingdom Peptide and Anticoagulant Drugs Market
- AstraZeneca
- GlaxoSmithKline (GSK)
- Pfizer Inc.
- Sanofi
- Bristol-Myers Squibb
- Others
Key Target Audience
- Market Players
- Investors
- End-users
- Government Authorities
- Consulting and Research Firm
- Venture capitalists
- Value-Added Resellers (VARs)
Market Segment
This study forecasts revenue at the United Kingdom, regional, and country levels from 2020 to 2035. Spherical Insights has segmented the United Kingdom peptide and anticoagulant drugs market based on the below-mentioned segments
United Kingdom Peptide and Anticoagulant Drugs Market, By Drug Type
- Peptide Drugs
- Anticoagulant Drugs
United Kingdom Peptide and Anticoagulant Drugs Market, By Route of Administration
- Oral
- Injectable
- Others
United Kingdom Peptide and Anticoagulant Drugs Market, By Distribution Channel
- Hospital Pharmacies
- Retail Pharmacies
- Online Pharmacies
Frequently Asked Questions (FAQ)
Q. How are regulatory frameworks influencing innovation in the UK peptide and anticoagulant drugs market?
A. Regulatory bodies such as the Medicines and Healthcare Products Regulatory Agency and National Institute for Health and Care Excellence play a crucial role by ensuring safety, efficacy, and cost-effectiveness. Streamlined approval pathways and early scientific advice help pharmaceutical companies bring innovative peptide therapies and anticoagulants to market faster while maintaining strict quality standards.
Q. What role do hospitals play in the adoption of peptide and anticoagulant drugs in the UK?
A. Hospitals, particularly under the National Health Service, are central to drug adoption due to centralized procurement systems and high patient inflow for cardiovascular treatments. They are the primary setting for administering injectable anticoagulants and advanced peptide therapies, ensuring consistent demand and structured distribution.
Q. What challenges are associated with the development of peptide-based therapeutics in the UK?
A. Peptide drug development faces challenges such as complex synthesis processes, stability issues, and high production costs. Despite advancements in biotechnology, maintaining bioavailability and ensuring efficient large-scale manufacturing remain key hurdles for companies operating in the UK pharmaceutical landscape.
Q. How is the shift toward personalized medicine impacting the anticoagulant drugs market in the UK?
A. Personalized medicine is transforming treatment approaches by enabling tailored anticoagulant therapies based on patient-specific risk factors, genetics, and clinical history. This trend improves treatment outcomes, minimizes adverse effects like bleeding risks, and encourages the adoption of next-generation therapies within the UK healthcare system.
Check Licence
Choose the plan that fits you best: Single User, Multi-User, or Enterprise solutions tailored for your needs.
We Have You Covered
- 24/7 Analyst Support
- Clients Across the Globe
- Tailored Insights
- Technology Tracking
- Competitive Intelligence
- Custom Research
- Syndicated Market Studies
- Market Overview
- Market Segmentation
- Growth Drivers
- Market Opportunities
- Regulatory Insights
- Innovation & Sustainability
Report Details
| Scope | Country |
| Pages | 245 |
| Delivery | PDF & Excel via Email |
| Language | English |
| Release | Apr 2026 |
| Access | Download from this page |
