United Kingdom Regulatory Information Management Systems Market
United Kingdom Regulatory Information Management Systems Market Size, Share, By Component (Software, Services), By Deployment (Cloud-Based, On-Premise), By End User (Pharmaceutical Companies, Biotechnology Firms, CROs), Analysis and Forecast 2026 ? 2035
Report Overview
Table of Contents
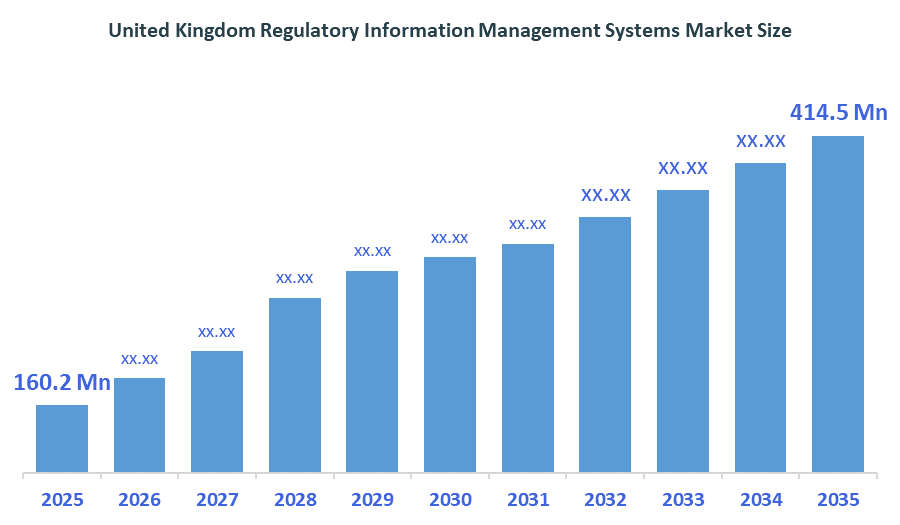
The United Kingdom Regulatory Information Management Systems Market size is forecast to grow from approximately USD 160.2 million in 2025 to around USD 414.5million by 2035. According to Decision Advisors, a detailed research report on the regulatory information management systems market indicates that the cloud-based RIMS platforms combined with the digital regulatory submission management trend dominates the market, accounting for approximately 42-46% of the total share in the United Kingdom. Veeva Systems Inc. leads the market with an estimated 17-20% United Kingdom market share. The company's annual revenue of approximately USD 2.75 billion is making it one of the most influential forces shaping industry trends and overall market growth.
Market Snapshot
- United Kingdom Regulatory Information Management Systems Market size (2025): USD 160.2 Million
- Projected United Kingdom Regulatory Information Management Systems Market Size (2035): USD 414.5Million
- United Kingdom Regulatory Information Management Systems Market Compound Annual Growth Rate (CAGR): 9.97%
- Base Year: 2025
- Historical Period: 2021–2024
- Forecast Period: 2026–2035
Market Overview/Introduction
The United Kingdom Regulatory Information Management Systems Market is the use of digital platforms in the life sciences industry to handle regulatory processes, paperwork, and submissions. The systems speed up regulatory processes while making sure that they follow new rules. They also keep all of the data in one place during the product development process. The market experiences rapid expansion because clinical trials become more frequent, companies seek quicker product approvals, and regulatory requirements become more complex. Advanced technologies such as cloud computing, artificial intelligence, and automation improve system performance and operational speed. The market expands because government support for digital healthcare transformation, together with its strict regulatory enforcement, creates new business opportunities. These standards also help drive innovation.
- The Innovative Devices Access Pathway (IDAP) is a major UK government initiative designed to accelerate the development, regulatory approval, and NHS adoption of innovative medical technologies. It provides an integrated regulatory support framework involving agencies like MHRA, NICE, and NHS England. This scheme streamlines regulatory processes and improves compliance efficiency, directly supporting the adoption of advanced RIMS platforms.
- The NHS Health Technology Pathway Programme outlines a structured, end-to-end framework for bringing healthcare innovations from development to patient use. It includes regulatory submission steps, compliance checkpoints, and stakeholder coordination. This program supports organizations in navigating complex regulatory environments, thereby increasing reliance on centralized regulatory information management systems.
Notable Insights: -
- The software segment dominated the market in 2025 and holds the largest market share, accounting for approximately 62% during the forecast period.
- The cloud-based segment dominated the market in 2025 and holds the largest market share, accounting for approximately 68% during the forecast period.
- The pharmaceutical companies segment dominated the market in 2025 and holds the largest market share, accounting for approximately 44% during the forecast period.
- The compound annual growth rate of the United Kingdom Regulatory Information Management Systems Market is 9.97%.
- The market is likely to achieve a valuation of USD 8110.9Million by 2035.
What is the role of technology in grooming the market?
Technology plays a transformative role in shaping the United Kingdom regulatory information management systems market by enabling automation, real-time data access, and improved compliance management. Cloud-based RIMS platforms allow seamless collaboration across global regulatory teams and ensure centralized data storage. Artificial intelligence and analytics help in tracking regulatory changes, managing submissions, and reducing approval timelines. Integration with other enterprise systems enhances workflow efficiency and minimizes manual errors. Additionally, advanced cybersecurity measures ensure data protection and compliance with strict regulatory standards. These technological advancements are significantly improving operational efficiency, transparency, and decision-making across the regulatory landscape.
Market Drivers
The United Kingdom regulatory information management systems market experiences growth because the pharmaceutical and biotechnology industries face greater regulatory requirements and need better compliance management solutions. Companies are implementing advanced RIMS solutions because they need to meet increasing demands for rapid drug development and international market entry. The rising number of clinical trials, together with product developments, creates additional demand for efficient regulatory procedures. System performance and user acceptance of cloud computing and automation technologies have enhanced system capabilities through their development. The healthcare sector, together with regulatory bodies, implements digital transformation projects that enable broader use of regulatory information management systems.
Restrain
The United Kingdom Regulatory Information Management Systems Market faces restraints due to high implementation and maintenance costs associated with advanced software solutions. Integration challenges with legacy systems and data migration complexities can hinder adoption. Small and mid-sized organizations may face financial and technical barriers in deploying RIMS platforms. Lack of skilled professionals to manage these systems can further limit market growth.
Study on the Supply, Demand, Distribution, and Market Environment of the United Kingdom Regulatory Information Management Systems Market
The United Kingdom Regulatory Information Management Systems Market demonstrates a structured ecosystem driven by regulatory compliance and digital transformation. On the supply side, software providers and service vendors offer advanced RIMS platforms supported by continuous innovation and technological upgrades. Demand is fueled by pharmaceutical companies, biotechnology firms, and CROs seeking efficient regulatory processes and compliance solutions. Distribution channels include direct sales, cloud-based delivery models, and strategic partnerships. The market environment is influenced by stringent regulatory frameworks, increasing globalization of clinical trials, and the growing need for faster approvals, which collectively drive adoption while maintaining compliance standards.
Price Analysis and Consumer Behaviour Analysis
Price analysis in the United Kingdom regulatory information management systems market is influenced by software licensing costs, customization requirements, and integration expenses. Cloud-based solutions offer flexible pricing models, making them attractive for organizations of varying sizes. While initial investment costs can be high, long-term benefits such as improved efficiency and faster approvals justify spending. Consumer behavior is driven by organizational priorities such as compliance, scalability, and operational efficiency. Pharmaceutical and biotechnology companies increasingly invest in advanced RIMS solutions to reduce regulatory risks and enhance productivity. Growing awareness about digital compliance and regulatory efficiency is further encouraging adoption.
Market Segmentation
The United Kingdom Regulatory Information Management Systems Market Share is classified into component, application, and end user
- The software segment dominated the market in 2025 and holds the largest market share, accounting for approximately 62% during the forecast period.
Based on the component, the regulatory information management systems market is divided into software and services. Among these, the software segment dominated the market in 2025 and holds the largest market share, accounting for approximately 62% during the forecast period. The software segment is driven by its critical role in managing regulatory data, submissions, and compliance workflows efficiently. Its scalability and integration capabilities make it essential for organizations handling complex regulatory processes.
- The cloud-based segment dominated the market in 2025 and holds the largest market share, accounting for approximately 68% during the forecast period.
Based on the deployment, the regulatory information management systems market is divided into cloud-based, and on-premise. Among these, the cloud-based segment dominated the market in 2025 and holds the largest market share, accounting for approximately 68% during the forecast period. The cloud-based segment in the U.K. is attributed to as organizations increasingly prefer scalable, flexible, and cost-effective deployment models. Cloud solutions enable real-time collaboration, remote accessibility, and faster updates while reducing infrastructure costs. Their ability to support global regulatory operations and ensure data security makes them highly attractive across the life sciences industry.
- The pharmaceutical companies segment dominated the market in 2025 and holds the largest market share, accounting for approximately 44% during the forecast period.
Based on the end user, the regulatory information management systems market is divided into pharmaceutical companies, biotechnology firms, and CROs. Among these, the pharmaceutical companies segment dominated the market in 2025 and holds the largest market share, accounting for approximately 44% during the forecast period. The pharmaceutical companies segment in the U.K. is driven by their extensive involvement in drug development, clinical trials, and regulatory processes. High investment capacity and continuous need for efficient data and compliance management systems drive adoption. Additionally, increasing demand for faster drug approvals and global expansion further strengthen their leading market position during the forecast period.
Recent Development
- In March 2026, life sciences organizations increasingly adopted AI-driven regulatory information management systems to automate document management, streamline submission tracking, and enable real-time monitoring of regulatory changes. These advancements significantly improved compliance efficiency, reduced manual workload, and accelerated approval timelines across the United Kingdom regulatory ecosystem.
- In July 2025, the Medicines and Healthcare products Regulatory Agency introduced significant reforms to medical device regulations, focusing on streamlined approval pathways and improved submission processes for medical devices and diagnostics. These updates encouraged companies to adopt advanced regulatory information management systems to efficiently manage submissions and comply with evolving UK regulatory standards.
- In March 2025, Rimsys entered into a strategic alliance with KPMG to drive digital transformation in the MedTech sector. The partnership focused on improving regulatory information management by combining advanced RIMS software with advisory expertise, enabling companies to streamline regulatory processes and enhance compliance efficiency.
- In January 2024, the Medicines and Healthcare products Regulatory Agency launched the International Recognition Procedure (IRP), enabling the UK to recognize approvals from trusted global regulators and streamline regulatory submissions. This development required pharmaceutical and MedTech companies to upgrade regulatory information management systems to handle new pathways and accelerated approval processes.
Competitive Analysis
The report offers the appropriate analysis of the key organisations/companies involved within the United Kingdom Regulatory Information Management Systems Market, along with a comparative evaluation primarily based on their product offering, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborative analysis focusing on the current news and developments of the companies, which includes product development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
Top Key Companies in the United Kingdom Regulatory Information Management Systems Market
- Avery Dennison Corporation
- Zebra Technologies Corporation
- Impinj Inc.
- CCL Industries (Smartrac)
- HID Global Corporation
- Others
Key Target Audience
- Market Players
- Investors
- End-users
- Government Authorities
- Consulting and Research Firm
- Venture capitalists
- Value-Added Resellers (VARs)
Market Segment
This study forecasts revenue at the United Kingdom, regional, and country levels from 2020 to 2035. Spherical Insights has segmented the United Kingdom Regulatory Information Management Systems Market based on the below-mentioned segments
United Kingdom Regulatory Information Management Systems Market, By Component
- Software
- Services
United Kingdom Regulatory Information Management Systems Market, By Deployment
- Cloud-Based
- On-Premise
United Kingdom Regulatory Information Management Systems Market, By End User
- Pharmaceutical Companies
- Biotechnology Firms
- CROs
Frequently Asked Questions (FAQ)
Q. How are global regulatory requirements influencing the adoption of RIMS in the United Kingdom?
A. Global regulatory requirements are pushing UK-based pharmaceutical and biotechnology companies to adopt RIMS solutions to manage multi-country submissions efficiently. These systems help standardize documentation, ensure compliance with international guidelines, and reduce delays in approvals, enabling companies to expand their presence in global healthcare markets.
Q. What impact does automation have on regulatory operations in the UK RIMS market?
A. Automation significantly improves regulatory operations by reducing manual tasks, minimizing errors, and accelerating submission processes. In the UK RIMS market, automated workflows enable faster document preparation, validation, and tracking, helping organizations improve productivity while ensuring compliance with evolving regulatory standards and timelines.
Q. Why is data centralization important in regulatory information management systems?
A. Data centralization allows organizations to store all regulatory information in a single platform, improving accessibility, consistency, and transparency. In the UK market, centralized systems help regulatory teams quickly retrieve documents, track submission statuses, and ensure accurate reporting, which is essential for maintaining compliance and operational efficiency.
Q. How are small and mid-sized enterprises (SMEs) adapting to RIMS adoption in the UK?
A. SMEs in the United Kingdom are increasingly adopting cloud-based RIMS solutions due to their lower upfront costs and scalability. These systems allow smaller organizations to access advanced regulatory tools without heavy infrastructure investment, helping them compete effectively while maintaining compliance with stringent regulatory requirements.
Check Licence
Choose the plan that fits you best: Single User, Multi-User, or Enterprise solutions tailored for your needs.
We Have You Covered
- 24/7 Analyst Support
- Clients Across the Globe
- Tailored Insights
- Technology Tracking
- Competitive Intelligence
- Custom Research
- Syndicated Market Studies
- Market Overview
- Market Segmentation
- Growth Drivers
- Market Opportunities
- Regulatory Insights
- Innovation & Sustainability
Report Details
| Scope | Country |
| Pages | 210 |
| Delivery | PDF & Excel via Email |
| Language | English |
| Release | Apr 2026 |
| Access | Download from this page |
