United Kingdom Sustained Release Excipients Market
United Kingdom Sustained release excipients market Size, Share, By Product Type (Polymers, Lipid-Based Excipients, Coating Materials, Matrix Forming Agents), By Application (Tablets, Capsules, Injectable Formulations, Implants), By End User (Pharmaceutical Companies, Contract Manufacturing Organizations, Research Institutes), Analysis and Forecast 2026 ? 2035
Report Overview
Table of Contents
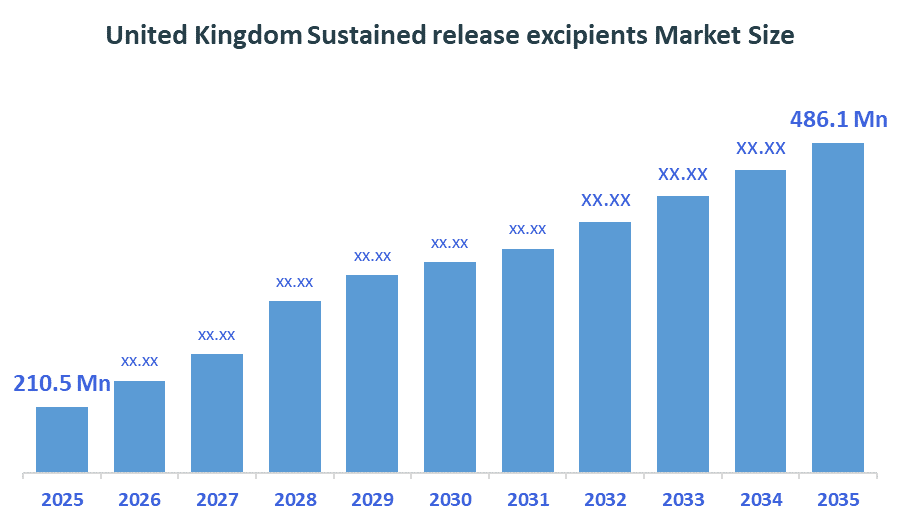
The United Kingdom Sustained Release Excipients Market size is forecast to grow from approximately USD 210.5million in 2025 to around USD 486.1million by 2035. According to Decision Advisors, a detailed research report on the sustained release excipients market indicates that the polymer-based sustained release excipients trend dominates the market, accounting for approximately 44-46% of the total share in the United Kingdom. BASF SE leads the market with an estimated 20-24% United Kingdom market share. The company's annual revenue of approximately USD 76-78 billion is making it one of the most influential forces shaping industry trends and overall market growth.
Market Snapshot
- United Kingdom Sustained release excipients market size (2025): USD 210.5Million
- Projected United Kingdom Sustained release excipients market Size (2035): USD 486.1Million
- United Kingdom Sustained release excipients market Compound Annual Growth Rate (CAGR): 8.73%
- Base Year: 2025
- Historical Period: 2021–2024
- Forecast Period: 2026–2035
Market Overview/Introduction
The United Kingdom Sustained Release Excipients Market comprises specialized pharmaceutical components that control active pharmaceutical ingredient (API) release rates across extended time periods in human body systems. These excipients serve essential functions in drug delivery systems, which maintain both sustained and controlled drug release because they enhance treatment results while decreasing the number of required doses. The common materials found in this category consist of cellulose derivatives, acrylic polymers, lipids, and wax-based systems. The market is expanding because patient-centric drug formulations have become more popular, and chronic diseases such as diabetes, cardiovascular disorders, and neurological conditions have become more common, and R&D investments in modified-release drug delivery technologies have increased. UK pharmaceutical companies are adopting advanced excipient engineering practices to enhance drug stability and performance.
- The UK’s SMMIP supports advanced pharmaceutical manufacturing, including sustained-release excipient development such as polymer-based matrices and coating technologies. It promotes greener, more efficient drug production and strengthens UK manufacturing capacity. The programme is funded under the VPAG framework and delivered through Innovate UK partnerships with industry and academia.
- Innovate UK provides grant funding for pharmaceutical R&D, including sustained-release formulations and functional excipients. It supports innovation in polymer science, drug delivery systems, and modified-release technologies through competitive funding calls. Collaboration between universities and pharma companies is encouraged to accelerate commercialization of advanced excipient technologies in the UK market.
- The MMIC is a UK government-backed initiative focused on transforming pharmaceutical manufacturing. It supports development of advanced excipients, continuous manufacturing, and controlled-release oral dosage systems. The centre enables collaboration between industry leaders like GSK and AstraZeneca with research institutions to improve formulation efficiency and scalability of sustained-release drug products.
Notable Insights: -
- The polymers segment dominated the market in 2025 and holds the largest market share, accounting for approximately 48% during the forecast period.
- The tablets segment dominated the market in 2025 and holds the largest market share, accounting for approximately 52% during the forecast period.
- The pharmaceutical companies segment dominated the market in 2025 and holds the largest market share, accounting for approximately 55% during the forecast period.
- The compound annual growth rate of the United Kingdom Sustained release excipients market is 8.73%.
- The market is likely to achieve a valuation of USD 8110.9Million by 2035.
What is the role of technology in grooming the market?
Technology plays a vital role in advancing the United Kingdom Sustained Release Excipients Market by enabling precision drug formulation, predictive release modeling, and advanced polymer engineering. Techniques such as hot-melt extrusion, spray drying, and nano-encapsulation improve controlled drug release performance and formulation stability. Computational modeling and AI-assisted formulation design help optimize excipient selection and predict drug release kinetics more accurately. Additionally, innovations in biocompatible polymers and smart excipients enhance targeted delivery and reduce side effects. Overall, technological advancements are significantly improving efficiency, product performance, and innovation in sustained release drug delivery systems.
Market Drivers
The United Kingdom Sustained Release Excipients Market is driven by more consumers need for long-acting drug formulations, which help them stay on medication while decreasing their need for multiple daily doses. The increasing cases of chronic diseases, that include hypertension, diabetes, and respiratory disorders, create a higher demand for controlled-release medications. Pharmaceutical companies are investing heavily in advanced drug delivery research to improve therapeutic outcomes. The market is experiencing growth because more people are using generic drugs, and contract manufacturing operations are becoming more common. The market experiences growth acceleration through ongoing advancements in polymer chemistry and excipient functionality.
Restrain
The United Kingdom Sustained Release Excipients Market faces restraints due to high development costs associated with advanced formulation technologies and complex regulatory approval processes. Limited compatibility of certain excipients with active pharmaceutical ingredients can restrict formulation flexibility. Smaller manufacturers often face difficulties in scaling production and meeting compliance requirements.
Study on the Supply, Demand, Distribution, and Market Environment of the United Kingdom Sustained release excipients market
The United Kingdom Sustained Release Excipients Market demonstrates a balanced ecosystem shaped by strong pharmaceutical R&D capabilities, regulatory oversight, and increasing demand for advanced drug delivery systems. On the supply side, global and regional excipient manufacturers provide a wide range of polymers, lipids, and coating agents, with polymer-based excipients holding the dominant share due to their versatility and controlled-release efficiency. On the demand side, pharmaceutical companies account for the majority usage as they increasingly develop sustained-release formulations for chronic disease management. Contract manufacturing organizations are also expanding their adoption due to rising outsourcing trends in drug development. Distribution channels primarily include direct supply agreements between excipient manufacturers and pharmaceutical firms, supported by specialized chemical distributors ensuring quality compliance and logistics efficiency. The overall market environment in the UK is favorable due to strong pharmaceutical innovation, government support for life sciences, and increasing investment in advanced formulation technologies, although cost pressures and regulatory complexity remain key challenges.
Price Analysis and Consumer Behaviour Analysis
The price structure of the United Kingdom Sustained Release Excipients Market is moderately high due to the specialized nature of materials and complex manufacturing processes. Advanced polymer-based excipients and functional coating systems typically command premium pricing due to their performance benefits and regulatory compliance requirements. Pricing is also influenced by batch consistency, purity levels, and application-specific customization. Consumer behavior in the UK pharmaceutical industry shows a strong preference for high-performance, regulatory-compliant, and scalable excipient solutions. Pharmaceutical companies prioritize reliability, formulation compatibility, and long-term supply stability over cost minimization. Increasing demand for controlled-release formulations in chronic disease therapies is further strengthening the adoption of premium excipient technologies.
Market Segmentation
The United Kingdom Sustained release excipients market Share is classified into type, source, and end user
- The polymers segment dominated the market in 2025 and holds the largest market share, accounting for approximately 48% during the forecast period.
Based on the product type, the sustained release excipients market is divided into polymers, lipid-based excipients, coating materials, and matrix-forming agents. Among these, the polymers segment dominated the market in 2025 and holds the largest market share, accounting for approximately 48% during the forecast period. The polymers segment is driven by their excellent controlled-release properties, high stability, and wide compatibility with active pharmaceutical ingredients. Their ability to modify drug release kinetics makes them highly preferred in sustained-release formulations across pharmaceutical manufacturing.
- The tablets segment dominated the market in 2025 and holds the largest market share, accounting for approximately 52% during the forecast period.
Based on the application, the sustained release excipients market is divided into tablets, capsules, injectable formulations, and implants. Among these, the tablets segment dominated the market in 2025 and holds the largest market share, accounting for approximately 52% during the forecast period. The tablets segment in the U.K. is attributed to their ease of production, cost efficiency, and high patient compliance. Sustained-release tablet formulations are widely used in chronic disease management, offering controlled drug release and improved therapeutic outcomes.
- The pharmaceutical companies segment dominated the market in 2025 and holds the largest market share, accounting for approximately 55% during the forecast period.
Based on the end user, the sustained release excipients market is divided into pharmaceutical companies, contract manufacturing organizations, and research institutes. Among these, the pharmaceutical companies segment dominated the market in 2025 and holds the largest market share, accounting for approximately 55% during the forecast period. The pharmaceutical companies segment in the U.K. is driven by their strong R&D capabilities and high adoption of advanced formulation technologies. Increasing investment in controlled-release drug development further strengthens their dominance in the market.
Recent Development
- In September 2025, a UK university spin-off, LipoActive, launched a novel liposomal encapsulation technology designed to enhance bioavailability and enable cleaner excipient use in nutraceutical and pharmaceutical formulations. The innovation uses clean-label phospholipid-based systems to replace restricted excipients like silicon dioxide, making it suitable for global markets including APAC. This development supports growth in the sustained-release and advanced excipients segment by improving ingredient stability, absorption efficiency, and formulation flexibility for next-generation drug delivery systems.
- In April 2025, several multinational pharmaceutical companies adjusted global R&D strategies while continuing collaborations in the United Kingdom with firms specializing in advanced excipients for complex drug formulations. These partnerships supported sustained-release and controlled-release drug development, reinforcing the UK’s role in innovative pharmaceutical research despite shifting global R&D priorities.
- In March 2025, major international pharmaceutical companies, including Pfizer, Novartis, and Eisai, maintained strong R&D partnerships in the United Kingdom, focusing on advanced drug formulation and delivery technologies. These collaborations supported innovation in sustained-release systems and excipient development, strengthening the UK’s pharmaceutical research ecosystem and controlled-release formulation capabilities.
Competitive Analysis
The report offers the appropriate analysis of the key organisations/companies involved within the United Kingdom Sustained Release Excipients Market, along with a comparative evaluation primarily based on their product offering, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborative analysis focusing on the current news and developments of the companies, which includes product development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
Top Key Companies in the United Kingdom Sustained Release Excipients Market
- BASF SE
- Dow Chemical Company
- Evonik Industries AG
- Ashland Global Holdings Inc.
- Roquette Frères
- Others
Key Target Audience
- Market Players
- Investors
- End-users
- Government Authorities
- Consulting and Research Firm
- Venture capitalists
- Value-Added Resellers (VARs)
Market Segment
This study forecasts revenue at the United Kingdom, regional, and country levels from 2020 to 2035. Spherical Insights has segmented the United Kingdom sustained release excipients market based on the below-mentioned segments
United Kingdom Sustained Release Excipients Market, By Product Type
- Polymers
- Lipid-Based Excipients
- Coating Materials
- Matrix Forming Agents
United Kingdom Sustained Release Excipients Market, By Application
- Tablets
- Capsules
- Injectable Formulations
- Implants
United Kingdom Sustained Release Excipients Market, By End User
- Pharmaceutical Companies
- Contract Manufacturing Organizations
- Research Institutes
Frequently Asked Questions (FAQ)
Q. What are the emerging technologies transforming the UK Sustained Release Excipients Market?
A. Emerging technologies such as nanotechnology-based drug delivery, hot-melt extrusion, 3D printing of pharmaceuticals, and AI-assisted formulation design are transforming the market by improving drug release precision, stability, and patient-specific customization. These innovations enhance therapeutic efficiency and accelerate development timelines for sustained-release formulations.
Q. How does regulatory framework influence the market in the UK?
A. The UK Medicines and Healthcare Products Regulatory Agency (MHRA) plays a key role in ensuring safety, efficacy, and quality of excipients used in drug formulations. Strict compliance requirements encourage manufacturers to invest in high-quality, well-characterized excipients, driving innovation while ensuring patient safety.
Q. What challenges do companies face in this market?
A. Companies face challenges such as high R&D costs, complex formulation development, strict regulatory approvals, and compatibility issues between excipients and active pharmaceutical ingredients. Additionally, scalability and long development cycles pose significant barriers for smaller manufacturers.
Q. How is sustainability influencing the market?
A. Sustainability is increasingly important, with companies focusing on biodegradable polymers, green chemistry-based excipients, and eco-friendly manufacturing processes. These initiatives help reduce environmental impact while aligning with global ESG goals and improving long-term industry sustainability.
Check Licence
Choose the plan that fits you best: Single User, Multi-User, or Enterprise solutions tailored for your needs.
We Have You Covered
- 24/7 Analyst Support
- Clients Across the Globe
- Tailored Insights
- Technology Tracking
- Competitive Intelligence
- Custom Research
- Syndicated Market Studies
- Market Overview
- Market Segmentation
- Growth Drivers
- Market Opportunities
- Regulatory Insights
- Innovation & Sustainability
Report Details
| Scope | Country |
| Pages | 210 |
| Delivery | PDF & Excel via Email |
| Language | English |
| Release | Apr 2026 |
| Access | Download from this page |
