United States Immunosuppressants Market
United States Immunosuppressants Market Size, Share, By Product Type (Calcineurin Inhibitors, Corticosteroids, Monoclonal Antibodies (mAbs), mTOR Inhibitors), By Indication (Organ Transplantation, Autoimmune Diseases, Non-Autoimmune Inflammatory Diseases), and By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), Analysis and Forecast 2025-2035
Report Overview
Table of Contents
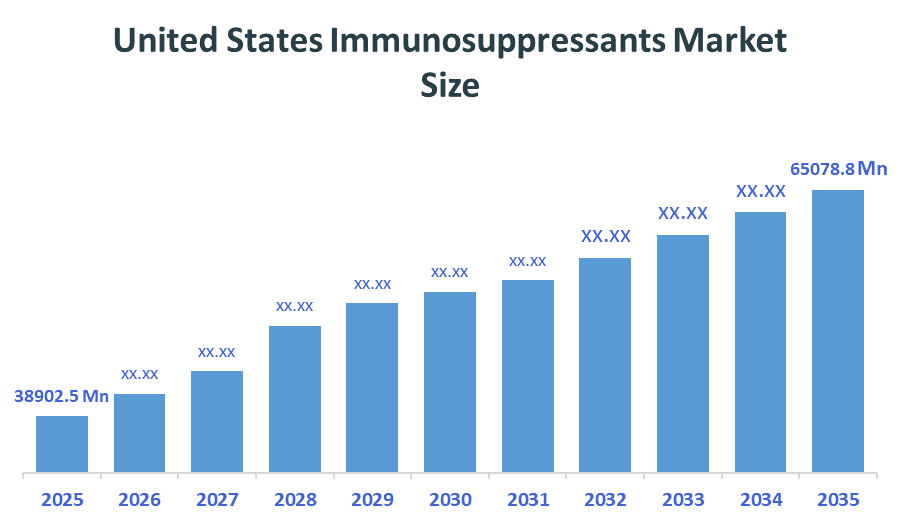
The United States Immunosuppressants Market size is forecast to grow from approximately USD 38902.5 Million in 2025 to around USD 65078.8 MIllion by 2035. According to Decision Advisors, a detailed research report on the immunosuppressants market indicates that the rapid shift toward monoclonal antibodies and biologic immunosuppressants trend dominates the market, accounting for approximately 35-45% of the total share in the United States. AbbVie Inc. leads the market with an estimated 18-22% United States market share. The company's annual revenue of approximately USD 8.6 billion is making it one of the most influential forces shaping industry trends and overall market growth.
Market Snapshot
- United States Immunosuppressants Market Size (2025): USD 38902.5 Million
Projected United States Immunosuppressants Market Size (2035): USD 65078.8 MIllion - United States Immunosuppressants Market Compound Annual Growth Rate (CAGR): 5.28%
- Base Year: 2025
- Historical Period: 2021–2025
- Forecast Period: 2026–2035
Market Overview/Introduction
The United States immunosuppressants market refers to the production and distribution of medicinal products, which can suppress the immune system to prevent rejection or treat autoimmune diseases. These medicines are a group of drugs that aid in treating Rheumatoid Arthritis and are used as part of transplant procedures. An increasing number of transplants, increasing prevalence of autoimmune diseases, along with growing R&D in biological medicines, are the key drivers of this market. Growth trends of the future market could be personalizing therapy in treatment, cell-based therapies, and improved ways of delivering drugs. The future success of the market would depend on continuing investments in R&D as well as the easy availability of health care services.
- The United States immunosuppressants market is the Medicare Part B Immunosuppressive Drug Benefit. Introduced in 2023, it provides continuous coverage for transplant patients’ immunosuppressive drugs, even after standard eligibility ends, reducing financial burden and improving long-term transplant success rates.
- The expanded lifetime immunosuppressive drug coverage under Medicare for kidney transplant recipients. This policy ensures lifelong access to essential anti-rejection medications for eligible patients, preventing graft failure and increasing treatment adherence, thereby supporting sustained demand and stability in the immunosuppressants market.
Notable Insights: -
- The calcineurin inhibitors segment dominated the market in 2025 and is projected to grow at a substantial CAGR of approximately 10.5% during the forecast period.
- The organ transplantation segment accounted for the largest share in 2025 and is anticipated to grow at a significant CAGR of approximately 9.5 % during the forecast period.
- The online pharmacies segment dominated the market in 2025 and is projected to grow at a substantial CAGR of approximately 12.4% during the forecast period.
- The compound annual growth rate of the United Kingdom Immunosuppressants Market is 5.28%.
- The market is likely to achieve a valuation of USD 65078.8 Million by 2035.
What is the role of technology in grooming the market?
Technology plays a crucial role in shaping the United States immunosuppressants market by enabling the development of advanced therapies, including targeted biologics and personalized medicine approaches. Innovations in Genomics and Biotechnology allow for precise identification of immune pathways, improving drug efficacy and reducing adverse effects. Digital health tools and AI-driven analytics support better patient monitoring, dosage optimization, and adherence tracking. Additionally, advancements in drug delivery systems enhance bioavailability and convenience. Technology also accelerates clinical trials and regulatory processes, enabling faster market entry of novel immunosuppressive therapies, ultimately improving patient outcomes and expanding treatment accessibility across the country.
Market Drivers
The United States Immunosuppressants Market is driven by growing occurrences of autoimmune disorders such as Rheumatoid Arthritis and Lupus, and the number of organ transplant surgeries performed in the US increases with each year. Increasing use of cutting-edge biologics and monoclonal antibodies will improve success of therapy and compliance with therapy. The US Immunosuppressants Market will be stimulated by increasing reimbursement scenarios and developed healthcare systems, and constant R & D efforts from major biopharmaceutical players, which would enable innovation in the field. The demographic profile of the US, where the aging population is on the rise, will further drive the demand for immunosuppressants, since most people develop long-lasting diseases for which immunosuppressants are used.
Restrain
The United States immunosuppressants market faces restraints due to rising organ transplant procedures and autoimmune disease prevalence. Advances in biologics, strong healthcare infrastructure, and increasing adoption of targeted therapies support growth. However, high drug costs and side effects remain challenges, while ongoing research continues to expand treatment options and improve patient outcomes.
Study on the Supply, Demand, Distribution, and Market Environment of the United States Immunosuppressants Market
The United States immunosuppressants market is characterized by a strong and complex supply-demand ecosystem supported by advanced healthcare infrastructure. On the supply side, production depends heavily on global active pharmaceutical ingredient sourcing, particularly from Asia, making the supply chain vulnerable to disruptions and regulatory issues. Demand is consistently high due to rising organ transplant procedures, increasing autoimmune disease prevalence, and an aging population requiring long-term therapy. Distribution is dominated by hospital pharmacies, which play a central role in transplant and acute care management, while retail and online channels support chronic treatment access. The market environment is highly regulated and innovation-driven, with growing adoption of biologics, biosimilars, and value-based healthcare models shaping competitive dynamics and accessibility.
Price Analysis and Consumer Behaviour Analysis
The price analysis of the United States immunosuppressants market shows a high-cost structure driven by branded biologics, complex manufacturing, and strong R&D investments. Drug prices remain elevated, especially for advanced therapies, although the increasing entry of generics and biosimilars is gradually improving affordability and intensifying competition. U.S. Food and Drug Administration approvals of innovative therapies further influence premium pricing. From a consumer behavior perspective, demand is largely necessity-driven, with patients relying on these drugs for organ transplants and autoimmune diseases. Purchasing decisions are heavily influenced by insurance coverage, reimbursement policies, and physician prescriptions. Additionally, there is a growing shift toward cost-effective generics and biologics with better efficacy and fewer side effects, reflecting value-based healthcare trends.
Market Segmentation
The United States Immunosuppressants Market share is classified into product type, indication, and distribution channel
- The calcineurin inhibitors segment dominated the market in 2025 and is projected to grow at a substantial CAGR of approximately 10.5% during the forecast period.
Based on the product type, the excipients market is divided into calcineurin inhibitors, corticosteroids, monoclonal antibodies (mAbs), and mTOR inhibitors. Among these, the calcineurin inhibitors segment dominated the market in 2025 and is projected to grow at a substantial CAGR of approximately 10.5% during the forecast period. The calcineurin inhibitors segment is driven by increasing adoption in targeted therapy for autoimmune diseases and transplant rejection management. These biologics offer higher specificity, fewer systemic side effects, and improved patient outcomes, leading to strong physician preference and continuous pipeline innovation.
- The organ transplantation segment accounted for the largest share in 2025 and is anticipated to grow at a significant CAGR of approximately 9.5 % during the forecast period.
Based on the indication, the excipients market is divided into organ transplantation, autoimmune diseases, and non-autoimmune inflammatory diseases. Among these, the organ transplantation segment accounted for the largest share in 2025 and is anticipated to grow at a significant CAGR of approximately 9.5 % during the forecast period. The organ transplantation segment leads the market due to the rising prevalence of chronic conditions such as rheumatoid arthritis and multiple sclerosis. Growing awareness, early diagnosis, and long-term treatment needs boost demand. Additionally, increasing use of advanced biologics and personalized medicine approaches further accelerates growth in this segment.
- The online pharmacies segment dominated the market in 2025 and is projected to grow at a substantial CAGR of approximately 12.4% during the forecast period.
Based on the distribution channel, the excipients market is divided into hospital pharmacies, retail pharmacies, and online pharmacies. Among these, the online pharmacies segment dominated the market in 2025, and is projected to grow at a substantial CAGR of approximately 8.5% during the forecast period. The online pharmacies segment dominates due to increasing digital adoption, convenience of home delivery, and improved access to specialty drugs. Patients managing chronic autoimmune conditions prefer online refills, while telehealth integration and cost discounts further strengthen this channel’s expansion.
Recent Development
- In October 2025, NATCO Pharma, in partnership with Breckenridge Pharmaceutical, launched a generic version of Everolimus (1 mg) in the United States. The product, a generic equivalent of Novartis’ Zortress, was introduced as an mTOR inhibitor immunosuppressant used to prevent organ rejection in kidney and liver transplant patients.
- In April 2025, Amgen announced that the U.S. Food and Drug Administration (FDA) approved UPLIZNA (inebilizumab-cdon) as the first and only treatment for Immunoglobulin G4-related disease (IgG4-RD). The therapy, a CD19-targeting monoclonal antibody, marked a significant advancement in immunosuppressive treatment by addressing a rare, chronic immune-mediated condition with no prior approved therapies.
- In February 2025, Teva Pharmaceuticals in partnership with Alvotech launched SELARSDI (ustekinumab-aekn) in the United States as a biosimilar to Stelara. The product was introduced as a lower-cost alternative for treating conditions such as Crohn’s disease and plaque psoriasis, expanding patient access to immunosuppressive therapies.
Competitive Analysis
The report offers the appropriate analysis of the key organisations/companies involved within the United States Immunosuppressants Market, along with a comparative evaluation primarily based on their product offering, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborative analysis focusing on the current news and developments of the companies, which includes product development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
Top Key Companies in the United States Immunosuppressants Market
- AbbVie
- Johnson & Johnson
- Pfizer
- Novartis
- Roche
- Others
Key Target Audience
- Market Players
- Investors
- End-users
- Government Authorities
- Consulting and Research Firm
- Venture capitalists
- Value-Added Resellers (VARs)
Market Segment
This study forecasts revenue at the United Kingdom, regional, and country levels from 2020 to 2035. Decision Advisor has segmented the United States Immunosuppressants market based on the below-mentioned segments
United States Immunosuppressants Market, By Product Type
- Calcineurin Inhibitors
- Corticosteroids
- Monoclonal Antibodies (mAbs)
United States Immunosuppressants Market, By Indication
- Organ Transplantation
- Autoimmune Diseases
- Non-Autoimmune Inflammatory Diseases
United States Immunosuppressants Market, By Distribution Channel
- Hospital Pharmacies
- Retail Pharmacies
- Online Pharmacies
Frequently Asked Questions (FAQ)
Q. How is the shift toward biologics influencing the United States immunosuppressants market?
A. The increasing adoption of biologics is transforming the market by offering more targeted and effective treatment options with fewer systemic side effects. These therapies are particularly beneficial for complex autoimmune conditions and transplant patients, leading to improved clinical outcomes. Their growing preference among healthcare providers is also encouraging pharmaceutical companies to invest heavily in biologics research and pipeline expansion.
Q. What impact does healthcare policy have on the growth of the immunosuppressants market in the United States?
A. Favorable healthcare policies, including extended drug coverage and reimbursement programs, play a critical role in improving patient access to immunosuppressants. These policies reduce financial barriers, increase treatment adherence, and support long-term therapy usage, thereby contributing to consistent market demand and overall growth.
Q. Why is patient adherence a critical factor in this market?
A. Patient adherence is essential because immunosuppressants require strict and long-term usage to prevent organ rejection or manage chronic autoimmune diseases. Non-adherence can lead to severe complications, increasing healthcare costs and negatively affecting treatment outcomes. As a result, companies are focusing on user-friendly drug delivery systems and digital monitoring tools to enhance adherence.
Q. What opportunities exist for new entrants in the United States immunosuppressants market?
A. New entrants can explore opportunities in developing biosimilars, innovative drug delivery technologies, and personalized medicine solutions. Additionally, expanding into underserved patient populations and leveraging digital healthcare platforms can provide a competitive advantage, especially as demand for cost-effective and accessible therapies continues to rise.
Check Licence
Choose the plan that fits you best: Single User, Multi-User, or Enterprise solutions tailored for your needs.
We Have You Covered
- 24/7 Analyst Support
- Clients Across the Globe
- Tailored Insights
- Technology Tracking
- Competitive Intelligence
- Custom Research
- Syndicated Market Studies
- Market Overview
- Market Segmentation
- Growth Drivers
- Market Opportunities
- Regulatory Insights
- Innovation & Sustainability
Report Details
| Scope | country |
| Pages | 240 |
| Delivery | PDF & Excel via Email |
| Language | English |
| Release | Apr 2026 |
| Access | Download from this page |
