United States Monoclonal Antibodies Market
United States Monoclonal Antibodies Market size, Share, By Source (Murine, Chimeric, Human, and Humanized), By Application (Cancer, Infectious Diseases, Auto-Immune Diseases, and Inflammatory Diseases), By Route of Administration (Intravenous (IV), and Subcutaneous (SC)) and By End-User (Hospital Pharmacies, Specialty Clinics, Retail Pharmacies, and Online Pharmacies) Analysis and Forecast 2026 ? 2035.
Report Overview
Table of Contents
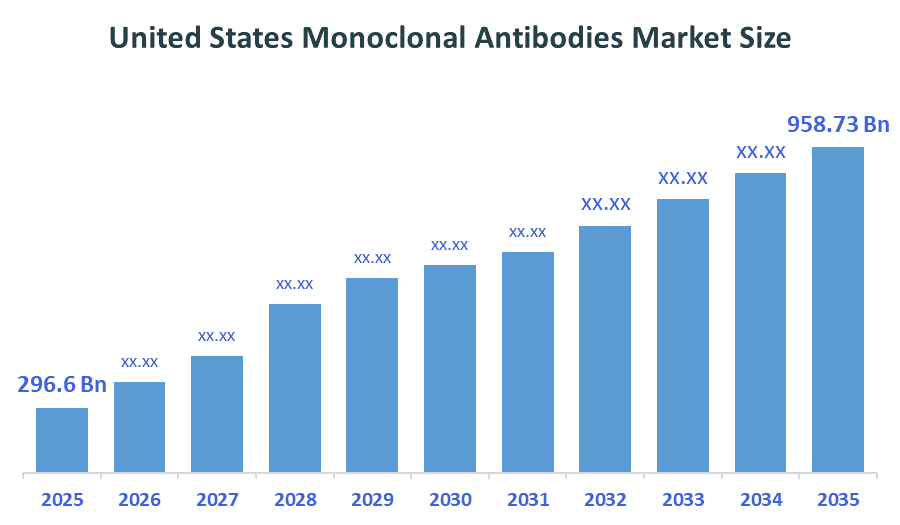
The United States Hip Replacement Devices Market size is forecast to grow from approximately USD 296.6 Billion in 2025 to around USD 958.73 Billion by 2035. According to Decision Advisors, a detailed research report on the anti-rheumatics market indicates that the Oncology mAbs trend dominates the market, accounting for approximately 55% of the total share in the United States. F. Hoffmann-La Roche leads the market with an estimated 13% United States market share. The company's annual revenue of approximately USD 74 billion is making it one of the most influential forces shaping industry trends and overall market growth.
Market Snapshot
- United States Monoclonal Antibodies Market Size (2025): USD 296.6Billion
- Projected United States Monoclonal Antibodies Market Size (2035): USD 958.73Billion
- United States Monoclonal Antibodies Market Compound Annual Growth Rate (CAGR): 12.45%
- Base Year: 2025
- Historical Period: 2021–2024
- Forecast Period: 2026–2035
Market Overview/Introduction
The United States Monoclonal Antibodies Market refers to all stages of development for antibody-based biotherapeutic medicines, which treat cancer, autoimmune diseases, and infectious diseases. Monoclonal antibodies are laboratory-produced molecules that scientists design to bind with particular antigens through their superior binding abilities, which enhances the effectiveness of medical treatments. The market benefits from the established healthcare system, which operates through the National Health Service. The market expansion will occur through the development of biosimilars and personalized medicine and the introduction of new antibody engineering methods, which encompass bispecific antibodies and antibody-drug conjugates. The market experiences growth because of rising disease rates, ongoing product development, and increasing regulatory approvals from organizations such as the Medicines and Healthcare products Regulatory Agency. The United States market experiences faster growth because of increased research funding and the industry transition toward targeted biologic treatments instead of standard medical approaches.
- The U.S. Food and Drug Administration (FDA) approved 46 novel drugs, including a significant share of biologics such as monoclonal antibodies across oncology and immunology. This program accelerates innovation and ensures faster patient access, strengthening growth in the U.S. monoclonal antibodies market.
- In December 2025, the FDA released draft guidance to streamline nonclinical safety studies for monoclonal antibodies, reducing regulatory complexity. The initiative aims to modernize development pathways, lower costs, and speed approvals, encouraging biotech investment and innovation in antibody therapeutics across the United States.
Notable Insights: -
- The humanized segment dominated the market in 2025 and holds the largest market share, accounting for approximately 50% during the forecast period.
- The cancer segment dominated the market in 2025 and holds the largest market share, accounting for approximately 45% during the forecast period.
- The intravenous (IV) segment dominated the market in 2025 and holds the largest market share, accounting for approximately 70% during the forecast period.
- The hospital pharmacies segment dominated the market in 2025 and holds the largest market share, accounting for approximately 55% during the forecast period.
- The compound annual growth rate of the United States Monoclonal Antibodies Market is 12.45%.
- The market is likely to achieve a valuation of USD 958.73Billion by 2035.
What is the role of technology in grooming the market?
Technology plays a pivotal role in shaping the United States monoclonal antibodies market by enhancing drug discovery, development, and manufacturing efficiency. Advanced platforms such as recombinant DNA technology, single-cell sequencing, and AI-driven drug design accelerate the identification of high-affinity antibodies. Automation and bioprocess optimization improve production scalability and reduce costs, supporting supply for the National Health Service. Innovations in continuous manufacturing and bioreactor systems ensure consistent quality and faster turnaround times. Additionally, digital health tools and data analytics enable better patient selection and personalized therapies, improving treatment outcomes. The adoption of biosimilar development technologies further drives affordability and competition, making advanced monoclonal antibody therapies more accessible across the UK healthcare landscape.
Market Drivers
The United States Monoclonal Antibodies Market has grown because more people are getting cancer, autoimmune diseases, and infectious diseases. This has led to a need for more advanced targeted therapies. The Medicines and Healthcare Products Regulatory Agency and the U.S. Food and Drug Administration have both given their OK to new monoclonal antibody treatments. This made more people use these treatments, especially for cancer. The market grew because it was easier for patients to get treatment with inexpensive biosimilar monoclonal antibodies. Better treatment outcomes and more accurate results came from focused research in genomics, antibody engineering, and the creation of personalized medicine. The UK market grew because advanced biologics became more popular than small-molecule drugs, which are more common. This made drug use safer, treatment outcomes better, and more effective.
Restrain
The United States monoclonal antibodies market faces restraints due to high treatment costs, limiting patient access despite support from the National Health Service. Stringent regulatory approvals, complex manufacturing processes, and supply chain dependencies also hinder rapid product availability and market expansion, particularly for innovative biologics and new entrants.
Study on the Supply, Demand, Distribution, and Market Environment of the United States Monoclonal Antibodies Market
The United States Monoclonal Antibodies Market is characterized by strong demand driven by the rising prevalence of cancer and autoimmune diseases, with procurement largely managed by the National Health Service. Supply is supported by global biopharma manufacturers and domestic production, though it remains sensitive to complex biologics manufacturing and regulatory approvals. Distribution is primarily hospital-based, supported by specialty pharmacies and cold-chain logistics. The market environment is shaped by stringent regulations and increasing biosimilar competition to control costs. The raw material industry, including cell culture media, bioreactors, and reagents, plays a crucial role, with suppliers ensuring high-quality inputs for consistent antibody production. Any disruption in these materials can significantly impact supply stability and production timelines.
Price Analysis and Consumer Behaviour Analysis
The United States Monoclonal Antibodies Market exhibits high pricing due to complex biologics manufacturing, stringent regulatory standards, and strong R&D investments. Prices are often moderated through centralized procurement by the National Health Service, which negotiates discounts and promotes biosimilar adoption to control costs. Consumer behavior is largely physician-driven, with demand influenced by clinical efficacy, safety profiles, and treatment guidelines rather than direct patient choice. There is an increasing preference for cost-effective biosimilars and subcutaneous formulations that reduce hospital visits. Additionally, rising awareness of targeted therapies and improved access to specialty care are boosting adoption. However, affordability pressures and reimbursement policies continue to shape purchasing decisions and overall market penetration across different patient segments in the United States.
Market Segmentation
The United States Monoclonal Antibodies Market share is classified into source, application, route of administration, and end-user
- The humanized segment dominated the market in 2025 and holds the largest market share, accounting for approximately 50% during the forecast period.
Based on the source, the monoclonal antibodies market is divided into murine, chimeric, human, and humanized. Among these, the humanized segment dominated the market in 2025 and holds the largest market share, accounting for approximately 50% during the forecast period. The humanized segment is driven by its reduced immunogenicity and improved safety compared to murine or chimeric antibodies. These antibodies offer higher efficacy and better patient tolerance, leading to wider adoption in treatments approved by the Medicines and Healthcare products Regulatory Agency, particularly in oncology and autoimmune diseases.
- The cancer segment dominated the market in 2025 and holds the largest market share, accounting for approximately 45% during the forecast period.
Based on the application, the monoclonal antibodies market is divided into cancer, infectious diseases, auto-immune diseases, and inflammatory diseases. Among these, the cancer segment dominated the market in 2025 and holds the largest market share, accounting for approximately 45% during the forecast period. The cancer segment in the U.S. is driven by high demand for oral solid dosage forms due to the high and rising prevalence of oncology cases, driving demand for targeted therapies. Strong clinical adoption, continuous approvals by the Medicines and Healthcare products Regulatory Agency, and proven efficacy of mAbs in improving survival rates further supported this segment’s leading market share.
- The intravenous (IV) segment dominated the market in 2025 and holds the largest market share, accounting for approximately 70% during the forecast period.
Based on the route of administration, the monoclonal antibodies market is divided into intravenous (IV), and subcutaneous (SC). Among these, the intravenous (IV) segment dominated the market in 2025 and holds the largest market share, accounting for approximately 70% during the forecast period. The intravenous (IV) segment in the U.S. is driven by its established use in hospital settings for delivering precise and controlled dosages. IV administration ensures rapid therapeutic action and is widely preferred for oncology and severe conditions, supported by treatment protocols under the National Health Service.
- The hospital pharmacies segment dominated the market in 2025 and holds the largest market share, accounting for approximately 55% during the forecast period.
Based on the end-user, the monoclonal antibodies market is divided into hospital pharmacies, specialty clinics, retail pharmacies, and online pharmacies. Among these, the hospital pharmacies segment dominated the market in 2025 and holds the largest market share, accounting for approximately 55% during the forecast period. The hospital pharmacies segment in the U.S. is driven by the high reliance on hospital-based administration of biologics, particularly intravenous therapies. Centralized procurement and distribution through the National Health Service, along with specialized handling and storage requirements, further strengthened the dominance of hospital pharmacies.
Recent Development
- In April 2026, AAX Biotech and evitria AG announced a partnership to integrate Opti-mAb technology into early-stage antibody engineering workflows. The collaboration aimed to enhance stability and developability of antibody candidates, supporting advanced biologics innovation and strengthening technological progress in the United States Monoclonal Antibodies Market.
- In October 2023, Daiichi Sankyo and Merck announced a global collaboration to co-develop three ADC candidates targeting cancer treatment. By 2025, the partnership had advanced multiple clinical trials across oncology indications, reinforcing innovation in monoclonal antibody-based therapies and strengthening the United States Monoclonal Antibodies Market.
Competitive Analysis
The report offers the appropriate analysis of the key organisations/companies involved within the United States monoclonal antibodies market, along with a comparative evaluation primarily based on their product offering, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborative analysis focusing on the current news and developments of the companies, which includes product development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
Top Key Companies in the United States Monoclonal Antibodies Market
- F. Hoffmann-La Roche Ltd.
- AbbVie Inc.
- Johnson & Johnson Services, Inc.
- Merck & Co., Inc.
- Bristol-Myers Squibb Company
- Others
Key Target Audience
- Market Players
- Investors
- End-users
- Government Authorities
- Consulting and Research Firm
- Venture capitalists
- Value-Added Resellers (VARs)
Market Segment
This study forecasts revenue at the United States, regional, and country levels from 2020 to 2035. Decision Advisor has segmented the United States Monoclonal Antibodies Market based on the below-mentioned segments
United States Monoclonal Antibodies Market, By Source
- Murine
- Chimeric
- Human
- Humanized
United States Monoclonal Antibodies Market, By Application
- Cancer
- Infectious Diseases
- Auto-Immune Diseases
- Inflammatory Diseases
United States Monoclonal Antibodies Market, By Route of Administration
- Intravenous (IV)
- Subcutaneous (SC)
United States Monoclonal Antibodies Market, By End-User
- Hospital Pharmacies
- Specialty Clinics
- Retail Pharmacies
- Online Pharmacies
United States Monoclonal Antibodies Market, By End-User
- Hospitals
- Clinics
- Home Healthcare
Frequently Asked Questions (FAQ)
Q. What role do biosimilars play in the United States monoclonal antibodies market?
A. Biosimilars play a critical role in reducing treatment costs and increasing patient access. They introduce competition to branded biologics, enabling the National Health Service to optimize spending while maintaining treatment quality and expanding therapy availability.
Q. How does regulatory support influence market growth in the US?
A. Strong regulatory frameworks led by the Medicines and Healthcare products Regulatory Agency ensure safety and efficacy while accelerating approvals, encouraging innovation, and supporting the rapid introduction of advanced monoclonal antibody therapies.
Q. Why is the US considered a favourable market for monoclonal antibody research?
A. The US offers a robust research ecosystem, strong academic-industry collaboration, and government funding initiatives, making it a hub for clinical trials and antibody innovation, particularly in oncology and immunology.
Q. What challenges do manufacturers face in the US monoclonal antibodies market?
A. Manufacturers face challenges such as high production costs, complex biologics manufacturing processes, strict regulatory compliance, and pricing pressures from centralized healthcare systems, which can impact profitability and market entry strategies.
Check Licence
Choose the plan that fits you best: Single User, Multi-User, or Enterprise solutions tailored for your needs.
We Have You Covered
- 24/7 Analyst Support
- Clients Across the Globe
- Tailored Insights
- Technology Tracking
- Competitive Intelligence
- Custom Research
- Syndicated Market Studies
- Market Overview
- Market Segmentation
- Growth Drivers
- Market Opportunities
- Regulatory Insights
- Innovation & Sustainability
Report Details
| Scope | Country |
| Pages | 210 |
| Delivery | PDF & Excel via Email |
| Language | English |
| Release | Apr 2026 |
| Access | Download from this page |
