United States Pharmaceutical Filtration Market
United States Pharmaceutical Filtration Market Size, Share, By Product Type (Membrane Filters, Prefilters & Depth Media, Single-Use Systems, Cartridge & Capsule Filters, Filter Holders, Filtration Accessories), By Technique (Microfiltration, Ultrafiltration, Nanofiltration, Cross-Flow Filtration, Reverse Osmosis), By End User (Pharmaceutical Companies, Biopharmaceutical Companies, Hospitals & Clinics, Research Institutes, Contract Manufacturing Organizations (CMOs)) Analysis and Forecast 2026-2035.
Report Overview
Table of Contents
Market Snapshot
- United States Pharmaceutical Filtration Market size (2025): USD 6.70Billion
- Projected United States Pharmaceutical Filtration Market Size (2035): USD 11.85 Billion
- United States Pharmaceutical Filtration Market Compound Annual Growth Rate (CAGR): 5.87%
- Base Year: 2025
- Historical Period: 2021–2024
- Forecast Period: 2026–2035
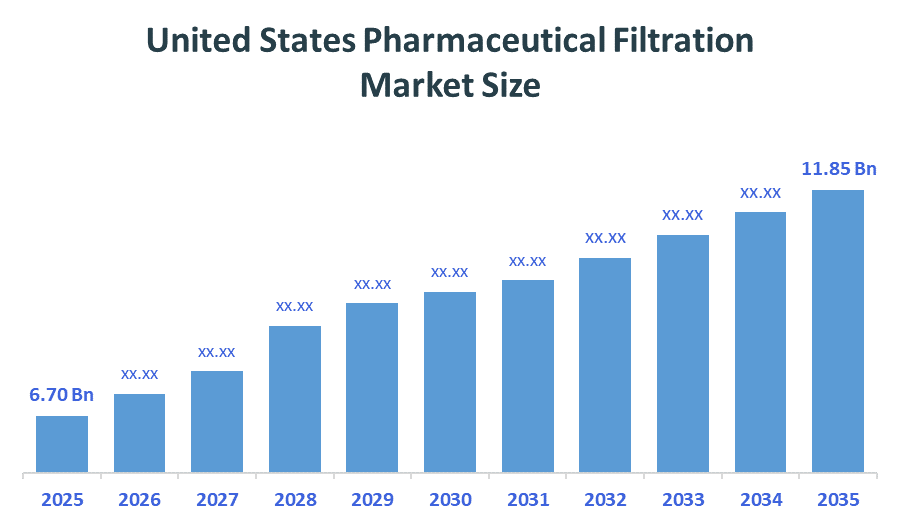
According to Decision Advisors, the United States Pharmaceutical Filtration Market size was grown from USD 6.70 Billion in 2025 to USD 11.85 Billion by 2035, at a CAGR of 5.87% during the forecast period 2026-2035. The United States pharmaceutical filtration market is projected to grow significantly over the next decade, fueled by increased R&D investments, advancements in membrane technology, the need for high-purity, single-use systems in drug manufacturing, the expansion of biologics/biosimilars, stringent regulatory standards, advancements in filtration technology, and focus on high-purity requirements.
Market Overview/Introduction
The United States pharmaceutical filtration market refers to the industry that provides advanced filtration technologies and systems for drug manufacturing operations, which need to maintain pharmaceutical product purity, sterility, and safety. Pharmaceutical filtration uses major methods, such as microfiltration, ultrafiltration, and nanofiltration, to remove three types of contaminants from solutions, which include contaminants, microbes, and particles. The market is expected to grow significantly because demand for biologics, vaccines, and sterile injectable drugs is increasing. Future opportunities include single-use filtration system expansion, personalized medicine growth, and continuous manufacturing adoption growth. The market experiences growth because of strict FDA rules, increasing biopharmaceutical production, technological advancements in membrane filters, and the rising research and development funding in the US healthcare and life sciences industry.
- The U.S. Food and Drug Administration (FDA) PreCheck Program is a government initiative designed to streamline regulatory processes and facilitate the development of new pharmaceutical manufacturing facilities in the United States, rather than directly “accelerating approvals.” It enhances early-stage communication, improves regulatory predictability, and supports faster establishment of drug production sites.
- The U.S. Food and Drug Administration (FDA) ANDA Prioritization Pilot Program is designed to support domestic generic drug manufacturing by prioritizing the review of Abbreviated New Drug Applications (ANDAs) for products that strengthen the U.S. drug supply chain, particularly those addressing shortages or public health needs. It does not strictly require local raw materials, but encourages U.S.-based production and supply reliability.
- Programs funded by the National Institutes of Health (NIH) provide extensive financial support for biologics, vaccines, and life sciences research, focusing on innovation in drug development and bioprocessing. These initiatives indirectly support the advancement of high-purity pharmaceutical manufacturing processes, including purification and sterile production technologies, rather than explicitly funding filtration systems alone.
Notable Insights: -
- The membrane filters segment dominated the market in 2025 and holds the largest market share, accounting for approximately 28% during the forecast period.
- The microfiltration segment dominated the market in 2025 and holds the largest market share, accounting for approximately 38% during the forecast period.
- The biopharmaceutical companies segment dominated the market in 2025 and holds the largest market share, accounting for approximately 45% during the forecast period.
- The compound annual growth rate of the United States Pharmaceutical Filtration Market is 5.87%.
- The market is likely to achieve a valuation of USD 11.85 Billion by 2035.
What is the role of technology in grooming the market?
Technology plays a vital role in advancing the United States Pharmaceutical Filtration Market by improving efficiency, safety, and product purity. Advanced filtration technologies such as membrane filtration, nanofiltration, and ultrafiltration enable precise separation of contaminants, ensuring high-quality drug production. Automation and digital monitoring systems enhance process control, reduce human error, and ensure compliance with strict FDA regulations. The integration of single-use filtration systems is increasing flexibility and reducing cross-contamination risks in biopharmaceutical manufacturing. Additionally, innovations in bioprocessing technologies support the production of complex biologics, vaccines, and gene therapies. Real-time data analytics and smart sensors help optimize filtration performance and maintenance schedules. Overall, technological advancements are transforming the U.S. pharmaceutical filtration market into a highly efficient, scalable, and quality-driven industry, supporting the growing demand for safe and effective pharmaceutical products.
Market Drivers
The United States pharmaceutical filtration market grows because the demand for complex and sensitive biologics and vaccines requires the use of precise and gentle filtration methods. The implementation of validated high-performance filtration systems, which include sterilization-grade filters, has increased because agencies now enforce stricter regulations about sterility and viral safety. The development of membrane technologies, which include single-use and disposable systems, has improved operational efficiency through contamination risk reduction. The market expansion benefits from increasing financial support that U.S. pharmaceutical companies provide for drug development and manufacturing facility construction. Companies adopt advanced filtration solutions because they need to maintain product purity, safety, and effectiveness.
Restrain
The United States pharmaceutical filtration market faces restraints such as high installation and operational costs, complex regulatory compliance requirements, and frequent filter replacement needs. Additionally, technical limitations in handling highly viscous solutions and potential product loss during filtration processes further challenge efficiency and increase overall production costs for manufacturers.
Study on the Supply, Demand, Distribution, and Market Environment of the United States Pharmaceutical Filtration Market
The United States pharmaceutical filtration market is driven by a strong and technologically advanced supply chain supported by leading global manufacturers of filtration systems and membranes. Supply is ensured through high-quality production of filtration media, while demand is rising due to increasing biologics, vaccines, and sterile drug manufacturing. Distribution occurs through direct partnerships between filtration providers and pharmaceutical companies, supported by specialized healthcare logistics networks ensuring regulatory compliance. The market environment is highly regulated by FDA standards, emphasizing product safety, sterility, and process validation. Key raw materials include polymeric membranes such as polyethersulfone (PES), polyvinylidene fluoride (PVDF), nylon, cellulose acetate, and advanced ceramic materials, which ensure high filtration efficiency and chemical compatibility. Growing demand for single-use systems and advanced bioprocessing solutions further strengthens market dynamics, while innovation in raw materials and membrane technologies continues to enhance performance, scalability, and cost efficiency across the U.S. pharmaceutical filtration industry.
Price Analysis and Consumer Behaviour Analysis
The United States pharmaceutical filtration market is characterized by high pricing due to advanced technology adoption, strict regulatory standards, and the use of high-quality filtration membranes and systems. Costs are influenced by raw materials such as polymeric membranes, single-use systems, and specialized bioprocessing components, which increase overall production expenses. Premium pricing is common for high-performance and sterile filtration solutions used in biologics and vaccine manufacturing. From a consumer behavior perspective, pharmaceutical and biotech companies prioritize product quality, regulatory compliance, and process efficiency over cost considerations. There is strong preference for reliable, validated, and scalable filtration systems that ensure product safety and consistency. Additionally, demand is increasing for single-use and disposable systems due to their ability to reduce contamination risks and operational downtime. Overall, purchasing decisions are value-driven, focusing on performance, compliance, and long-term operational efficiency rather than low-cost alternatives.
Market Segmentation
The United States Pharmaceutical Filtration Market share is classified into material type, application, and end user
- The membrane filters segment dominated the market in 2025 and holds the largest market share, accounting for approximately 28% during the forecast period.
Based on the product type, the pharmaceutical filtration market is divided into membrane filters, prefilters & depth media, single-use systems, cartridge & capsule filters, filter holders, and filtration accessories. Among these, the membrane filters segment dominated the market in 2025 and holds the largest market share, accounting for approximately 28% during the forecast period. The membrane filters segment is driven by the system, which demonstrates exceptional performance through its ability to conduct sterile filtration and microbial removal, and particle separation while maintaining drug integrity. The system has become a standard equipment choice for manufacturing biologics, vaccines, and injectable drugs because it provides essential accuracy and meets all required regulatory standards. The system secures its top market position through its adaptable nature and availability of advanced materials, which include PES, PVDF, nylon, and its ability to meet GMP standards.
- The microfiltration segment dominated the market in 2025 and holds the largest market share, accounting for approximately 38% during the forecast period.
Based on the technique, the pharmaceutical filtration market is divided into microfiltration, ultrafiltration, nanofiltration, cross-flow filtration, and reverse osmosis. Among these, the microfiltration segment dominated the market in 2025 and holds the largest market share, accounting for approximately 38% during the forecast period. The microfiltration segment in the U.K. is attributed to the product serving three main purposes, which include sterile filtration, bio burden reduction, and final product clarification. The system provides high efficiency and cost-effectiveness while operating efficiently in large-scale biopharmaceutical production. The product maintains its dominant market presence because it sees widespread use in vaccine development, biologics production, and injectable drug manufacturing.
- The biopharmaceutical companies segment dominated the market in 2025 and holds the largest market share, accounting for approximately 45% during the forecast period.
Based on the end user, the biopharmaceutical filtration market is divided into pharmaceutical companies, biopharmaceutical companies, hospitals & clinics, research institutes, and contract manufacturing organizations (CMOs). Among these, the biopharmaceutical companies segment dominated the market in 2025 and holds the largest market share, accounting for approximately 45% during the forecast period. The biopharmaceutical companies segment in the U.K. is driven by the process of developing monoclonal antibodies and vaccines,, cell and gene therapies and recombinant proteins requires scientists to use advanced filtration methods. The pharmaceutical industry in the United States needs filtration technologies because its operations depend on sterile processing and single-use systems and must follow strict regulatory standards.
Recent Development
- In June 2025, Cytiva (a subsidiary of Danaher Corporation) announced the expansion of its global manufacturing capacity, including North America, as part of a $1.6 billion investment program to support growing demand for bioprocessing and liquid filtration solutions.
- In March 2024, Merck KGaA (operating as MilliporeSigma in the U.S.) launched the Stericup Quick Release-GP Filter System, designed to improve efficiency, ease of handling, and safety in sterile filtration workflows. The system features a quick-release mechanism that simplifies filter handling and reduces contamination risks in laboratory and pharmaceutical applications.
Competitive Analysis
The report offers the appropriate analysis of the key organisations/companies involved within the United States Pharmaceutical filtration market, along with a comparative evaluation primarily based on their product offering, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborative analysis focusing on the current news and developments of the companies, which includes product development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
Top Key Companies in the United States Pharmaceutical Filtration Market
- Danaher Corporation (Pall Corporation & Cytiva)
- Thermo Fisher Scientific Inc.
- 3M Company
- Merck KGaA (MilliporeSigma)
- Parker Hannifin Corporation
- Others
Key Target Audience
- Market Players
- Investors
- End-users
- Government Authorities
- Consulting and Research Firm
- Venture capitalists
- Value-Added Resellers (VARs)
Market Segment
This study forecasts revenue at the United States, regional, and country levels from 2020 to 2035. Spherical Insights has segmented the United States pharmaceutical filtration market based on the below-mentioned segments
United States Pharmaceutical Filtration Market, By Product Type
- Membrane Filters
- Prefilters & Depth Media
- Single-Use Systems
- Cartridge & Capsule Filters
- Filter Holders
- Filtration Accessories
United States Pharmaceutical Filtration Market, By Technique
- Microfiltration
- Ultrafiltration
- Nanofiltration
- Cross-Flow Filtration
- Reverse Osmosis
United States Pharmaceutical Filtration Market, By End User
- Pharmaceutical Companies
- Biopharmaceutical Companies
- Hospitals & Clinics
- Research Institutes
- Contract Manufacturing Organizations (CMOs)
Frequently Asked Questions (FAQ)
Q. What challenges do manufacturers face in scaling up pharmaceutical filtration processes in the U.S.?
A. Scaling up filtration processes is challenging due to maintaining consistent performance across different production volumes. Issues such as filter fouling, pressure drop variations, and product yield loss become more significant at a large scale. Additionally, ensuring validation consistency, regulatory approval, and cost efficiency during scale-up adds complexity, especially in biologics and vaccine manufacturing environments.
Q. How is the rise of personalized medicine influencing the U.S. pharmaceutical filtration market?
A. The growth of personalized medicine is increasing demand for highly precise and small-batch drug production, which requires advanced filtration systems. Technologies like single-use filters and ultra-clean membrane systems are becoming essential to maintain sterility and product integrity. This shift is pushing manufacturers to adopt flexible, scalable filtration solutions that can handle customized therapies efficiently while ensuring regulatory compliance and consistent quality.
Q. Why is sustainability becoming important in pharmaceutical filtration systems in the U.S.?
A. Sustainability is gaining importance due to increasing environmental regulations and industry focus on reducing waste. Traditional filtration systems generate significant plastic and disposable waste, especially single-use components. Companies are now exploring recyclable materials, reusable filter systems, and greener manufacturing processes to reduce environmental impact while maintaining high sterility and performance standards.
Q. How does supply chain resilience impact the U.S. pharmaceutical filtration market?
A. Supply chain resilience is critical because filtration systems depend on specialized raw materials like advanced polymers and membranes. Disruptions can delay drug production and impact critical therapies. As a result, companies are diversifying suppliers, increasing local manufacturing, and building strategic inventory reserves to ensure continuous availability of filtration components, especially during high-demand periods like pandemics or vaccine rollouts.
Check Licence
Choose the plan that fits you best: Single User, Multi-User, or Enterprise solutions tailored for your needs.
We Have You Covered
- 24/7 Analyst Support
- Clients Across the Globe
- Tailored Insights
- Technology Tracking
- Competitive Intelligence
- Custom Research
- Syndicated Market Studies
- Market Overview
- Market Segmentation
- Growth Drivers
- Market Opportunities
- Regulatory Insights
- Innovation & Sustainability
Report Details
| Scope | Country |
| Pages | 210 |
| Delivery | PDF & Excel via Email |
| Language | English |
| Release | Apr 2026 |
| Access | Download from this page |
