United States Rheumatoid Arthritis Therapeutics Market
United States Rheumatoid Arthritis Therapeutics Market Size, Share, By Drug Class (Biologics, Conventional DMARDs, Targeted Synthetic DMARDs (JAK inhibitors), NSAIDs, Corticosteroids), By Route of Administration (Oral, Injectable, Intravenous), By End User (Hospitals, Specialty Clinics, Homecare Settings), By End User (Hospitals, Clinics, Research Institutes, Ambulatory Surgical Centers) Analysis and Forecast 2026-2035
Report Overview
Table of Contents
Market Snapshot
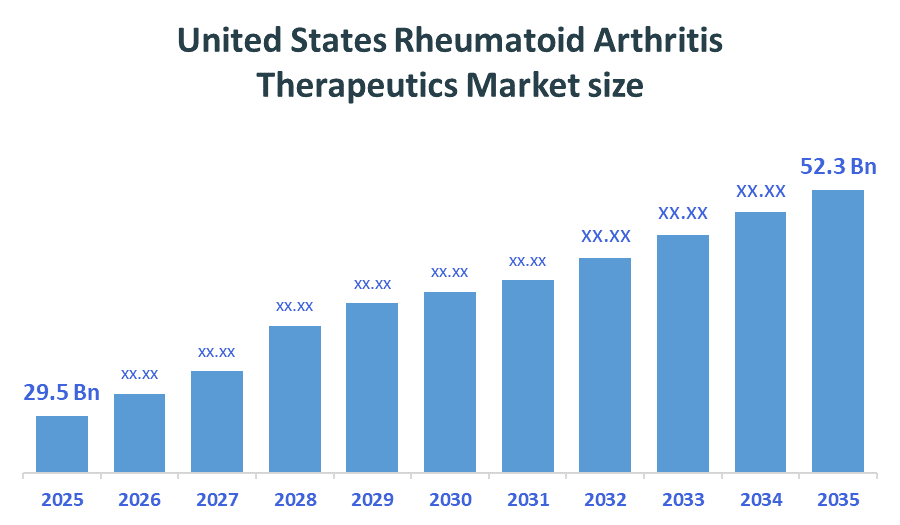
- United States Rheumatoid Arthritis Therapeutics Market size (2025): USD 29.5Billion
- Projected United States Rheumatoid Arthritis Therapeutics Market Size (2035): USD 52.3Billion
- United States Rheumatoid Arthritis Therapeutics Market Compound Annual Growth Rate (CAGR): 5.89%
- Base Year: 2025
- Historical Period: 2021–2024
- Forecast Period: 2026–2035
Market Overview/Introduction
The United States Rheumatoid Arthritis (RA) therapeutics market refers to the healthcare segment that focuses on creating, manufacturing, selling drugs that treat rheumatoid arthritis, an ongoing autoimmune disease. The treatments for rheumatoid arthritis use biologic agents, disease-modifying antirheumatic drugs, JAK inhibitors, and biosimilars to reduce inflammation and prevent joint damage, which increases patient happiness. The market will experience substantial expansion because rheumatoid arthritis cases increase, together with the elderly population growth and the rising adoption of innovative biologic medicines. The future holds potential for personalized medicine, biosimilar development, and AI-based drug discovery research. The United States market expansion depends on multiple factors, that include substantial research and development funding, together with supportive reimbursement systems and ongoing progress in targeted therapy development, improved diagnostic capabilities, and wider access to specialized medical services.
- The U.S. FDA provides regulatory frameworks and guidance to speed up development of RA therapies, including biologics and biosimilars. These include structured clinical development pathways, fast-track designations, and biologics evaluation programs that help bring innovative RA treatments to market faster.
- Medicare in the United States supports rheumatoid arthritis patients by covering biologics and DMARDs under Part B (physician-administered drugs) and Part D (prescription drugs). This improves access to high-cost therapies such as monoclonal antibodies and JAK inhibitors, especially among elderly patients, thereby increasing treatment adoption and supporting market growth.
- Programs such as Medicare Extra Help and various copay assistance initiatives help reduce out-of-pocket expenses for rheumatoid arthritis patients in the United States. These financial support mechanisms improve affordability of biologics and DMARDs, enhance patient adherence to long-term treatment regimens, and support sustained use of advanced RA therapies, driving market growth.
Notable Insights: -
- The biologics segment dominated the market in 2025 and holds the largest market share, accounting for approximately 58% during the forecast period.
- The injectable segment dominated the market in 2025 and holds the largest market share, accounting for approximately 68% during the forecast period.
- The hospitals segment dominated the market in 2025 and holds the largest market share, accounting for approximately 50% during the forecast period.
- The compound annual growth rate of the United States Rheumatoid Arthritis Therapeutics Market is 5.89%.
- The market is likely to achieve a valuation of USD 52.3 Billion by 2035.
What is the role of technology in grooming the market?
Technology plays a crucial role in shaping the United States Rheumatoid Arthritis (RA) therapeutics market by improving drug discovery, diagnosis, treatment precision, and patient management. Advanced technologies such as AI and machine learning accelerate biologics and small-molecule drug development by identifying molecular targets and predicting drug responses. Digital health tools, including wearable devices and mobile health apps, enable real-time monitoring of disease progression and treatment effectiveness. Precision medicine and genomics support personalized RA therapies, improving patient outcomes. Additionally, automation in biopharmaceutical manufacturing enhances production efficiency and quality of biologics. Telemedicine platforms also expand access to rheumatology care, ensuring timely diagnosis and follow-ups, ultimately driving market growth and improving long-term disease management in the U.S.
Market Drivers
The United States Rheumatoid Arthritis therapeutics market is driven by the rising prevalence of autoimmune disorders and an aging population increasingly affected by chronic inflammatory diseases. The development of biologics and JAK inhibitors, together with targeted DMARDs, has resulted in improved treatment results, which clinicians now use at higher rates. Pharmaceutical companies that invest heavily in research and development, together with the continuous FDA authorization of new therapies, will drive market expansion. The increasing understanding of the importance of early diagnosis and treatment will create a greater need for solutions that provide lasting results. Patients can access expensive biologics because insurance plans and reimbursement systems provide favourable coverage; meanwhile, biosimilars become more accessible, which reduces costs and allows their penetration throughout the entire U.S. healthcare market.
Restrain
The United States rheumatoid arthritis therapeutics market faces restraints such as high treatment costs of biologics, strict FDA regulatory requirements, and complex clinical approval processes. Patent expirations and biosimilar competition also impact revenues. Additionally, adverse side effects, limited accessibility for uninsured patients, and reimbursement challenges further restrict market growth.
Study on the Supply, Demand, Distribution, and Market Environment of the United States Rheumatoid Arthritis Therapeutics Market
The United States Rheumatoid Arthritis (RA) therapeutics market is characterized by a well-structured ecosystem driven by strong biologic drug supply chains, high patient demand, and advanced distribution networks. On the supply side, the market is dominated by major pharmaceutical companies producing biologics, biosimilars, and JAK inhibitors, with high manufacturing complexity and strict FDA regulations limiting new entrants. Continuous innovation and biosimilar expansion are gradually improving supply availability and cost efficiency. On the demand side, rising RA prevalence, aging population, and early diagnosis rates are increasing long-term therapy needs, with biologics being the most prescribed treatment category. The distribution system is highly centralized, relying on specialty pharmacies, hospital pharmacies, and insurance-linked reimbursement channels, ensuring controlled access to high-cost therapies. The market environment is highly competitive and patent-driven, with strong dominance by leading firms and significant barriers due to R&D costs, regulatory approvals, and intellectual property protection. However, growing biosimilar penetration and value-based healthcare policies are gradually reshaping pricing and accessibility dynamics.
Price Analysis and Consumer Behaviour Analysis
In the United States Rheumatoid Arthritis (RA) therapeutics market, price analysis shows consistently high treatment costs, mainly driven by biologics and targeted synthetic DMARDs such as monoclonal antibodies and JAK inhibitors. Biologic therapies often range from several thousand to approximately USD 20,000 annually per patient, although biosimilars are gradually improving affordability and increasing price competition. Insurance coverage, Medicare reimbursement policies, and specialty pharmacy distribution significantly influence net patient costs. Consumer behaviour is strongly shaped by long-term disease management needs, leading patients to prioritize efficacy, safety, and insurance coverage over price sensitivity. Physicians heavily influence treatment decisions, especially in biologic switching and initiation. Patients increasingly show willingness to adopt biosimilars due to lower co-pays and formulary incentives. Additionally, growing awareness of early diagnosis and advanced therapies is increasing demand for premium, long-acting injectable and oral RA treatments across the U.S.
Market Segmentation
The United States Rheumatoid Arthritis Therapeutics Market share is classified into Drug Class, Route of Administration, and end user
- The biologics segment dominated the market in 2025 and holds the largest market share, accounting for approximately 58% during the forecast period.
Based on the drug class, the rheumatoid arthritis therapeutics market is divided into biologics, conventional DMARDs, targeted synthetic DMARDs (JAK inhibitors), NSAIDs, and corticosteroids. Among these, he biologics segment dominated the market in 2025 and holds the largest market share, accounting for approximately 58% during the forecast period. The biologics segment is driven by its superior clinical efficacy in treating moderate to severe RA cases and its ability to slow disease progression. High physician preference, strong patient response rates, and widespread insurance reimbursement further support adoption. Additionally, continuous innovation in monoclonal antibodies and TNF inhibitors, along with expanding FDA approvals and biosimilar availability, has strengthened market penetration.
- The injectable segment dominated the market in 2025 and holds the largest market share, accounting for approximately 68% during the forecast period.
Based on the route of administration, the rheumatoid arthritis therapeutics market is divided into immunology, hematology, neurology, and critical care. Among these, the injectable segment dominated the market in 2025 and holds the largest market share, accounting for approximately 68% during the forecast period. The injectable segment in the U.K. is attributed to its strong association with biologic and advanced RA therapies, which require subcutaneous or intravenous administration for higher effectiveness and rapid symptom control. These formulations ensure better bioavailability and sustained therapeutic action in moderate-to-severe cases. Additionally, hospital-based administration, strong physician preference, and widespread insurance reimbursement support further drive adoption.
- The hospitals segment dominated the market in 2025 and holds the largest market share, accounting for approximately 50% during the forecast period.
Based on the end user, the rheumatoid arthritis therapeutics market is divided into hospitals, clinics, research institutes, and ambulatory surgical centers. Among these, the hospitals segment dominated the market in 2025 and holds the largest market share, accounting for approximately 50% during the forecast period. The hospitals segment in the U.K. is driven by the high volume of complex RA cases requiring specialist supervision and advanced treatment administration, particularly biologics and infusion therapies. Hospitals provide better diagnostic infrastructure, insurance-supported treatment access, and emergency care for severe flare-ups. Additionally, the presence of rheumatology specialists and access to advanced biologic drugs further strengthen hospital-based treatment adoption.
Recent Development
- In October 2025, Celltrion launched Eydenzelt, an aflibercept biosimilar, expanding biologic competition and improving affordability and access within the broader U.S. autoimmune and rheumatoid arthritis therapeutics market.
- In April 2025, Biocon received FDA approval for Jobevne, a bevacizumab biosimilar, strengthening the U.S. biosimilars landscape relevant to autoimmune and inflammatory conditions.
- In March 2025, the U.S. FDA continued its expansion of biosimilar approvals for autoimmune and inflammatory diseases relevant to rheumatoid arthritis, including adalimumab biosimilars. These approvals improved affordability, increased market competition, and enhanced patient access to biologic therapies, supporting wider adoption of advanced rheumatoid arthritis therapeutics across the United States healthcare system.
Competitive Analysis
The report offers the appropriate analysis of the key organisations/companies involved within the United States Rheumatoid Arthritis Therapeutics Market, along with a comparative evaluation primarily based on their product offering, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborative analysis focusing on the current news and developments of the companies, which includes product development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
Top Key Companies in the United States Rheumatoid Arthritis Therapeutics Market
- AbbVie Inc.
- Johnson & Johnson (Janssen Biotech)
- Amgen Inc.
- Pfizer Inc.
- Bristol Myers Squibb
- Others
Key Target Audience
- Market Players
- Investors
- End-users
- Government Authorities
- Consulting and Research Firm
- Venture capitalists
- Value-Added Resellers (VARs)
Market Segment
This study forecasts revenue at the United States, regional, and country levels from 2020 to 2035. Spherical Insights has segmented the United States rheumatoid arthritis therapeutics market basedon the below-mentioned segments
United States Rheumatoid Arthritis Therapeutics Market, By Drug Class
- Biologics
- Conventional DMARDs
- Targeted Synthetic DMARDs (JAK inhibitors)
- NSAIDs
- Corticosteroids
United States Rheumatoid Arthritis Therapeutics Market, By Route of Administration
- Oral
- Injectable
- Intravenous
United States Rheumatoid Arthritis Therapeutics Market, By End User
- Hospitals
- Specialty Clinics
- Homecare Settings
Frequently Asked Questions (FAQ)
Q. What are the emerging innovations shaping treatment approaches in the U.S. Rheumatoid Arthritis therapeutics market?
A. Emerging innovations include precision medicine, AI-based drug discovery, and next-generation biologics such as bispecific antibodies. These advancements are improving treatment targeting, reducing side effects, and enabling personalized therapy selection based on genetic and biomarker profiling, leading to better patient outcomes and faster disease control.
Q. How does patient adherence impact the growth of the Rheumatoid Arthritis therapeutics market in the United States?
A. Patient adherence significantly influences market growth because RA requires long-term, continuous treatment. Improved drug formulations like long-acting injectable and oral JAK inhibitors enhance compliance. Better adherence reduces disease progression, increases repeat prescriptions, and supports sustained demand for advanced therapies.
Q. What role do biosimilars play in reshaping the competitive landscape of the U.S. RA therapeutics market?
A. Biosimilars are increasing affordability and expanding patient access to biologic therapies. They introduce price competition against originator biologics, reduce healthcare spending, and are accelerating market penetration in insurance-driven systems. This is gradually shifting market share dynamics among leading pharmaceutical companies.
Q. How is the aging population influencing demand in the U.S. Rheumatoid Arthritis therapeutics market?
A. The aging population is a major growth driver because older adults are more prone to autoimmune and inflammatory diseases like RA. This demographic shift increases long-term therapy demand, boosts biologics usage, and expands healthcare utilization, thereby strengthening overall market growth.
Check Licence
Choose the plan that fits you best: Single User, Multi-User, or Enterprise solutions tailored for your needs.
We Have You Covered
- 24/7 Analyst Support
- Clients Across the Globe
- Tailored Insights
- Technology Tracking
- Competitive Intelligence
- Custom Research
- Syndicated Market Studies
- Market Overview
- Market Segmentation
- Growth Drivers
- Market Opportunities
- Regulatory Insights
- Innovation & Sustainability
Report Details
| Scope | country |
| Pages | 240 |
| Delivery | PDF & Excel via Email |
| Language | English |
| Release | Apr 2026 |
| Access | Download from this page |
