US Transdermal Drug Delivery System Market
US Transdermal Drug Delivery System Market Size, Share, By Type (Matrix Patch, Reservoir Patch, Drug-in-Adhesive), By Application (Pain Management, Hormonal Therapy, Cardiovascular Diseases, CNS Disorders, Others), By End User (Hospitals, Home Care Settings, Specialty Clinics), Analysis and Forecast 2025?2035.
Report Overview
Table of Contents
Market Snapshot
- US Transdermal Drug Delivery System Market Size (2025): USD 41.9 Billion
- Projected US Transdermal Drug Delivery System Market Size (2035): USD 123.3 Billion
- US Transdermal Drug Delivery System Market Compound Annual Growth Rate (CAGR): 11.4%
- Market Base Year: 2025
- Historical Period: 2021–2024
- Forecast Period: 2026–2035
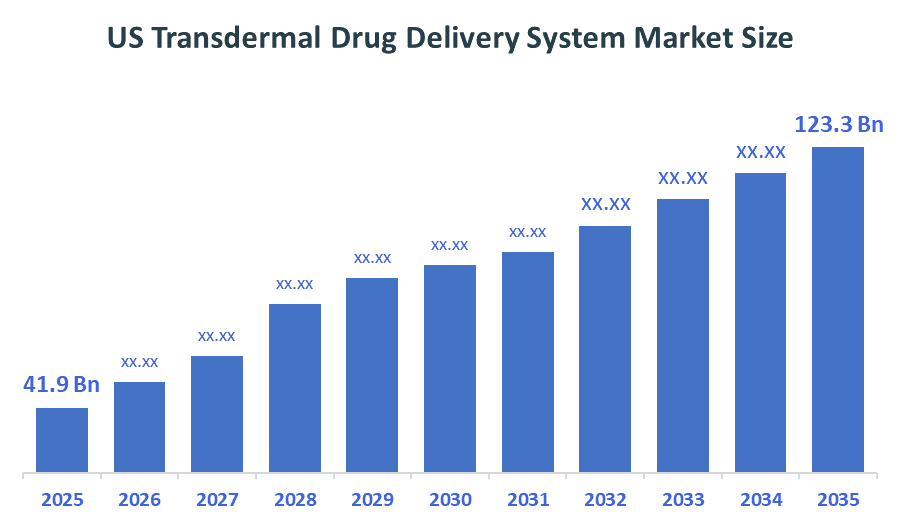
According to Decision Advisors, the US Transdermal Drug Delivery System Market Size is expected to grow from USD 41.9 Billion in 2025 to USD 123.3 Billion by 2035, at a CAGR of 11.4% during the forecast period 2025-2035. The market is projected to grow significantly over the next decade, driven by the rising prevalence of chronic diseases, increasing demand for non-invasive and self-administered drug delivery methods, and growing adoption of home healthcare solutions.
Market Overview/Introduction
The U.S. Transdermal Drug Delivery System Market is defined by the systemic administration of therapeutic agents through the skin, typically via specialized patches that bypass gastrointestinal degradation. Because these patches bypass the stomach, they’re easier on the body and much more convenient for patients. The market is boom is mostly because more people are dealing with long-term health issues and, frankly, nobody likes needles if they can avoid them. Big names like 3M, Johnson & Johnson, and Novartis are leading the charge, putting money into better materials and buying up smaller biotech firms to make these patches more effective at absorbing medicine. Looking ahead, the real game-changer is going to be microneedles and smart patches. These next-gen devices will use tiny sensors to monitor your health and could finally allow us to deliver complex drugs like insulin or vaccines through a simple patch, something that used to require a painful injection.
- FDA’s GDUFA regulatory science program, over USD 20 million has been allocated toward research projects focusing on transdermal and topical delivery systems, including bioequivalence and adhesion testing.
Notable Insights: -
- By type, the drug-in-adhesive segment dominated the market in 2025, accounting for approximately 55% of market share, and is projected to grow at a substantial CAGR during the forecast period.
- By end user, the home care settings segment dominated the market in 2025, accounting for approximately 48% of market share, and is projected to grow at a substantial CAGR during the forecast period.
- The compound annual growth rate of the US transdermal drug delivery System Market is 11.4%.
- The market is likely to achieve a valuation of USD 123.3 Billion by 2035.
What is role of technology in grooming the market?
Technology plays a transformative role in the transdermal drug delivery system market through advancements in patch design, drug permeability enhancement, and smart delivery systems. Innovations such as microneedle patches, iontophoresis, and thermal ablation technologies improve drug penetration across the skin barrier. Additionally, the integration of wearable and smart patches with digital monitoring capabilities allows real-time tracking of drug release and patient adherence. Advanced polymer science and nanotechnology are also enabling controlled and sustained drug delivery, enhancing therapeutic outcomes and reducing side effects
Market Drivers
The expansion of the U.S. Transdermal Patch Market is really about a shift toward healthcare that feels more human. As more Americans live with long-term conditions like diabetes or chronic pain, the daily grind of swallowing pills or facing painful needles can become a heavy emotional and physical burden. We're seeing a move toward patches because they offer a gentler, set-it-and-forget-it way to stay healthy. They are discreet, easy to use, and they skip the stomach issues that many oral meds cause. Technological breakthroughs are also meeting us where we are. We are moving into an era of smart patches that use tiny sensors to keep an eye on your vitals and adjust your dose automatically. This is especially vital as we look at the rising impact of heart disease on American women, having a simple, wearable patch to manage blood pressure or cholesterol can be a lifesaver for a busy woman who doesn’t want her diagnosis to define her day.
In February 2026, the American Heart Association warns 60% of US women will have cardiovascular disease by 2050, which boosts the future growth of the market.
Study on the Supply, Demand, Distribution, and Market Environment of the US Transdermal Drug Delivery System Market
The U.S. Transdermal Drug Delivery System (TDDS) market operates within a well-established and innovation-driven healthcare ecosystem, characterized by strong supply capabilities, rising demand, and an efficient distribution network. On the supply side, the market is supported by advanced pharmaceutical manufacturing infrastructure and continuous R&D investments in polymer science, drug permeability technologies, and smart patch systems. Leading pharmaceutical companies are increasingly focusing on developing next-generation transdermal patches, including microneedle-based and digitally integrated systems, to enhance drug delivery efficiency and patient outcomes. On the demand side, the market is primarily driven by the growing prevalence of chronic diseases, with over 6 in 10 adults in the U.S. suffering from at least one chronic condition, alongside a rapidly aging population that prefers non-invasive and easy-to-use therapies. Additionally, increasing awareness of self-administration and home healthcare has significantly boosted demand for transdermal systems, particularly for pain management and hormonal therapies. In terms of distribution, TDDS products are widely accessible through retail and hospital pharmacies, online platforms, and direct supply agreements with healthcare providers, ensuring broad market penetration. The market environment is highly regulated, with strict oversight by the FDA governing product approval, safety, and quality standards, which, while increasing compliance costs, ensures high product reliability and patient safety. Overall, the U.S. TDDS market reflects a balanced and competitive environment where strong demand, technological innovation, and robust distribution channels collectively drive sustained growth.
Price Analysis and Consumer Behaviour Analysis
The U.S. Transdermal Drug Delivery System (TDDS) market reflects a value-driven pricing structure, where products are moderately priced but deliver high therapeutic and economic benefits over time. In 2025, the average cost of branded transdermal patches remains higher than conventional oral medications due to advanced formulation technologies, stringent FDA compliance, and complex manufacturing processes. However, the presence of over 25 FDA-approved transdermal patches and a pipeline exceeding 40 products in clinical development has increased competitive pricing pressure, especially from generic alternatives. Additionally, fluctuations in demand, such as recent shortages in hormone replacement patches, have led to temporary price increases in specific segments. Despite higher upfront costs, TDDS products help reduce overall healthcare expenditure by lowering hospital visits and improving adherence, making them cost-effective in long-term treatment scenarios.
Market Segmentation
The US Transdermal Drug Delivery System Market share is classified into type, application, and end user.
- The drug-in-adhesive segment dominated the market in 2025, accounting for approximately 55% of market share, and is projected to grow at a substantial CAGR during the forecast period.
Based on the type, the US transdermal drug delivery system market is divided into matrix patches, reservoir patches, and drug-in-adhesive systems. Among these, the drug-in-adhesive segment dominated the market in 2025, accounting for approximately 55% of market share, and is projected to grow at a substantial CAGR during the forecast period. Drug-in-adhesive systems dominate due to their simplified design, improved patient comfort, and cost-effectiveness, as they combine the drug and adhesive layer into a single system, reducing manufacturing complexity and enhancing drug stability.
Recent developments in the U.S. pharmaceutical sector highlight increasing investments in next-generation adhesive technologies and microneedle-integrated patches, further supporting segment growth. Additionally, news around FDA approvals of advanced transdermal formulations for pain and neurological conditions has accelerated adoption. However, reservoir patches are gaining traction in specialized therapies requiring controlled and sustained drug release, particularly for hormonal and chronic disease treatments.
- The pain management segment accounted for the largest share in 2025 and is anticipated to grow at a significant CAGR of approximately 9.5% during the forecast period.
Based on the application, the US transdermal drug delivery system Market is divided into pain management, hormonal therapy, cardiovascular diseases, CNS disorders, and others. Among these, the pain management segment accounted for the largest share in 2025 and is anticipated to grow at a significant CAGR of approximately 9.5% during the forecast period. Transdermal patches are widely used in pain management due to their ability to provide continuous, controlled drug release, minimizing the need for frequent dosing and reducing systemic side effects.
Recent news trends indicate a strong push in the U.S. toward opioid alternatives, with transdermal patches emerging as a safer option for chronic pain relief. Additionally, increasing approvals and clinical trials for non-opioid analgesic patches and lidocaine-based systems are further boosting this segment. The use of transdermal systems in CNS disorders and hormonal therapies is also expanding, driven by innovations in drug permeability technologies and wearable drug delivery systems.
- The home care settings segment dominated the market in 2025, accounting for approximately 48% of market share, and is projected to grow at a substantial CAGR during the forecast period.
Based on the end user, the US transdermal drug delivery system market is divided into hospitals, home care settings, and specialty clinics. Among these, the home care settings segment dominated the market in 2025, accounting for approximately 48% of market share, and is projected to grow at a substantial CAGR during the forecast period. Home care settings dominate due to the increasing preference for self-administration, convenience, and reduced hospital visits, particularly among elderly and chronically ill patients.
Recent Development
- In February 2026, The U.S. has witnessed a significant increase in demand for estradiol transdermal patches, with sales rising from 46.5 million units in 2020 to over 142 million units in 2025, leading to supply shortages across multiple products. This reflects strong market demand driven by increased awareness and regulatory changes supporting hormone therapy.
- In October 2025, a U.S.based biotechnology company reported positive Phase 2/3 clinical trial results for a nanoengineered cisplatin-loaded transdermal patch (PRV111) designed for oral cancer treatment. The patch demonstrated the ability to deliver localized chemotherapy effectively while reducing the need for surgical intervention, highlighting the growing role of advanced transdermal systems in oncology.
Competitive Analysis
The report offers the appropriate analysis of the key organisations/companies involved within the US transdermal drug delivery system market, along with a comparative evaluation primarily based on their product offering, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborative analysis focusing on the current news and developments of the companies, which includes product development, innovations, joint ventures, partnerships, mergers & acgquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
Top Key Companies in the US Transdermal Drug Delivery System Market
- Novartis AG
- Johnson & Johnson
- Viatris Inc. (formerly Mylan)
- Hisamitsu Pharmaceutical Co., Inc.
- 3M Company
Key Target Audience
- Market Players
- Investors
- End-users
- Government Authorities
- Consulting and Research Firm
- Venture capitalists
- Value-Added Resellers (VARs)
Market Segment
This study forecasts revenue at the United Kingdom, regional, and country levels from 2020 to 2035. Decision Advisors has segmented the US transdermal drug delivery system market based on the below-mentioned segments
US Transdermal Drug Delivery System Market, By Type
- Matrix Patch
- Reservoir Patch
- Drug-in-Adhesive
US Transdermal Drug Delivery System Market, By Application
- Pain Management
- Hormonal Therapy
- Cardiovascular Diseases
- CNS Disorders
- Others
US Transdermal Drug Delivery System Market, By End User
- Hospitals
- Home care Settings
- Specialty Clinics
Frequently Asked Questions (FAQ)
Q. What types of drugs are most suitable for transdermal drug delivery systems?
A. Drugs with low molecular weight, high potency, and good skin permeability, such as nicotine, hormones (e.g., estrogen), and certain pain medications, are most suitable for transdermal delivery. Large-molecule drugs typically require advanced technologies like microneedles.
Q. How do transdermal patches compare with oral medications in terms of bioavailability?
A. Transdermal patches often provide higher and more consistent bioavailability by bypassing the gastrointestinal tract and first-pass liver metabolism, resulting in more stable drug concentrations in the bloodstream.
Q. What are the key challenges in developing transdermal drug delivery systems?
A. Major challenges include limited drug permeability through the skin barrier, risk of skin irritation, difficulty in delivering large or hydrophilic molecules, and high development and formulation costs.
Q. How is nanotechnology influencing the transdermal drug delivery market?
A. Nanotechnology enhances drug penetration through the skin by using nanoparticles, liposomes, and nano-carriers, enabling improved delivery of complex drugs and expanding the range of treatable conditions.
Q. What role does patient lifestyle play in the adoption of transdermal patches?
A. Patient lifestyle significantly impacts adoption, as individuals with busy schedules or those requiring long-term therapy prefer patches due to their convenience, reduced dosing frequency, and discreet usage.
Check Licence
Choose the plan that fits you best: Single User, Multi-User, or Enterprise solutions tailored for your needs.
We Have You Covered
- 24/7 Analyst Support
- Clients Across the Globe
- Tailored Insights
- Technology Tracking
- Competitive Intelligence
- Custom Research
- Syndicated Market Studies
- Market Overview
- Market Segmentation
- Growth Drivers
- Market Opportunities
- Regulatory Insights
- Innovation & Sustainability
Report Details
| Scope | Country |
| Pages | 210 |
| Delivery | PDF & Excel via Email |
| Language | English |
| Release | Apr 2026 |
| Access | Download from this page |
