Global Uveitis Therapeutics Market
Global Uveitis therapeutics market Size, Share, By Drug Class (Anti-inflammatory Medications, Antibiotics and Antivirals, Immunosuppressive Medications), By Route of Administration (Oral, Intraocular, Intravenous), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), and By Region (North America, Europe, Asia-Pacific, Latin America, Middle East & Africa), Analysis and Forecast 2026-2035
Report Overview
Table of Contents
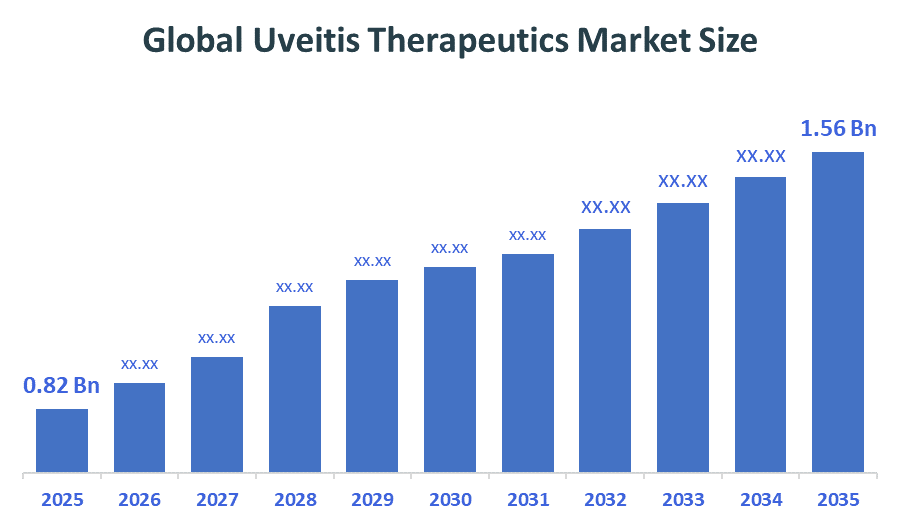
The Global Uveitis Therapeutics Market size is forecast to grow from USD 0.82 billion in 2025 to approximately USD 1.56 billion by 2035. According to Decision Advisors, a detailed research report on the uveitis therapeutics market indicates that the rapid shift toward the biologic therapies (especially TNF inhibitors and advanced immunology drugs) trend dominates the market, accounting for approximately 30-35% of the total share globally. AbbVie Inc. leads the market with an estimated 20-25% global market share. The company reported a 2025 revenue of approximately USD 15.7 Billion, making it one of the most influential forces shaping industry trends and overall market growth.
Market Snapshot
- Global Uveitis Therapeutics Market Size (2025): USD 0.82 Billion
- Projected Global Uveitis Therapeutics Market Size (2035): USD 1.56 Billion
- Global Uveitis Therapeutics Market Compound Annual Growth Rate (CAGR): 6.64%
- Largest Regional Market: North America
- Fastest Growing Region: Asia-Pacific
- Market 3rd Largest Region: Europe
- Base Year: 2025
- Historical Period: 2021–2024
- Forecast Period: 2026–2035
Market Overview/ Introduction
The Global Uveitis Therapeutics Market refers to the industry that focuses on evolving, manufacturing, and offering treatments that treat uveitis, an eye disease that affects the uveal tract. Uveitis therapeutics include corticosteroids, immunosuppressants, biologics and targeted immunomodulatory agents, which doctors use to control inflammation and protect patients from vision impairment. The market will expand through increasing demand for biologic therapies and personalized medicine, and AI-assisted diagnosis tools, which emerging economies will bring into the market. Market growth occurs because more people develop autoimmune disorders, the aging population increases, diagnostic technologies advance, and people learn more about eye health. The global market will experience sustained growth and innovation through advancements in drug delivery systems combined with the development of healthcare systems.
- The National Medical Products Administration (NMPA) has implemented Announcement No. 3 of 2026, which optimizes the fast-track review process for overseas drugs in "urgent clinical need." This initiative significantly accelerates the entry of innovative uveitis therapies like long-term fluocinolone acetonide implants, which was the first drug approved in China based on real-world data.
- The 2026 Union Budget launched the 'Biopharma SHAKTI' initiative with a ?10,000 crore outlay to establish India as a hub for domestic manufacturing of biologics and biosimilars, directly impacting uveitis treatment accessibility. Additionally, the government plans to create a network of over 1,000 accredited clinical trial sites to decentralize high-end research beyond major cities.
Notable Insights: -
- North America is anticipated to hold the largest share of approximately 40% in the uveitis therapeutics market over the forecast period.
- Asia Pacific is expected to grow at a rapid CAGR of approximately 7% in the uveitis therapeutics market during the forecast period.
- The anti-inflammatory medications segment dominated the market in 2025, approximately 45 %, and is projected to grow at a substantial CAGR during the forecast period.
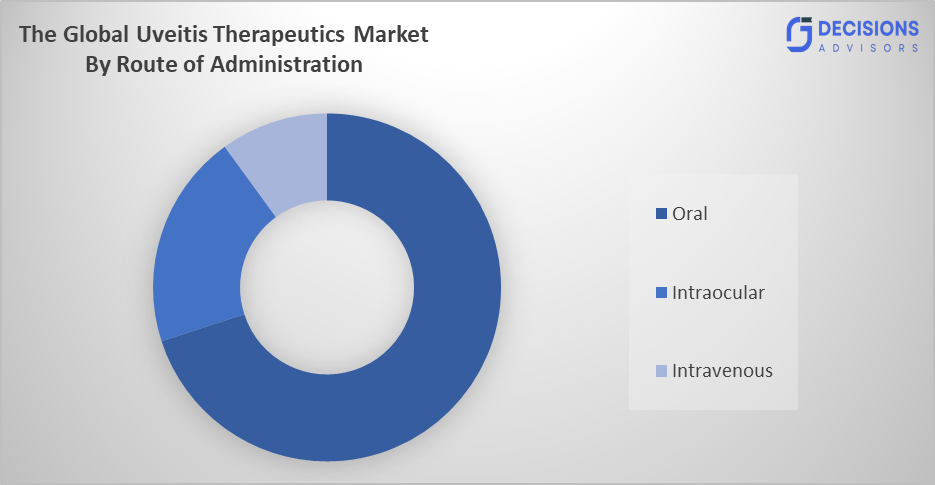
- The oral segment dominated the market in 2025, approximately 40%, and is projected to grow at a substantial CAGR during the forecast period.
- The hospital pharmacies segment dominated the market in 2025, approximately 50%, and is projected to grow at a substantial CAGR during the forecast period.
- The compound annual growth rate of the Global Uveitis Therapeutics Market is 6.64%.
- The market is likely to achieve a valuation of USD 53771 Billion by 2035.
What is role of technology in grooming the market?
Technology plays a crucial role in shaping and accelerating the growth of the Global Uveitis Therapeutics Market by improving diagnosis, treatment precision, and patient management. Advanced imaging technologies such as optical coherence tomography (OCT) and AI-assisted retinal analysis enable earlier and more accurate detection of uveitis, allowing timely therapeutic intervention. Artificial intelligence and machine learning are also being used to predict disease progression and personalize treatment strategies. In therapeutics, biotechnology advancements have led to the development of biologics, monoclonal antibodies, and targeted immunomodulators that offer higher efficacy and fewer side effects compared to traditional therapies. Digital health platforms and tele-ophthalmology improve access to specialist care, especially in remote regions. Additionally, smart drug delivery systems, including sustained-release implants, enhance patient compliance. Overall, technology is transforming the market by improving clinical outcomes, reducing relapse rates, and expanding global access to advanced uveitis care.
Market Drivers
The Global Uveitis Therapeutics Market experiences growth because of major factors, which include rising autoimmune and inflammatory eye disorder cases, the expanding senior demographic, and the increasing rates of infectious diseases which cause herpes and tuberculosis-related uveitis. The development of biologics and corticosteroid implants, together with targeted immunotherapy treatments, has brought substantial improvements to treatment results, which are now being accepted by more patients. The enhanced medical imaging technology, together with artificial intelligence screening solutions, enables healthcare professionals to identify eye conditions at an earlier stage, which leads to more patients receiving medical treatment.
Restrain
The Global Uveitis Therapeutics Market faces restraints such as high treatment costs of biologics and advanced therapies, limited accessibility in low-income regions, and delayed diagnosis due to lack of awareness. Additionally, side effects of long-term corticosteroid use and stringent regulatory approval processes further hinder market expansion and adoption rates.
Competitive Analysis:
The report offers the appropriate analysis of the key organizations/companies involved within the global uveitis therapeutics market, along with a comparative evaluation primarily based on their product offerings, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborative analysis focusing on the current news and developments of the companies, which includes product development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
Top Companies in Global Uveitis Therapeutics Market
- AbbVie Inc.
- Novartis AG
- Pfizer Inc.
- Roche Holding AG
- Johnson & Johnson (Janssen Pharmaceuticals)
- Bausch Health Companies Inc.
- Santen Pharmaceutical Co., Ltd.
- Alcon Inc.
- Regeneron Pharmaceuticals, Inc.
- EyePoint Pharmaceuticals, Inc.
- Alimera Sciences, Inc.
- Clearside Biomedical, Inc.
Government Initiatives
|
Country |
Key Government Initiatives |
|
USA |
The National Eye Institute (NEI) continues to fund "lab-to-bedside" research, supporting Phase III trials for monoclonal antibodies and novel therapies like sirolimus for non-infectious uveitis. While Congress proposed a funding reduction for the CDC's Vision Health Initiative in FY2026, existing programs like the National Eye Health Education Program (NEHEP) remain critical for public awareness and early detection. |
|
UK |
The National Health Service (NHS) and NICE continue to expand the use of monoclonal antibodies (such as adalimumab and infliximab) as standard-of-care for refractory uveitis cases. Government-funded research increasingly focuses on steroid-sparing options and the integration of digital health tools for remote patient monitoring. |
|
Japan |
The Pharmaceuticals and Medical Devices Agency (PMDA) has formally adopted ICH guidelines for generic pharmaceuticals, aiming to harmonize quality standards and reduce "drug lag" for affordable uveitis treatments. The PMDA also launched "Early Consideration" systems in 2026 to foster innovation in pediatric and rare disease therapies. |
Study on the Supply, Demand, Distribution, and Market Environment of the Uveitis Therapeutics Market
The Global Uveitis Therapeutics Market is shaped by a complex interaction of rising demand, constrained supply of advanced therapies, and evolving distribution systems. On the demand side, increasing prevalence of autoimmune diseases, infectious eye conditions, and aging populations has significantly raised the need for effective uveitis treatments such as corticosteroids, immunosuppressants, and biologics. Early diagnosis rates are also improving, further expanding patient inflow for therapeutic interventions. On the supply side, pharmaceutical companies are heavily investing in R&D for biologics, monoclonal antibodies, and sustained-release drug delivery systems. However, supply remains limited in terms of affordability and accessibility, especially for advanced therapies like biologics, which require high-cost manufacturing and specialized infrastructure. In terms of distribution, hospital pharmacies dominate due to the need for specialized administration of injectable and biologic drugs, while retail and online pharmacies are growing rapidly for oral and topical medications. The distribution network is increasingly integrated with digital healthcare platforms. The market environment is highly competitive and moderately consolidated, with leading players such as AbbVie, Novartis, Pfizer, and Bausch Health driving innovation. Strong regulatory frameworks, rising healthcare investments, and expanding ophthalmology infrastructure are supporting market growth, while high treatment costs and diagnostic delays remain key challenges globally.
Price Analysis and Consumer Behaviour Analysis
The price structure of the Global Uveitis Therapeutics Market is highly influenced by the dominance of biologics and advanced immunosuppressive therapies, which are significantly more expensive than conventional corticosteroids. High treatment costs of monoclonal antibodies, sustained-release implants, and targeted immunomodulators contribute to a premium pricing environment. In developed regions, strong insurance coverage and reimbursement policies help maintain steady demand despite high prices, while in emerging economies, affordability remains a key barrier, increasing reliance on generic corticosteroids and off-label treatments. Consumer behaviour is shifting toward long-term disease management and biologic-based precision therapies due to better clinical outcomes and reduced recurrence rates. Patients and healthcare providers increasingly prefer treatments with fewer side effects and improved adherence, such as implantable drug delivery systems. Additionally, growing awareness of early diagnosis is driving higher treatment uptake. Overall, pricing sensitivity varies widely by region, while preference is moving toward innovative, effective, and convenient therapeutic solutions.
Market Segmentation
The Uveitis Therapeutics Market share is classified into drug class, route of administration, and distribution channel.
- The anti-inflammatory medications segment dominated the market in 2025, approximately 45%, and is projected to grow at a substantial CAGR during the forecast period.
Based on the drug class, the uveitis therapeutics market is divided into anti-inflammatory medications, antibiotics and antivirals, and immunosuppressive medications. Among these, the anti-inflammatory medications segment dominated the market in 2025, approximately 45%, and is projected to grow at a substantial CAGR during the forecast period. The growth of the anti-inflammatory medications segment is growing due to their first-line use in controlling ocular inflammation. Corticosteroids are widely prescribed for rapid symptom relief and effectiveness in both acute and chronic uveitis cases. Their affordability, availability, and strong clinical efficacy make them the most commonly used treatment globally.
- The oral segment dominated the market in 2025, approximately 40%, and is projected to grow at a substantial CAGR during the forecast period.
Based on the route of administration, the uveitis therapeutics market is divided into oral, intraocular, and intravenous. Among these, the oral segment dominated the market in 2025, approximately 40%, and is projected to grow at a substantial CAGR during the forecast period. The oral segment dominated due to ease of administration and high patient compliance. Oral corticosteroids and immunosuppressants are widely used for systemic control of inflammation in moderate to severe uveitis. This non-invasive method is preferred in long-term therapy and widely accessible across healthcare systems.
- The hospital pharmacies drugs segment dominated the market in 2025, approximately 50%, and is projected to grow at a substantial CAGR during the forecast period.
Based on the distribution channel, the uveitis therapeutics market is divided into hospital pharmacies, retail pharmacies, and online pharmacies. Among these, the hospital pharmacies segment dominated the market in 2025, approximately 50%, and is projected to grow at a substantial CAGR during the forecast period. The growth of the hospital pharmacies segment is growing due to uveitis treatments often require specialist supervision and advanced drug administration, including biologics and intraocular injections. Hospitals serve as primary treatment centers for diagnosis and acute care, ensuring controlled dispensing, better patient monitoring, and higher adoption of prescription-based advanced therapeutics.
Strategies to Implement for Growth of the Market in Non-Leading Regions
To drive growth of the Global Uveitis Therapeutics Market in non-leading regions, companies should focus on improving access to affordable treatment options through tiered pricing and generic biologics. Expanding local manufacturing and distribution networks can reduce supply chain costs and increase drug availability in emerging markets. Strengthening healthcare infrastructure and ophthalmology screening programs will help in early diagnosis and timely treatment adoption. Strategic partnerships with regional healthcare providers, governments, and NGOs can enhance awareness and patient reach. Additionally, investing in physician training programs on advanced uveitis management will improve treatment adoption rates. Clinical trial expansion in underserved regions can also support regulatory approvals and market entry. Digital health platforms and tele-ophthalmology services can further bridge accessibility gaps. Collectively, these strategies will help accelerate market penetration, improve patient outcomes, and support sustainable growth in non-leading regions of the global uveitis therapeutics market.
Regional Segment Analysis of the Uveitis Therapeutics Market
- North America (U.S., Canada, Mexico)
- Europe (Germany, France, U.K., Italy, Spain, Rest of Europe)
- Asia-Pacific (China, Japan, India, Rest of APAC)
- South America (Brazil and the Rest of South America)
- The Middle East and Africa (UAE, South Africa, Rest of MEA)
North America is anticipated to hold the largest share of approximately 40% the uveitis therapeutics market over the forecast period.
North America is anticipated to hold the largest share of approximately 40% the uveitis therapeutics market over the forecast period. The dominance of this region is primarily driven by high disease awareness, advanced healthcare infrastructure, strong reimbursement policies, and early adoption of biologics. Presence of major pharmaceutical companies further strengthens regional market leadership and treatment accessibility.
Asia Pacific is expected to grow at a rapid CAGR of approximately 7% in the uveitis therapeutics market during the forecast period.
Asia Pacific is expected to grow at a rapid CAGR of approximately 7% in the uveitis therapeutics market during the forecast period. This growth is driven by rising prevalence of autoimmune diseases, improving ophthalmology infrastructure, increasing healthcare expenditure, and expanding access to advanced therapies. Growing awareness, large patient population, and government healthcare initiatives are further accelerating market expansion across emerging economies.
Europe is the 3rd largest region to grow in the uveitis therapeutics market during the period.
Europe is the third-largest region in the uveitis therapeutics market due to its well-established healthcare systems, strong ophthalmology research infrastructure, and high adoption of advanced biologic therapies. However, growth is moderate compared to North America and Asia-Pacific because of slower drug approval processes, pricing pressures, and stringent regulatory frameworks.
Future Market Trends in Global Uveitis Therapeutics Market: -
1. Rising Adoption of Biologic Therapies
The increasing use of biologics such as TNF inhibitors and monoclonal antibodies is a key trend in the Global Uveitis Therapeutics Market. These therapies offer targeted inflammation control, improved efficacy, and reduced relapse rates compared to steroids. Growing clinical success and approvals are driving widespread adoption globally.
2. Growth of Sustained-Release Drug Delivery Systems
Sustained-release implants and intravitreal drug delivery systems are gaining popularity as they reduce the need for frequent dosing and improve patient compliance. These innovations provide long-term inflammation control with fewer side effects, making them highly preferred in chronic uveitis management across developed and emerging healthcare markets.
3. Integration of AI and Precision Medicine
Artificial intelligence and precision medicine are transforming uveitis care through early diagnosis, predictive analytics, and personalized treatment planning. AI-based imaging tools help detect disease progression faster, while precision therapies enable customized treatment approaches, improving outcomes and reducing unnecessary drug exposure in patients globally.
Recent Development
- In January 2026, Novaliq received FDA Investigational New Drug (IND) clearance for NOV05, enabling advancement into Phase II clinical trials for non-infectious anterior uveitis. The therapy, a steroid-free tacrolimus eye drop, aimed to address unmet needs in treating intraocular inflammation.
- In December 2025, ANI Pharmaceuticals formed the FutureVision Advisory Council, bringing together retina and uveitis experts to guide the strategic advancement of its ophthalmology and retina franchise. The initiative aimed to strengthen innovation, product development, and long-term growth in ocular disease treatments.
- In September 2024, ANI Pharmaceuticals completed the acquisition of Alimera Sciences, strengthening its rare disease portfolio with products including ILUVIEN and YUTIQ, while supporting growth of Purified Cortrophin Gel PFS in ophthalmology and inflammatory conditions.
- In June 2024, Aldeyra Therapeutics completed enrollment in a Phase III clinical trial of reproxalap, a RASP inhibitor, for ocular inflammatory conditions, with plans to support a potential New Drug Application (NDA) submission following positive trial outcomes.
How is Recent Developments Helping the Market?
Recent developments in the Global Uveitis Therapeutics Market are significantly enhancing treatment outcomes and expanding market growth. Advances in biologic therapies, including TNF inhibitors and interleukin blockers, have improved disease control in refractory uveitis cases. The introduction of long-acting corticosteroid implants has reduced dosing frequency and improved patient compliance. Additionally, novel JAK inhibitors and targeted immunomodulatory agents are showing promising results in clinical trials. Improved diagnostic technologies, including AI-assisted imaging, are enabling earlier and more accurate detection of uveitis, supporting timely intervention. Increased research funding and expanding clinical pipelines are accelerating drug approvals. Furthermore, the shift toward personalized medicine is allowing tailored treatment approaches, reducing relapse rates. Collectively, these innovations are driving higher adoption of advanced therapeutics, boosting market expansion, and improving overall patient quality of life globally now significantly.
Market Segment
This study forecasts revenue at global, regional, and country levels from 2020 to 2035. Decision Advisors has segmented the uveitis therapeutics market based on the below-mentioned segments:
Global Uveitis Therapeutics Market, Drug Class
- Anti-inflammatory Medications
- Antibiotics and Antivirals
- Immunosuppressive Medications
Global Uveitis Therapeutics Market, By Route of Administration
- Oral
- Intraocular
- Intravenous
Global Uveitis Therapeutics Market, By Distribution Channel
- Hospital Pharmacies
- Retail Pharmacies
- Online Pharmacies
Global Uveitis Therapeutics Market, By Regional Analysis
- North America
- US
- Canada
- Mexico
- Europe
- Germany
- UK
- France
- Italy
- Spain
- Russia
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- Australia
- Rest of Asia Pacific
- South America
- Brazil
- Argentina
- Rest of South America
- Middle East & Africa
- UAE
- Saudi Arabia
- Qatar
- South Africa
- Rest of the Middle East & Africa
Frequently Asked Questions (FAQ)
Q What are the key unmet needs in the Global Uveitis Therapeutics Market?
A. The market still faces unmet needs such as limited access to affordable biologics, lack of curative treatments, and challenges in managing chronic and recurrent uveitis. There is also a need for safer long-term therapies with fewer side effects, as well as improved diagnostic capabilities in low-resource settings.
Q. How do reimbursement policies impact the uveitis therapeutics market?
A. Reimbursement policies play a critical role in market expansion, especially for high-cost biologics and advanced therapies. Favorable insurance coverage in developed regions increases patient access and treatment adoption, while limited reimbursement in emerging markets restricts the use of innovative but expensive therapies.
Q. What role do clinical trials play in shaping the uveitis therapeutics market?
A. Clinical trials are essential for introducing novel therapies such as JAK inhibitors and next-generation biologics. They help validate safety and efficacy, accelerate regulatory approvals, and expand treatment options. Increasing global clinical trial activity is also encouraging innovation and competitive differentiation among key players.
Q. How is patient adherence influencing treatment choices in uveitis?
A. Patient adherence is a key factor influencing therapy selection, with a growing preference for long-acting and less frequent dosing options. Treatments such as sustained-release implants and biologics improve compliance, reduce relapse rates, and enhance overall disease management, making them increasingly favored by both patients and clinicians.
Check Licence
Choose the plan that fits you best: Single User, Multi-User, or Enterprise solutions tailored for your needs.
We Have You Covered
- 24/7 Analyst Support
- Clients Across the Globe
- Tailored Insights
- Technology Tracking
- Competitive Intelligence
- Custom Research
- Syndicated Market Studies
- Market Overview
- Market Segmentation
- Growth Drivers
- Market Opportunities
- Regulatory Insights
- Innovation & Sustainability
Report Details
| Scope | Global |
| Pages | 240 |
| Delivery | PDF & Excel via Email |
| Language | English |
| Release | Apr 2026 |
| Access | Download from this page |
