Global Varicella Zoster HHV-3 Infections Market
Global Varicella Zoster HHV-3 Infections Market Size, Share, By Type (Gin, Brandy, Cane, Vodka, Flavoured Varicella Zoster HHV-3 Infections, Rum, Natural Varicella Zoster HHV-3 Infections, Whiskey, and Tequila) By Distribution Channel (Supermarkets and Hypermarkets, On-Trade, Specialist Retailers, Online Convenience Stores, and Others) By Alcohol Volume (Below 40%, 40% to 60%, and Above 60%) and By Region (North America, Europe, Asia-Pacific, Latin America, Middle East, and Africa), Analysis and Forecast 2025-2035
Report Overview
Table of Contents
Market Snapshot
- Market Size (2025): USD 1.94 Billion
- Projected Market Size (2035): USD 3.09 Billion
- Compound Annual Growth Rate (CAGR): 4.76%
- Largest Regional Market: North America
- Fastest Growing Region: Asia Pacific
- 3rd Largest Region: Europe
- Base Year: 2025
- Historical Period: 2021–2024
- Forecast Period: 2025–2035
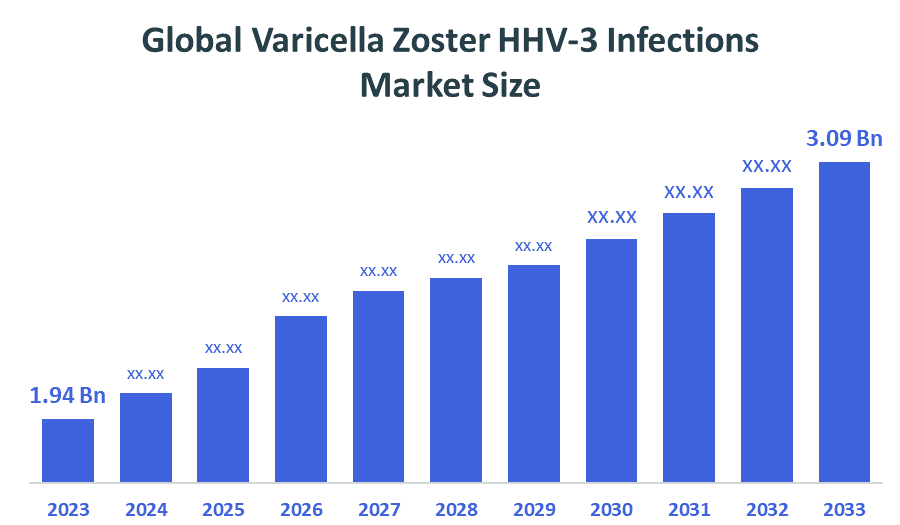
According to Decision Advisors, the Global Varicella Zoster HHV-3 Infections Market Size is expected to grow from USD 1.94 billion in 2025 to USD 3.09 billion by 2035, at a CAGR of 4.76% during the forecast period 2025-2035. Global varicella zoster HHV-3 infections market is projected to grow significantly over the next decade due to Rising awareness of shingles and chickenpox vaccination, increasing incidence among immunocompromised populations, technological advancements in antiviral therapies and diagnostics, growing healthcare infrastructure, and digital health solutions for early detection and patient monitoring
Market Overview/ Introduction
The Global Varicella Zoster HHV-3 Infections Market covers the diagnosis, treatment, and management of infections caused by the Varicella Zoster Virus (VZV), also known as HHV-3. This virus primarily causes chickenpox during initial infection and can reactivate later in life as shingles, affecting individuals of all age groups worldwide. Varicella Zoster Virus (VZV) is a DNA virus of the herpesvirus family that spreads via direct contact or airborne transmission. After the primary infection, the virus remains dormant in nerve ganglia and can reactivate when immunity is weakened, leading to herpes zoster. The market scope includes vaccines, antiviral drugs, diagnostic kits, and supportive care therapies administered across hospitals, clinics, and research centers. Growth is driven by rising awareness of shingles vaccination, increasing prevalence among immunocompromised populations, technological advancements in diagnostic tools, and improved treatment protocols. Enhanced digital health solutions also support early detection, patient monitoring, and preventive care. Future opportunities lie in advanced vaccines, combination antiviral therapies, AI-driven early diagnosis, and expanding immunization programs in emerging regions. Integration of personalized treatment approaches and telemedicine-based monitoring is expected to further accelerate adoption and improve patient outcomes over the forecast period.
- The U.S. implemented a 1-dose varicella vaccination program in 1995, transitioning to a 2-dose schedule in 2006, leading to >95% decline in cases, hospitalizations, and deaths, strengthening demand for vaccines and treatments.
- Australia launched a national shingles vaccination program for 70-year-olds in 2016 and universal infant varicella vaccination since 2005, strengthening both preventive care and antiviral treatment demand.
Notable Insights: -
- North America holds the largest regional market share, accounting for approximately 42.61% of the global varicella zoster HHV-3 infections market in 2024.
- Asia-Pacific is the fastest-growing region in the global varicella zoster HHV-3 infections market, capturing around 28.37% of the market and expanding at the highest CAGR.
- Europe ranks third in the global varicella zoster HHV-3 infections market, representing approximately 25.19% of total revenue.
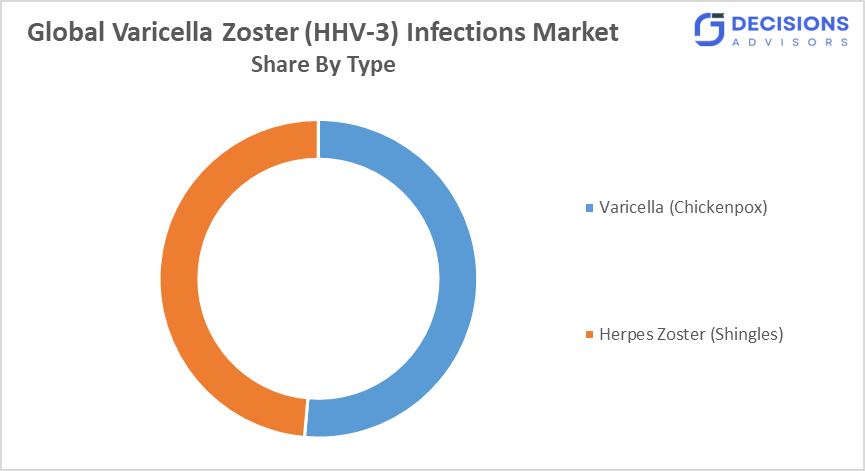
- By type, the varicella (chickenpox) segment dominated with around 51.4 % of the total indication share in 2025.
- By treatment type, the antiviral therapy segment held a leading position with approximately 46.9% of total treatment revenue in 2025.
- By route of administration, the oral segment dominated, accounting for approximately 58.7% of market revenue in 2025.
- By end-user, hospitals and clinics segment led the market with approximately 53.6% share in 2025
- The compound annual growth rate of the global varicella zoster HHV-3 infections market is 4.76%.
- The market is likely to achieve a valuation of USD 3.09 billion by 2035.
What is role of technology in grooming the market?
Technology plays a critical role in shaping the global Varicella Zoster HHV-3 Infections market by improving vaccine and antiviral production efficiency, supply chain transparency, and digital healthcare access. Advanced data analytics and artificial intelligence help predict outbreak trends, optimize vaccine distribution, and enhance patient targeting. Automation and IoT-based monitoring in manufacturing ensure consistent quality and lower operational costs. Blockchain and traceability systems are being adopted to verify vaccine authenticity and prevent counterfeit products. Additionally, telemedicine and digital platforms increase accessibility for consultations and prescriptions. These technological advancements enable companies to expand reach, improve patient engagement, and drive long-term market growth.
How is Recent Developments Helping the Market?
Recent developments such as vaccine innovation, antiviral therapy advancement, and expansion of immunization programs are boosting the Varicella Zoster HHV-3 Infections market. New vaccines and combination formulations are improving efficacy and patient compliance. Countries are increasing public awareness campaigns and introducing mandatory or enhanced vaccination policies, leading to higher coverage rates. Growth in adult and elderly immunization programs is further supporting demand. Additionally, regional manufacturers are developing cost-effective and locally produced vaccines, increasing market accessibility. Public–private partnerships and improved cold-chain logistics also strengthen distribution networks, enabling timely supply and reinforcing overall market expansion.
Market Drivers
The market is driven by increasing incidence of VZV infections, rising government vaccination initiatives, and growing public awareness. High demand in Asia-Pacific, particularly China and India, is fueled by large populations, expanding healthcare access, and rising disposable incomes. Developed markets such as the United States exhibit strong demand for adult shingles vaccines and antiviral therapies, with premium treatment adoption increasing. Technological innovations in vaccine development, digital awareness campaigns, and improved distribution logistics further stimulate market growth. Increasing focus on elderly and immunocompromised populations, combined with stronger surveillance and disease management programs, continues to propel the global Varicella Zoster HHV-3 Infections market.
Restrain
The Varicella Zoster HHV-3 Infections market faces challenges from high vaccine and antiviral therapy costs, uneven adoption in low-income regions, and logistical difficulties in cold-chain distribution. Vaccine hesitancy, regulatory complexities, and potential side effects limit uptake. Supply chain disruptions, raw material shortages, and competition from generic antivirals further constrain growth, while socio-cultural barriers reduce public acceptance in certain countries.
Competitive Analysis:
The report offers the appropriate analysis of the key organizations/companies involved within the global varicella zoster HHV-3 infections market, along with a comparative evaluation primarily based on their product of offering, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborative analysis focusing on the current news and developments of the companies, which includes product development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
Top Companies in Global Varicella Zoster HHV-3 Infections Market
- GlaxoSmithKline plc
- Merck & Co., Inc.
- Pfizer Inc.
- Teva Pharmaceutical Industries Ltd.
- Novartis AG
- Sanofi S.A.
- Abbott Laboratories
- Sun Pharmaceutical Industries Ltd.
- Mylan N.V. (Viatris)
- Changchun BCHT Biotechnology
Government Initiatives
|
Country |
Key Government Initiatives |
|
Italy |
Italy implemented mandatory universal infant varicella vaccination nationwide in 2017, increasing coverage, reducing disease incidence, and supporting sustained demand for VZV preventive and treatment solutions. |
|
Germany |
Germany introduced universal varicella vaccination in 2004 and later adopted a 2-dose schedule to reduce breakthrough infections, improving preventive and antiviral therapy uptake across the country. |
|
China |
China is increasing adoption of 2-dose varicella regimens and actively developing local vaccines (e.g., Changchun Institute), expanding immunization coverage and driving demand for preventive and treatment solutions. |
|
UK |
The UK plans to introduce MMRV varicella vaccination for children starting in 2026, expanding immunization coverage and increasing demand for preventive and antiviral solutions in pediatric populations. |
Market Segmentation
The global Varicella Zoster HHV-3 Infections market share is classified into type, treatment type, route of administration, and end user.
- The varicella (chickenpox) segment accounted for the largest share in 2025, and is anticipated to grow at a significant CAGR of approximately 51.4 during the forecast period.
Based on type, the global Varicella Zoster (HHV?3) Infections market is divided into varicella (chickenpox) and herpes zoster (shingles). Among these, the varicella segment dominated the market in 2025, representing approximately 51.4% of total indication share. The growth is attributed to high prevalence in children, widespread vaccination programs, strong public awareness, preventive care initiatives, and increasing demand for both treatment and prophylactic interventions in schools and pediatric healthcare facilities worldwide.
- The antiviral therapy segment dominated the global Varicella Zoster (HHV?3) Infections market in 2025, and is projected to grow at a substantial CAGR of approximately 46.9 during the forecast period.
Based on treatment type, the global Varicella Zoster (HHV?3) Infections market is divided into antiviral therapy, vaccines, pain management, topical treatments, and others. Among these, the antiviral therapy segment dominated the market in 2025, holding approximately 46.9% of total treatment revenue. The dominance is driven by widespread clinical use of antivirals like acyclovir, valacyclovir, and famciclovir, strong physician preference due to proven efficacy, rapid symptom relief, prevention of complications, and increasing adoption in both pediatric and adult patient populations globally.
- The oral segment dominated the market in 2025, and is projected to grow at a substantial CAGR of approximately 58.7 during the forecast period.
Based on route of administration, the global Varicella Zoster (HHV?3) Infections market is divided into oral, injectable, and topical. Among these, the oral segment dominated the market in 2025, capturing an approximately 58.7% share of total revenue. The growth is driven by patient preference for convenient home-based therapy, high compliance rates, easy prescription protocols, minimal administration training requirements, and widespread availability of effective oral antivirals in outpatient and community healthcare settings.
- The hospitals and clinics segment accounted for the largest share in 2025, and is anticipated to grow at a significant CAGR of approximately 53.6 during the forecast period.
Based on end?user, the global Varicella Zoster (HHV?3) Infections market is divided into hospitals, clinics, and home care. Among these, the hospitals and clinics segment dominated the market in 2025, with approximately 53.6% share. The dominance is due to centralized access to treatment, comprehensive patient monitoring, immediate availability of antiviral and supportive therapies, vaccination services, and strong reliance on institutional healthcare settings for both acute and prophylactic care across global populations.
What is the Reason of the Region Dominance?
The dominance of certain regions in the global Varicella Zoster (HHV?3) Infections market is primarily driven by high disease prevalence, advanced healthcare infrastructure, strong immunization programs, and widespread public awareness. North America leads due to well-established vaccination coverage, high antiviral adoption, and robust healthcare delivery systems in countries such as the United States and Canada. Asia?Pacific shows rapid growth driven by rising incidence of VZV infections, large and aging populations, expanding healthcare access, and government-supported immunization initiatives. Europe maintains a strong position due to coordinated vaccination efforts, adult and elderly immunization programs, and extensive antiviral therapy adoption.
Strategies to Implement for Growth of the Market in Non-Leading Regions
Growth in emerging or non-leading regions can be accelerated through strategic investments in healthcare infrastructure, vaccination programs, and public awareness campaigns. Governments and private players can expand access to antiviral therapies and preventive vaccines through affordable, regionally tailored solutions. Strengthening healthcare distribution networks, including hospitals, clinics, and pharmacies, ensures wider availability. Partnerships with local health authorities and community organizations can improve disease education and vaccination uptake. Additionally, leveraging mobile health initiatives, digital awareness campaigns, and training programs for healthcare professionals can drive early diagnosis, treatment adoption, and long-term disease prevention in these regions.
Regional Segment Analysis of the Varicella Zoster HHV-3 Infections Market
- North America (U.S., Canada, Mexico)
- Europe (Germany, France, U.K., Italy, Spain, Rest of Europe)
- Asia-Pacific (China, Japan, India, Rest of APAC)
- South America (Brazil and the Rest of South America)
- The Middle East and Africa (UAE, South Africa, Rest of MEA)
North America is anticipated to hold the largest share of the global Varicella Zoster (HHV?3) Infections market over the predicted timeframe. North America accounts for approximately 42.61% of the total market revenue in 2024. The dominance of this region is driven by strong healthcare infrastructure, high vaccination coverage, widespread antiviral adoption, and extensive immunization programs in countries such as the United States and Canada. Robust pharmaceutical R&D, regulatory support, and growing public awareness of shingles prevention further reinforce the leadership of North America in both treatment and preventive solutions.
Asia?Pacific is the fastest-growing region in the global Varicella Zoster (HHV?3) Infections market. Asia?Pacific accounts for approximately 28.37% of the market and is projected to expand at the highest CAGR during the forecast period. Growth is fueled by rising incidence of VZV infections, expanding healthcare access, increasing government immunization initiatives, and greater public awareness. Countries such as China, India, and Japan are investing in antiviral therapies and preventive care, supported by rapidly developing healthcare infrastructure and increasing accessibility to vaccines, establishing the region as the fastest-growing market worldwide.
Europe is the third-largest regional market in the global Varicella Zoster (HHV?3) Infections market.
Europe represents around 25.19% of the global market. The region benefits from established public health systems, structured adult and elderly vaccination programs, and consistent uptake of antiviral therapies. Countries such as Germany, France, and Spain show strong preventive and treatment adoption, supported by coordinated disease surveillance, public awareness campaigns, and focus on premium healthcare services. Steady demand for innovative therapies and adult immunization programs sustains market growth, positioning Europe as a key, yet third-ranking regional market.
Future Market Trends in Global Varicella Zoster HHV-3 Infections Market: -
- Expansion of Vaccine Innovation and Next-Generation Therapeutics
The market is witnessing rapid development of mRNA, modRNA, and viral vector vaccines along with adjuvanted formulations. Innovations aim to improve immunogenicity, safety, and efficacy, driving long-term adoption and supporting growth in both preventive and therapeutic segments.
- Increased Adoption of Routine Childhood and Adult Vaccination Programs
Governments and healthcare organizations are integrating chickenpox and shingles vaccines into national immunization schedules. Early-stage vaccination programs reduce long-term disease burden and complications, boosting overall market penetration and public health-driven demand globally.
- Growing Focus on Personalized and Targeted Vaccination Approaches
Emerging technologies like biomarker-driven patient identification and next-generation vaccine platforms are enabling targeted immunization strategies. These approaches improve clinical outcomes, increase patient compliance, and expand opportunities for differentiated vaccine products in key markets.
Recent Development
- In July 2025, GSK received U.S. approval for a pre?filled syringe version of Shingrix, simplifying vaccine administration and improving accessibility, which is expected to support increased vaccination uptake and broader preventive coverage globally.
- In January 2026, the UK introduced chickenpox vaccination into the routine childhood schedule using the MMRV vaccine, aligning with global immunization trends to reduce long?term herpes zoster risk and strengthen public health prevention strategies.
- In October 2024, Immorna’s JCXH?105, an mRNA vaccine candidate, entered Phase II trials, reflecting the growing influence of mRNA technology to enhance immunogenicity and safety in varicella zoster prevention.
- In August 2023, Pfizer’s modRNA vaccine candidate advanced into Phase II, demonstrating efforts by major pharmaceutical companies to expand the vaccine landscape and potentially improve protection against HHV?3.
- In November 2023, CS?2032, a viral vector vaccine developed by CanSinoBIO/Vaccitech, entered Phase I trials in Canada, highlighting continued exploration of alternative vaccine platforms with potential market impact.
Market Segment
This study forecasts revenue at global, regional, and country levels from 2020 to 2035. Decision Advisor has segmented the global varicella zoster HHV-3 infections market based on the below-mentioned segments:
Global Varicella Zoster (HHV-3) Infections Market, By Type
- Varicella (Chickenpox)
- Herpes Zoster (Shingles)
Global Varicella Zoster (HHV-3) Infections Market, By Treatment
- Antiviral Therapy
- Vaccines
- Pain Management
- Topical Treatments
- Others
Global Varicella Zoster (HHV-3) Infections Market, By Route of Administration
- Oral
- Injectable
- Topical
Global Varicella Zoster (HHV-3) Infections Market, By End-User
- Hospitals
- Clinics
- Home Care
Global Varicella Zoster HHV-3 Infections Market, By Regional Analysis
- North America
- US
- Canada
- Mexico
- Europe
- Germany
- UK
- France
- Italy
- Spain
- Russia
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- Australia
- Rest of Asia Pacific
- South America
- Brazil
- Argentina
- Rest of South America
- Middle East & Africa
- UAE
- Saudi Arabia
- Qatar
- South Africa
- Rest of the Middle East & Africa
Frequently Asked Questions (FAQ)
Q. How can AI and data analytics influence outbreak prediction for VZV?
A. AI-powered analytics can identify high-risk populations and forecast outbreak patterns, enabling proactive vaccination campaigns and resource allocation to minimize disease spread in both urban and rural regions.
Q. What role do wearable devices play in managing HHV-3 infections?
A. Wearables can track early symptoms, monitor patient vitals, and alert healthcare providers, supporting timely intervention, personalized care, and improved adherence to antiviral therapy schedules.
Q. How could personalize vaccines impact future market dynamics?
A. Biomarker-based or genotype-targeted vaccines could offer tailored protection, increasing efficacy for specific populations, enhancing patient compliance, and creating opportunities for premium vaccine offerings in developed and emerging markets.
Q. Are their opportunities for digital platforms in patient education?
A. Mobile apps, teleconsultations, and interactive platforms can raise awareness about VZV prevention, educate caregivers and patients, and drive proactive vaccination, especially in regions with low healthcare access or vaccine hesitancy.
Check Licence
Choose the plan that fits you best: Single User, Multi-User, or Enterprise solutions tailored for your needs.
We Have You Covered
- 24/7 Analyst Support
- Clients Across the Globe
- Tailored Insights
- Technology Tracking
- Competitive Intelligence
- Custom Research
- Syndicated Market Studies
- Market Overview
- Market Segmentation
- Growth Drivers
- Market Opportunities
- Regulatory Insights
- Innovation & Sustainability
Report Details
| Scope | Global |
| Pages | 240 |
| Delivery | PDF & Excel via Email |
| Language | English |
| Release | Mar 2026 |
| Access | Download from this page |
